Abstract
Background
Preoperative chemotherapy/chemoradiotherapy has been generally considered for the treatment of esophageal squamous cell carcinoma (ESCC) to improve prognosis. We examined the effects of anticancer drugs on the expression of kallikrein-related peptidase 13 (KLK13), a potential ESCC prognostic marker, and its clinical relevance in patients who received chemotherapy/chemoradiotherapy for ESCC.
Methods
Overall, 105 patients with ESCC who received chemotherapy or chemoradiotherapy before esophagectomy were enrolled. The expression of KLK13 in biopsy samples obtained before chemotherapy/chemoradiotherapy and resected ESCC tumors was assessed by immunohistochemical staining. The effects of 5-fluorouracil (5-FU) and/or cisplatin (CDDP) exposure on the expressions of KLK13 and ten-eleven translocation dioxygenases (TET) in ESCC cells were examined by reverse transcription-polymerase chain reaction.
Results
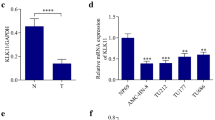
Immunohistochemical staining of paired ESCC specimens before (biopsy samples) and after (resected specimens) chemotherapy/chemoradiotherapy demonstrated a change in KLK13 expression. KLK13 and TET2/3 transcriptions were induced when human ESCC cell lines were treated with 5-FU and/or CDDP. Among patients with KLK13-negative status before chemotherapy/chemoradiotherapy, those with KLK13-positive resected tumors had a significantly poorer prognosis than those with KLK13-negative resected tumors (p = 0.0477). By using tumor cells isolated from ESCC biopsy tissues obtained before chemotherapy/chemoradiotherapy, we established a primary culture system and detected the induction of KLK13 expression by anticancer drugs.
Conclusions
Preoperative treatments alter KLK13 expression in ESCC. The conversion of KLK13 expression from a negative status in biopsy samples to a positive status in resected tumor samples is a predictor of poor prognosis. KLK13 status is a potential marker for decision making to avoid harmful chemotherapy/chemoradiotherapy in patients with ESCC.





Similar content being viewed by others
References
Lu CL, Lang HC, Luo JC, et al. Increasing trend of the incidence of esophageal squamous cell carcinoma, but not adenocarcinoma, in taiwan. Cancer Causes Control. 2010;21(2):269–74.
Fujita H. Ways and tradition of Japan in esophageal surgery for cancer. Gen Thorac Cardiovasc Surg. 2020;68(10):1187–92.
Then EO, Lopez M, Saleem S, et al. Esophageal cancer: an updated surveillance epidemiology and end results database analysis. World J Oncol. 2020;11(2):55–64.
Miyata H, Sugimura K, Motoori M, et al. Clinical implications of conversion surgery after induction therapy for T4b thoracic esophageal squamous cell carcinoma. Ann Surg Oncol. 2019;26(13):4737–43.
Natsugoe S, Okumura H, Matsumoto M, et al. The role of salvage surgery for recurrence of esophageal squamous cell cancer. Eur J Surg Oncol. 2006;32(5):544–7.
Zhang M, Hu D, Wang S, Qin C. Clinicopathologic significance of HMGA2 expression’s correlation with prognosis of esophageal squamous cell carcinoma after Ivor Lewis esophagectomy. Minerva Chir. 2016;71(4):239–44.
Nakamura K, Kato K, Igaki H, et al. Three-arm phase III trial comparing cisplatin plus 5-FU (CF) versus docetaxel, cisplatin plus 5-FU (DCF) versus radiotherapy with CF (CF-RT) as preoperative therapy for locally advanced esophageal cancer (JCOG1109, NExT study). Jpn J Clin Oncol. 2013;43(7):752–5.
Ando N, Kato H, Igaki H, et al. A randomized trial comparing postoperative adjuvant chemotherapy with cisplatin and 5-fluorouracil versus preoperative chemotherapy for localized advanced squamous cell carcinoma of the thoracic esophagus (JCOG9907). Ann Surg Oncol. 2012;19(1):68–74.
