Abstract
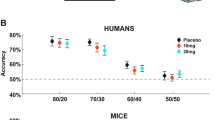
Translation of drug targets from preclinical studies to clinical trials has been aided by cross-species behavioral tasks, but evidence for brain-based engagement during task performance is still required. Cross-species progressive ratio breakpoint tasks (PRBTs) measure motivation-related behavior and are pharmacologically and clinically sensitive. We recently advanced elevated parietal alpha power as a cross-species electroencephalographic (EEG) biomarker of PRBT engagement. Given that amphetamine increases breakpoint in mice, we tested its effects on breakpoint and parietal alpha power in both humans and mice. Twenty-three healthy participants performed the PRBT with EEG after amphetamine or placebo in a double-blind design. C57BL/6J mice were trained on PRBT with EEG (n = 24) and were treated with amphetamine or vehicle. A second cohort of mice was trained on PRBT without EEG (n = 40) and was treated with amphetamine or vehicle. In humans, amphetamine increased breakpoint. In mice, during concomitant EEG, 1 mg/kg of amphetamine significantly decreased breakpoint. In cohort 2, however, 0.3 mg/kg of amphetamine increased breakpoint consistent with human findings. Increased alpha power was observed in both species as they reached breakpoint, replicating previous findings. Amphetamine did not affect alpha power in either species. Amphetamine increased effort in humans and mice. Consistent with previous reports, elevated parietal alpha power was observed in humans and mice as they performed the PRBT. Amphetamine did not affect this EEG biomarker of effort. Hence, these findings support the pharmacological predictive validity of the PRBT to measure effort in humans and mice and suggest that this EEG biomarker is not directly reflective of amphetamine-induced changes in effort.



Similar content being viewed by others
Data Availability
Data are made available upon request.
References
Amitai, N., Powell, S. B., & Young, J. W. (2019). Phencyclidine increased while isolation rearing did not affect progressive ratio responding in rats: Investigating potential models of amotivation in schizophrenia. Behavior Brain Research, 364, 413–422. https://doi.org/10.1016/j.bbr.2017.11.026
Barch, D. M., Carter, C. S., Arnsten, A., Buchanan, R. W., Cohen, J. D., Geyer, M., ..., Heinssen, R. (2009). Selecting paradigms from cognitive neuroscience for translation into use in clinical trials: Proceedings of the third CNTRICS meeting. Schizophrenia Bulletin, 35(1), 109-114. https://doi.org/10.1093/schbul/sbn163
Barkus, C., Feyder, M., Graybeal, C., Wright, T., Wiedholz, L., Izquierdo, A., ..., Holmes, A. (2012). Do GluA1 knockout mice exhibit behavioral abnormalities relevant to the negative or cognitive symptoms of schizophrenia and schizoaffective disorder? Neuropharmacology, 62(3), 1263-1272. https://doi.org/10.1016/j.neuropharm.2011.06.005
Barrett, S. P., Pihl, R. O., Benkelfat, C., Brunelle, C., Young, S. N., & Leyton, M. (2008). The role of dopamine in alcohol self-administration in humans: individual differences. Eur Neuropsychopharmacology, 18(6), 439–447. https://doi.org/10.1016/j.euroneuro.2008.01.008
Bensadoun, J. C., Brooks, S. P., & Dunnett, S. B. (2004). Free operant and discrete trial performance of mice in the nine-hole box apparatus: validation using amphetamine and scopolamine. Psychopharmacology (Berlin), 174(3), 396–405. https://doi.org/10.1007/s00213-003-1751-0
Berlin, I., Givry-Steiner, L., Lecrubier, Y., & Puech, A. J. (1998). Measures of anhedonia and hedonic responses to sucrose in depressive and schizophrenic patients in comparison with healthy subjects. European Psychiatry, 13(6), 303–309. https://doi.org/10.1016/S0924-9338(98)80048-5
Bhakta, S. G., Cavanagh, J. F., Talledo, J. A., Kotz, J. E., Benster, L., Roberts, B. Z., ..., Young, J. W. (2022). EEG reveals that dextroamphetamine improves cognitive control through multiple processes in healthy participants. Neuropsychopharmacology, 47(5), 1029-1036. https://doi.org/10.1038/s41386-021-01257-2
Bismark, A. W., Thomas, M. L., Tarasenko, M., Shiluk, A. L., Rackelmann, S. Y., Young, J. W., & Light, G. A. (2017). Relationship between effortful motivation and neurocognition in schizophrenia. Schizophrenia Research, https://doi.org/10.1016/j.schres.2017.06.042.epub ahead of print.
