Summary
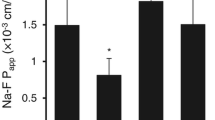
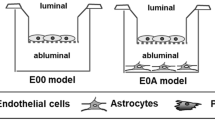
Lipids of brain tissue and brain microvascular endothelial cells contain high proportions of long-chain polyunsaturated fatty acids (long PUFAs). The blood-brain barrier (BBB) is formed by the brain endothelial cells under the inductive influence of brain cells, especially perivascular glia, and coculture of endothelial cells and glial cells has been used to examine this induction. The objective of this study was to investigate whether C6 glioma cells are able to influence the lipid composition and shift the fatty acid (FA) patterns of the BBB model cell lines RBE4 and ECV304 toward the in vivo situation. Lipid classes of the three cell lines were analyzed by thin-layer chromatography and lipid FA patterns by high-performance liquid chromatography. Only ECV304 cells showed altered lipid composition in coculture, with C6 cells. The fractions of triglycerides and cholesteryl esters (depending on the support filter) were about twice as high in coculture as when the cells were grown alone. Triglyceride fractions reached 13 to 15% of total lipids in coculture. The three cell lines showed an increase in the percentage of long PUFAs with respect to unsaturated FAs, mainly because of an increase in the percentages of arachidonic acid, all cis-7,10,13,16-docosatetraenoic, acid, and all cis-7,10,13,16,19-docosapentaenoic acid. It is concluded that glioma C6 cells are able to induce a more in vivo-like FA pattern in BBB cell culture models. However, changes were not significant for the individual PUFAs, and their levels did not reach in vivo values.
Similar content being viewed by others
References
Alberghina, M. Fatty acid and lipid intermediate transport. In: Pardridge, W. M., ed. Introduction to the blood-brain barrier. Cambridge: Cambridge University Press; 1998:227–237.
Begley, D. J.; Bradbury, M. W.; Kreuter, J., eds. The blood-brain barrier and drug delivery to the CNS. New York: Dekker; 2000.
Begley, D. J.; Lechardeur, D.; Chen, Z.-D., et al. Functional expression of P-glycoprotein in an immortalised cell line of rat brain endothelial cells, RBE4. J. Neurochem. 67:988–995; 1996.
Bénistant, C.; Dehouck, M. P.; Fruchart, J.-C., et al. Fatty acid composition of brain capillary endothelial cells: effect of the conculture with astrocytes. J. Lipid Res. 36:2311–2319; 1995.
Bernoud, N.; Fenart, L.; Bénistant, C., et al. Astrocytes are mainly responsible for the polyunsaturated fatty acid enrichment in blood-brain barrier endothelial cells in vitro. J. Lipid Res. 39:1816–1824; 1998.
Bourre, J.-M. Lipid composition of brain microvessels. In: Pardridge W. M., ed. Introduction to the blood-brain barrier. Cambridge: Cambridge University Press; 1998:308–313.
Braun, A.; Hämmerle, S.; Suda, K., et al. Cell cultures as tools in biopharmacy. Eur. J. Pharm. Sci. 11:S51-S60; 2000.
Buckland, A. G.; Wilton, D. C. Antonic phospholipids, interfacial binding and the regulation of cell functions. Biochim. Biophys. Acta 1483:199–216; 2000.
Chishty, M.; Reichel, A.; Begley, D. J.; Abbott, N. J. Glial induction of blood-brain barrier like L-system amino acid transport in the ECV304 cell line. Glia 39:99–104; 2002.
Delton-Vandenbroucke, I.; Grammas, P.; Anderson, R. E. Polyunsaturated fatty acid metabolism in retinal and cerebral microvascular endothelial cells. J. Lipid Res. 38:147–159; 1997.
Dirks, W. G.; Macleod, R. A. F.; Drexler, H. G. ECV304 (endothelial) is really T24 (bladder carcinoma): cell line cross-contamination at source. In Vitro Cell. Dev. Biol. 35A:558–559; 1999.
Dolman, D. E. M.; Anderson, P.; Rollinson, C.; Abbott, N. J. Characterisation of a new in vitro model of the blood-brain barrier (BBB). J. Physiol. 505:56P-57P; 1997.
Easton, A. S.; Abbott, N. J. Bradykinin increases permeability by calcium and 5-lipoxygenase in the ECV304/C6 cell culture model of the blood-brain barrier. Brain Res. 953:157–169; 2002.
Findlay, J. B. C.; Evans, W. H. Biological membranes: a practical approach. Oxford: IRL Press; 1987.
Homayoun, P.; Durand, G.; Pascal, G.; Bourre, J. M. Alteration in fatty acid composition of adult rat brain capillaries and choroid plexus induced by a diet deficient in n-3 fatty acids: slow recovery after substitution with a nondeficient diet. J. Neurochem. 51:45–48; 1988.
Hunter, G. W.; Negash, S.; Squier, T. C. Phosphatidylethanolamine modulates Ca-ATPase function and dynamics. Biochemistry 38:1356–1364; 1999.
Hurst, R. D.; Fritz, I. B. Properties of an immortalised vascular endothelial/glioma cell co-culture model of the blood-brain barrier. J. Cell. Physiol. 167:81–88; 1996.
Issandou, M.; Grand-Perret, T. Multidrug resistance P-glycoprotein is not involved in cholesterol esterification. Biochem. Biophys. Res. Commun. 279:369–377; 2000.
Krämer, S. D.; Abbott, N. J.; Begley, D. J. Biological models to predict blood-brain barrier permeation. In: Testa, B.; van de Waterbeemd, H.; Folkers, G.; Guy, R., ed. Pharmacokinetic optimization in drug research: biological physicochemical and computational strategies. Weinheim, Germany: VCH Verlag; 2001.