Shapiro J, van Lanschot JJB, Hulshof MCCM, et al. Neoadjuvant chemoradiotherapy plus surgery versus surgery alone for oesophageal or junctional cancer (CROSS): long-term results of a randomised controlled trial. Lancet Oncol. 2015;16(9):1090–8.
van Hagen P, Hulshof MC, van Lanschot JJ, et al. Preoperative chemoradiotherapy for esophageal or junctional cancer. N Engl J Med. 2012;366(22):2074–84.
Otsubo T, Yamada K, Hagiwara T, et al. DNA hypermethyation and silencing of PITX1 correlated with advanced stage and poor postoperative prognosis of esophageal squamous cell carcinoma. Oncotarget. 2017;8(48):84434–48.
Nohara K, Yamada K, Yamada L, et al. Expression of kallikrein-related peptidase 13 is associated with poor prognosis in esophageal squamous cell carcinoma. Gen Thorac Cardiovasc Surg. 2018;66(6):351–7.
Maeda O, Ando T, Ohmiya N, et al. Alteration of gene expression and DNA methylation in drug-resistant gastric cancer. Oncol Rep. 2014;31(4):1883–90.
Florou D, Mavridis K, Scorilas A. The kallikrein-related peptidase 13 (KLK13) gene is substantially up-regulated after exposure of gastric cancer cells to antineoplastic agents. Tumour Biol. 2012;33(6):2069–78.
Ito S, Shen L, Dai Q, et al. Tet proteins can convert 5-methylcytosine to 5-formylcytosine and 5-carboxylcytosine. Science. 2011;333(6047):1300–3. https://doi.org/10.1126/science.1210597.
Brierley JD, Gospodarowicz MK, Wittekind C. TNM classification of malignant tumours. Hoboken: Wiley; 2017.
Eisenhauer EA, Therasse P, Bogaerts J, et al. New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1). Eur J Cancer. 2009;45(2):228–47.
Japan Esophageal Society. Japanese classification of esophageal cancer, 11th edition: part II and III. Esophagus. 2017;14(1):37–65.
Kang KA, Piao MJ, Kim KC, et al. Epigenetic modification of Nrf2 in 5-fluorouracil-resistant colon cancer cells: involvement of TET-dependent DNA demethylation. Cell Death Dis. 2014;5(4):e1183. https://doi.org/10.1038/cddis.2014.149.
Chou RH, Lin SC, Wen HC, Wu CW, Chang WS. Epigenetic activation of human kallikrein 13 enhances malignancy of lung adenocarcinoma by promoting N-cadherin expression and laminin degradation. Biochem Biophys Res Commun. 2011;409(3):442–7.
Zhou K, Guo H, Zhang J, et al. Potential role of TET2 in gastric cancer cisplatin resistance. Pathol Res Pract. 2019;215(11):152637. https://doi.org/10.1016/j.prp.2019.152637.
Dekker E, Tanis PJ, Vleugels JLA, Kasi PM, Wallace MB. Colorectal cancer. Lancet. 2019;394(10207):1467–80.
Acknowledgment
The authors thank Drs. Miwa Tamura-Nakano and Chinatsu Oyama in the NCGM EM Support Unit for their technical support for histological analysis. They would also like to thank Enago for the English language review, and Ms. Yasuko Nozaki for her technical assistance. This work was supported by JSPS KAKENHI Grant Numbers JP15K10124, JP19K08457, and by grants from the National Center for Global Health and Medicine (26-117, 29-1019, 20A1017 and 20A3002).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosures
Akira Shimomura, Teruki Hagiwara, Kazuhiko Yamada, Chizu Yokoi, Masayoshi Terayama, Kyoko Nohara, Toru Igari, and Yuki I. Kawamura have no financial or other relations that could lead to a commercial interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Shimomura, A., Hagiwara, T., Yamada, K. et al. Induction of Kallikrein-Related Peptidase 13 and TET2/3 by Anticancer Drugs and Poor Prognosis of Patients with Esophageal Squamous Cell Carcinoma After Preoperative Treatment. Ann Surg Oncol 31, 251–261 (2024). https://doi.org/10.1245/s10434-023-14364-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-023-14364-9