Carter, C. S., & Barch, D. M. (2007). Cognitive neuroscience-based approaches to measuring and improving treatment effects on cognition in schizophrenia: the CNTRICS initiative. Schizophrenia Bulletin, 33(5), 1131–1137. https://doi.org/10.1093/schbul/sbm081
Cavanagh, J. F., Gregg, D., Light, G. A., Olguin, S. L., Sharp, R. F., Bismark, A. W., ..., Young, J. W. (2021). Electrophysiological biomarkers of behavioral dimensions from cross-species paradigms. Translational Psychiatry, 11(1), 482. https://doi.org/10.1038/s41398-021-01562-w
Cavanagh, J. F., Olguin, S. L., Talledo, J. A., Kotz, J. E., Roberts, B. Z., Nungaray, J. A., ..., Brigman, J. L. (2022). Amphetamine alters an EEG marker of reward processing in humans and mice. Psychopharmacology (Berlin), 239(3), 923-933. https://doi.org/10.1007/s00213-022-06082-z
Chou, H. H., Talledo, J. A., Lamb, S. N., Thompson, W. K., & Swerdlow, N. R. (2013). Amphetamine effects on MATRICS Consensus Cognitive Battery performance in healthy adults. Psychopharmacology (Berlin), 227(1), 165–176. https://doi.org/10.1007/s00213-012-2948-x
Cilia, J., Piper, D. C., Upton, N., & Hagan, J. J. (2001). Clozapine enhances breakpoint in common marmosets responding on a progressive ratio schedule. Psychopharmacology (Berlin), 155(2), 135–143. https://doi.org/10.1007/s002130100682
Cocker, P. J., Hosking, J. G., Benoit, J., & Winstanley, C. A. (2012). Sensitivity to cognitive effort mediates psychostimulant effects on a novel rodent cost/benefit decision-making task. Neuropsychopharmacology, 37(8), 1825–1837. https://doi.org/10.1038/npp.2012.30
Cooper, Z. D., Foltin, R. W., & Evans, S. M. (2013). Effects of menstrual cycle phase on cocaine self-administration in rhesus macaques. Hormone Behavior, 63(1), 105–113. https://doi.org/10.1016/j.yhbeh.2012.10.008
Cuthbert, B. N. (2014). The RDoC framework: facilitating transition from ICD/DSM to dimensional approaches that integrate neuroscience and psychopathology. World Psychiatry, 13(1), 28–35. https://doi.org/10.1002/wps.20087
Distler, M. G., Opal, M. D., Dulawa, S. C., & Palmer, A. A. (2012). Assessment of behaviors modeling aspects of schizophrenia in Csmd1 mutant mice. PLoS One, 7(12), e51235. https://doi.org/10.1371/journal.pone.0051235
Drew, M. R., Simpson, E. H., Kellendonk, C., Herzberg, W. G., Lipatova, O., Fairhurst, S., ..., Balsam, P. D. (2007). Transient overexpression of striatal D2 receptors impairs operant motivation and interval timing. Journal of Neuroscience, 27(29), 7731-7739. https://doi.org/10.1523/JNEUROSCI.1736-07.2007
First, M. B., Spitzer, R. L., Gibbon, M., & Williams, J. B. W. (2002). Structured clinical interview for DSM-IV-TR axis I disorders, research version, non-patient edition. (SCID-I/NP): Biometrics Research. New York: New York State Psychiatric Institute.