Krämer, S. D.; Hurley, J. A.; Abbott, N. J.; Begley, D. J. Lipids in blood-brain barrier models in vitro I: thin-layer chromatography and high-performance liquid chromatography for the analysis of lipid classes and long-chain polyunsaturated fatty acids. In Vitro Cell. Dev. Biol. 38A:557–565; 2002.
Lerique, B.; Lepetit-Thevenim, J.; Verine, A., et al. Triacylglycerol in biomembranes. Life Sci. 54:831–840; 1994.
Luker, G. D.; Nilsson, K. R.; Covey, D. F.; Piwnica-Worms, D. Multidrug resistance (MDR1) P-glycoprotein enhances esterification of plasma membrane cholesterol. J. Biol. Chem. 274:6979–6991; 1999.
Marsh, D. Handbook of lipid bilayers. Boston, MA: CRC Press; 1990.
Moore, S. A.; Yoder, E.; Murphy, S., et al. Astrocytes, not neurons, produce docosahexaenoic acid (22∶6 omega-3) and arachidonic acid (20∶4 omega-6). J. Neurochem. 56:518–524; 1991.
Moore, S. A.; Yoder, E.; Spector, A. A. Role of the blood-brain barrier in the formation of long-chain omega-3 and omega-6 fatty acids from essential fatty acid precursors. J. Neurochem. 55:391–402; 1990.
Mukherjee, S.; Ghosh, R. N.; Maxfield, F. R. Endocytosis. Physiol. Rev. 77:759–803; 1997.
O'Brien, J. S.; Sampson, E. L. Fatty acid and aldehyde composition of the major brain lipids in normal gray matter, white matter and myelin. J. Lipid Res. 6:545–552; 1965.
Rist, R. J.; Romero, I. A.; Chart, M. W. K., et al. F-actin cytoskeleton and sucrose permeability of immortalised rat brain microvascular endothelial cell monolayers: effects of cyclic AMP and astrocytic factors. Brain Res. 768:10–18; 1997.
Romsicki, Y.; Sharom, J. The membrane lipid environment modulates drug interactions with the P-glycoprotein multidrug transporter. Biochemistry 38:6887–6896; 1999.
Rubin, L. L.; Staddon, J. M. The cell biology of the blood-brain barrier. Annu. Rev. Neurosci. 22:11–28; 1999.
Scism, J. L.; Laska, D. A.; Horn, J. W., et al. Evaluation of an in vitro coculture model for the blood-brain barrier: comparison of human umbilical vein endothelial cells (ECV304) and rat glioma cells (C6) from two commercial sources. In Vitro Cell. Dev. Biol. 35A:580–595; 1999.
Shand, J. H.; Noble, R. C. Quantification of lipid mass by a liquid scintillation counting procedure following charring on thin-layer plates. Anal. Biochem. 101:427–434; 1980.
Spector, A. A. Essentially of fatty acids. Lipids 34:S1-S3; 1999.
Spector, A. A.; Mathur, S. N.; Kaduce, T. L.; Hyman, B. T. Lipid nutrition and metabolism of cultured mammalian cells. Progr. Lipid Res. 19:155–186; 1980.
Stein, O.; Stein, Y. Bovine aortic endothelial cells display macrophage-like properties towards acetylated 125I-labelled low density lipoprotein. Biochim. Biophys. Acta 620:631–635; 1980.
Suda, K.; Rothen-Rutishauser, B.; Günthert, M.; Wunderli-Allenspach, H. Phenotypical characterisation of ECV304 and T24 cells: endothelial versus epithelial features. In Vitro Cell. Dev. Biol. 37A:505–514; 2000.
Tan, K. H.; Dobbie, M. S.; Felix, R. A., et al. A comparison of the induction of immortalized endothelial cell impermeability by astrocytes. Neuroreport 12:1329–1334; 2001.
Tayarani, I.; Chaudiere, J.; Lefauconnier, J.-M.; Bourre, J.-M. Enzymatic protection against peroxidative damage in isolated brain capillaries. J. Neurochem. 48:1399–1402; 1987.
van't Hof, W.; Malik, A.; Vijayakumar, S., et al. The effect of apical and basolateral lipids on the function of the band 3 anion exchange protein. J. Cell Biol. 139:941–949; 1997.
Williard, D. E.; Harmon, S. D.; Kaduce, T. L., et al. Docosahexaenoic acid synthesis from n−3 polyunsaturated fatty acids in differentiated rat brain astrocytes. J. Lipid Res. 42:1368–1376; 2001.
Wunderli-Allenspach, H. Methodologies in cell culture. In: Testa, B.; van de Waterbeemd, H.; Folkers, G.; Guy, R., ed. Pharmacokinetic optimization in drug research: biological, physicochemical and computational strategies. New York: VCH Verlagsgesellschaft mbH; 2001.
Yamaguchi, M.; Matsunaga, R.; Fukuda, K., et al. Highly sensitive determination of free polyunsaturated, long-chain fatty acids in human serum by high-performance liquid chromatography with fluorescence detection. Anal. Biochem. 155:256–261; 1986.
Youdim, K. A.; Dobbie, M. S.; Kuhnle, G., et al. Interaction between flavonoids and the blood-brain barrier; in vitro studies. J. Neurochem. 85:180–192; 2003.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Krämer, S.D., Schütz, Y.B., Wunderli-Allenspach, H. et al. Lipids in blood-brain barrier models in vitro II: Influence of glial cells on lipid classes and lipid fatty acids. In Vitro Cell.Dev.Biol.-Animal 38, 566–571 (2002). https://doi.org/10.1290/1543-706X(2002)38<566:LIBBMI>2.0.CO;2
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1290/1543-706X(2002)38<566:LIBBMI>2.0.CO;2