Green, M. F., Horan, W. P., Barch, D. M., & Gold, J. M. (2015). Effort-Based Decision Making: A Novel Approach for Assessing Motivation in Schizophrenia. Schizophrenia Bulletin, 41(5), 1035–1044. https://doi.org/10.1093/schbul/sbv071
Heath, C. J., Bussey, T. J., & Saksida, L. M. (2015). Motivational assessment of mice using the touchscreen operant testing system: effects of dopaminergic drugs. Psychopharmacology (Berlin), 232(21–22), 4043–4057. https://doi.org/10.1007/s00213-015-4009-8
Heath, C. J., O'Callaghan, C., Mason, S. L., Phillips, B. U., Saksida, L. M., Robbins, T. W., ..., Sahakian, B. J. (2019). A Touchscreen Motivation Assessment Evaluated in Huntington's Disease Patients and R6/1 Model Mice. Frontiers in Neurology, 10, 858. https://doi.org/10.3389/fneur.2019.00858
Hershenberg, R., Satterthwaite, T. D., Daldal, A., Katchmar, N., Moore, T. M., Kable, J. W., & Wolf, D. H. (2016). Diminished effort on a progressive ratio task in both unipolar and bipolar depression. Journal of Affective Disorder, 196, 97–100. https://doi.org/10.1016/j.jad.2016.02.003
Higley, A. E., Kiefer, S. W., Li, X., Gaal, J., Xi, Z. X., & Gardner, E. L. (2011). Dopamine D(3) receptor antagonist SB-277011A inhibits methamphetamine self-administration and methamphetamine-induced reinstatement of drug-seeking in rats. European Journal of Pharmacology, 659(2–3), 187–192. https://doi.org/10.1016/j.ejphar.2011.02.046
Horan, W. P., Reddy, L. F., Barch, D. M., Buchanan, R. W., Dunayevich, E., Gold, J. M., ..., Green, M. F. (2015). Effort-based decision-making paradigms for clinical trials in schizophrenia: Part 2-External Validity and Correlates. Schizophrenia Bulletin, 41(5), 1055-1065. https://doi.org/10.1093/schbul/sbv090
Karlsson, R. M., Tanaka, K., Saksida, L. M., Bussey, T. J., Heilig, M., & Holmes, A. (2009). Assessment of glutamate transporter GLAST (EAAT1)-deficient mice for phenotypes relevant to the negative and executive/cognitive symptoms of schizophrenia. Neuropsychopharmacology, 34(6), 1578–1589. https://doi.org/10.1038/npp.2008.215
Kirkpatrick, B., Fenton, W. S., Carpenter, W. T., Jr., & Marder, S. R. (2006). The NIMH-MATRICS consensus statement on negative symptoms. Schizophrenia Bulletin, 32(2), 214–219. Retrieved from http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=16481659
Klawohn, J., Joyner, K., Santopetro, N., Brush, C. J., & Hajcak, G. (2022). Depression reduces neural correlates of reward salience with increasing effort over the course of the progressive ratio task. Journal of Affective Disorders, 307, 294–300. https://doi.org/10.1016/j.jad.2022.03.051
Klimesch, W., Doppelmayr, M., Russegger, H., Pachinger, T., & Schwaiger, J. (1998). Induced alpha band power changes in the human EEG and attention. Neuroscience Letter, 244(2), 73–76. https://doi.org/10.1016/s0304-3940(98)00122-0
Laughren, T., & Levin, R. (2011). Food and Drug Administration commentary on methodological issues in negative symptom trials. Schizophrenia Bulletin, 37(2), 255–256. https://doi.org/10.1093/schbul/sbq162
MacQueen, D. A., Minassian, A., Kenton, J. A., Geyer, M. A., Perry, W., Brigman, J. L., & Young, J. W. (2018). Amphetamine improves mouse and human attention in the 5-choice continuous performance test. Neuropharmacology, 138, 87–96. https://doi.org/10.1016/j.neuropharm.2018.05.034
Marder, S. R., Daniel, D. G., Alphs, L., Awad, A. G., & Keefe, R. S. (2011). Methodological issues in negative symptom trials. Schizophrenia Bulletin, 37(2), 250–254. https://doi.org/10.1093/schbul/sbq161
Markou, A., Paterson, N. E., & Semenova, S. (2004). Role of gamma-aminobutyric acid (GABA) and metabotropic glutamate receptors in nicotine reinforcement: potential pharmacotherapies for smoking cessation. Annals of the New York Academy of Sciences, 1025, 491–503. https://doi.org/10.1196/annals.1316.061
Markou, A., Salamone, J. D., Bussey, T. J., Mar, A. C., Brunner, D., Gilmour, G., & Balsam, P. (2013). Measuring reinforcement learning and motivation constructs in experimental animals: relevance to the negative symptoms of schizophrenia. Neuroscience Biobehavior Review, 37(9(Pt B)), 2149–2165. https://doi.org/10.1016/j.neubiorev.2013.08.007
Milienne-Petiot, M., Groenink, L., Minassian, A., & Young, J. W. (2017). Blockade of dopamine D(1)-family receptors attenuates the mania-like hyperactive, risk-preferring, and high motivation behavioral profile of mice with low dopamine transporter levels. Journal of Psychopharmacology, 31(10), 1334–1346. https://doi.org/10.1177/0269881117731162
Milienne-Petiot, M., Kesby, J. P., Graves, M., van Enkhuizen, J., Semenova, S., Minassian, A., ..., Young, J. W. (2017). The effects of reduced dopamine transporter function and chronic lithium on motivation, probabilistic learning, and neurochemistry in mice: Modeling bipolar mania. Neuropharmacology, 113(Pt A), 260-270. https://doi.org/10.1016/j.neuropharm.2016.07.030
Orio, L., Edwards, S., George, O., Parsons, L. H., & Koob, G. F. (2009). A role for the endocannabinoid system in the increased motivation for cocaine in extended-access conditions. Journal of Neuroscience, 29(15), 4846–4857. https://doi.org/10.1523/JNEUROSCI.0563-09.2009
Paterson, N. E., Froestl, W., & Markou, A. (2004). The GABAB receptor agonists baclofen and CGP44532 decreased nicotine self-administration in the rat. Psychopharmacology (Berlin), 172(2), 179–186. https://doi.org/10.1007/s00213-003-1637-1
Reddy, L. F., Waltz, J. A., Green, M. F., Wynn, J. K., & Horan, W. P. (2016). Probabilistic reversal learning in schizophrenia: stability of deficits and potential causal mechanisms. Schizophrenia Bulletin, 42(4), 942–951. https://doi.org/10.1093/schbul/sbv226
Reddy, L. F., Horan, W. P., Barch, D. M., Buchanan, R. W., Dunayevich, E., Gold, J. M., ..., Green, M. F. (2015). Effort-based decision-making paradigms for clinical trials in schizophrenia: Part 1-Psychometric Characteristics of 5 Paradigms. Schizophrenia Bulletin, 41(5), 1045-1054. https://doi.org/10.1093/schbul/sbv089
Romoli, B., Lozada, A. F., Sandoval, I. M., Manfredsson, F. P., Hnasko, T. S., Berg, D. K., & Dulcis, D. (2019). Neonatal Nicotine Exposure Primes Midbrain Neurons to a Dopaminergic Phenotype and Increases Adult Drug Consumption. Biological Psychiatry, 86(5), 344–355. https://doi.org/10.1016/j.biopsych.2019.04.019
Ryu, I. S., Kim, O. H., Kim, J. S., Sohn, S., Choe, E. S., Lim, R. N., ..., Jang, E. Y. (2021). Effects of beta-Phenylethylamine on Psychomotor, Rewarding, and Reinforcing Behaviors and Affective State: The Role of Dopamine D1 Receptors. International Journal of Molecular Science, 22(17). https://doi.org/10.3390/ijms22179485
Stoops, W. W., Lile, J. A., Fillmore, M. T., Glaser, P. E., & Rush, C. R. (2005). Reinforcing effects of modafinil: influence of dose and behavioral demands following drug administration. Psychopharmacology (Berl), 182(1), 186–193. https://doi.org/10.1007/s00213-005-0044-1
Swerdlow, N. R., Platten, A., Hanlon, F. M., Martinez, Z. A., Printz, M. P., & Auerbach, P. (2003). Sensitivity to sensorimotor gating-disruptive effects of apomorphine in two outbred parental rat strains and their F1 and N2 progeny. Neuropsychopharmacology, 28(2), 226–234. Retrieved from http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=12589375
Vardigan, J. D., Huszar, S. L., McNaughton, C. H., Hutson, P. H., & Uslaner, J. M. (2010). MK-801 produces a deficit in sucrose preference that is reversed by clozapine, D-serine, and the metabotropic glutamate 5 receptor positive allosteric modulator CDPPB: relevance to negative symptoms associated with schizophrenia? Pharmacology, Biochemistry, & Behavior, 95(2), 223–229. https://doi.org/10.1016/j.pbb.2010.01.010
Wardle, M. C., Treadway, M. T., Mayo, L. M., Zald, D. H., & de Wit, H. (2011). Amping up effort: effects of d-amphetamine on human effort-based decision-making. Journal of Neuroscience, 31(46), 16597–16602. https://doi.org/10.1523/JNEUROSCI.4387-11.2011
White, E. J., Nacke, M., Akeman, E., Cannon, M. J., Mayeli, A., Touthang, J., ..., Aupperle, R. L. (2021). P300 amplitude during a monetary incentive delay task predicts future therapy completion in individuals with major depressive disorder. Journal of Affect Disorder, 295, 873-882. https://doi.org/10.1016/j.jad.2021.08.106
Wolf, D. H., Satterthwaite, T. D., Kantrowitz, J. J., Katchmar, N., Vandekar, L., Elliott, M. A., & Ruparel, K. (2014). Amotivation in schizophrenia: Integrated assessment with behavioral, clinical, and imaging measures. Schizophrenia Bulletin.https://doi.org/10.1093/schbul/sbu026
Young, J. W. (2023). Development of cross-species translational paradigms for psychiatric research in the Research Domain Criteria era. Neuroscience Biobehavioral Review, 148, 105119. https://doi.org/10.1016/j.neubiorev.2023.105119
Young, J. W., & Geyer, M. A. (2010). Action of Modafinil-Increased Motivation Via the Dopamine Transporter Inhibition and D1 Receptors? Biological Psychiatry, 67(8), 784–787. https://doi.org/10.1016/j.biopsych.2009.12.015
Young, J. W., & Geyer, M. A. (2013). Evaluating the role of the alpha-7 nicotinic acetylcholine receptor in the pathophysiology and treatment of schizophrenia. Biochemical Pharmacology, 86(8), 1122–1132. https://doi.org/10.1016/j.bcp.2013.06.031
Young, J. W., & Light, G. A. (2018). Cross-Species Neurophysiological Biomarkers of Attentional Dysfunction in Schizophrenia: Bridging the Translational Gap. Neuropsychopharmacology, 43(1), 230–231. https://doi.org/10.1038/npp.2017.218
Young, J. W., Zhou, X., & Geyer, M. A. (2010). Animal models of schizophrenia. Current Topics in Behaviorial Neuroscience, 4, 391–433. https://doi.org/10.1007/7854_2010_62
Young, J. W., Meves, J. M., Tarantino, I. S., Caldwell, S., & Geyer, M. A. (2011). Delayed procedural learning in alpha7-nicotinic acetylcholine receptor knockout mice. Genes Brain Behavior, 10(7), 720–733. https://doi.org/10.1111/j.1601-183X.2011.00711.x
Young, J., & W., Kamenski, M., Higa, K., Light, G., A., Geyer, M., A., Zhou X. (2015). GlyT-1 inhibition attenuates the attentional but not learning or motivational deficits of the Sp4-hypomorphic mouse model relevant to psychiatric disorders. Neuropsychopharmacology, 40(12), 2715–26. https://doi.org/10.1038/npp.2015.120
Acknowledgments
The authors thank Dr. Mark A. Geyer, Ms. Mahalah R. Buell, and Mr. Richard F. Sharp for their support on these studies.
Funding
These studies were funded by NIMH UH3MG109168.
Author information
Authors and Affiliations
Contributions
MN: Manuscript writing, Data Curation, Data Analysis, Investigation.
SGB: Methodology, Investigation, Data curation, Supervision, Project Administration, Editing.
JAT: Investigation, Data curation, Supervision, Project Administration.
JEK: Investigation, Data curation, Supervision, Project Administration.
LB: Investigation, Data Curation.
BZR: Investigation, Data curation, Supervision, Project Administration.
JAN: Investigation, Data curation.
GAL: Conceptualization, Methodology, Resources, Writing— Review & Editing, Funding acquisition.
NRS: Conceptualization, Methodology, Writing—Review & Editing, Investigation, Data curation, Medical Monitoring of Test Subjects, Supervision, Project Administration, Funding acquisition.
JLB: Conceptualization, Methodology, Resources, Writing— Review & Editing, Funding acquisition
JFC: Conceptualization, Methodology, Software, Formal analysis, Writing, Funding acquisition.
JWY: Conceptualization, Methodology, Writing—Review & Editing, Funding acquisition.
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Open Practices
The data and materials for all experiments are available upon request.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Noback, M., Bhakta, S.G., Talledo, J.A. et al. Amphetamine increases motivation of humans and mice as measured by breakpoint, but does not affect an Electroencephalographic biomarker. Cogn Affect Behav Neurosci 24, 269–278 (2024). https://doi.org/10.3758/s13415-023-01150-z
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.3758/s13415-023-01150-z