Abstract
Background
The role of viral hepatitis status in post-hepatectomy outcomes has yet to be delineated. This large, multicentred contemporary study aimed to evaluate the effect of viral hepatitis status on 30-day post-hepatectomy complications in patients treated for hepatocellular carcinoma (HCC).
Methods
Patients from the National Surgical Quality Improvement Program (NSQIP) database with known viral hepatitis status, who underwent hepatectomy for HCC between 2014 and 2018, were included. Patients were classified as HBV-only, HCV-only, HBV and HCV co-infection (HBV/HCV), or no viral hepatitis (NV). Multivariable models were used to assess outcomes of interest. The primary outcome was any 30-day post-hepatectomy complication. The secondary outcomes were major complications and post-hepatectomy liver failure (PHLF). Subgroup analyses were performed for cirrhotic and noncirrhotic patients.
Results
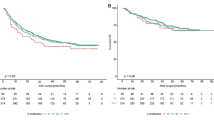
A total of 3234 patients were included. The 30-day complication rate was 207/663 (31.2%) HBV, 356/1077 (33.1%) HCV, 29/81 (35.8%) HBV/HCV, and 534/1413 (37.8%) NV (p = 0.01). On adjusted analysis, viral hepatitis status was not associated with occurrence of any 30-day post-hepatectomy complications (ref: NV, HBV odds ratio (OR) 0.89 [95% confidence interval (CI): 0.71–1.12]; HCV OR 0.91 [95% CI: 0.75–1.10]; HBV/HCV OR 1.17 [95% CI: 0.71–1.93]). Similar results were found in cirrhotic and noncirrhotic subgroups, and for secondary outcomes: occurrence of any major complications and PHLF.
Conclusions
In patients with HCC managed with resection, viral hepatitis status is not associated with 30-day post-hepatectomy complications, major complications, or PHLF compared with NV. This suggests that clinical decisions and prognostication of 30-day outcomes in this population likely should not be made based on viral hepatitis status.


Similar content being viewed by others
References
Liu Z, Xu K, Jiang Y, et al. Global trend of aetiology-based primary liver cancer incidence from 1990 to 2030: a modelling study. Int J Epidemiol. 2021;50(1):128–42. https://doi.org/10.1093/ije/dyaa196.
de Mattos AZ, Debes JD, Boonstra A, et al. Current impact of viral hepatitis on liver cancer development: The challenge remains. World J Gastroenterol. 2021;27(24):3556–67. https://doi.org/10.3748/wjg.v27.i24.3556.
Lee CW, Tsai HI, Sung CM, et al. Risk factors for early mortality after hepatectomy for hepatocellular carcinoma. Med (Baltim). 2016;95(39):e5028. https://doi.org/10.1097/MD.0000000000005028.
Zhong JH, Ke Y, Gong WF, et al. Hepatic resection associated with good survival for selected patients with intermediate and advanced-stage hepatocellular carcinoma. Ann Surg. 2014;260(2):329–40. https://doi.org/10.1097/SLA.0000000000000236.
Belghiti J, Hiramatsu K, Benoist S, Massault P, Sauvanet A, Farges O. Seven hundred forty-seven hepatectomies in the 1990s: an update to evaluate the actual risk of liver resection. J Am Coll Surg. 2000;191(1):38–46. https://doi.org/10.1016/s1072-7515(00)00261-1.
Spolverato G, Ejaz A, Kim Y, et al. Patterns of care among patients undergoing hepatic resection: a query of the National Surgical Quality Improvement Program-targeted hepatectomy database. J Surg Res. 2015;196(2):221–8. https://doi.org/10.1016/j.jss.2015.02.016.
Utsunomiya T, Shimada M, Kudo M, et al. A comparison of the surgical outcomes among patients with HBV-positive, HCV-positive, and non-B non-C hepatocellular carcinoma: a nationwide study of 11,950 patients. Ann Surg. 2015;261(3):513–20. https://doi.org/10.1097/SLA.0000000000000821.
Wu CC, Ho WL, Chen JT, Tang JS, Yeh DC, P’Eng FK. Hepatitis viral status in patients undergoing liver resection for hepatocellular carcinoma. Br J Surg. 1999;86(11):1391–6. https://doi.org/10.1046/j.1365-2168.1999.01272.x.
Jia HD, Liang L, Li C, et al. Long-term surgical outcomes of liver resection for hepatocellular carcinoma in patients with HBV and HCV co-infection: a multicenter observational study. Front Oncol. 2021;11:700228. https://doi.org/10.3389/fonc.2021.700228.
Lee JJ, Kim PT, Fischer S, et al. Impact of viral hepatitis on outcomes after liver resection for hepatocellular carcinoma: results from a North American center. Ann Surg Oncol. 2014;21(8):2708–16. https://doi.org/10.1245/s10434-014-3609-6.
Kao WY, Su CW, Chau GY, Lui WY, Wu CW, Wu JC. A comparison of prognosis between patients with hepatitis B and C virus-related hepatocellular carcinoma undergoing resection surgery. World J Surg. 2011;35(4):858–67. https://doi.org/10.1007/s00268-010-0928-z.
Kabir T, Syn NL, Tan ZZX, et al. Predictors of post-operative complications after surgical resection of hepatocellular carcinoma and their prognostic effects on outcome and survival: a propensity-score matched and structural equation modelling study. Eur J Surg Oncol. 2020;46(9):1756–65. https://doi.org/10.1016/j.ejso.2020.03.219.
Lai Y, Lee JC, Hung HC, et al. Models to predict disease-free survival for hepatocellular carcinoma patients with surgical resections. J Surg Oncol. 2020;122(7):1444–52. https://doi.org/10.1002/jso.26169.
Li Z, Zhao X, Jiang P, et al. HBV is a risk factor for poor patient prognosis after curative resection of hepatocellular carcinoma: a retrospective case-control study. Med (Baltim). 2016;95(31):e4224. https://doi.org/10.1097/MD.0000000000004224.
Panel AIHG. Hepatitis C guidance: AASLD-IDSA recommendations for testing, managing, and treating adults infected with hepatitis C virus. Hepatology. 2015;62(3):932–54. https://doi.org/10.1002/hep.27950.
Lockart I, Hajarizadeh B, Buckley N, et al. All-cause hepatocellular carcinoma survival in the era of direct-acting antiviral therapy. J Gastroenterol Hepatol. 2021;36(12):3515–23. https://doi.org/10.1111/jgh.15687.
Tabrizian P, Saberi B, Holzner ML, et al. Outcomes of transplantation for HBV- versus HCV-related HCC: impact of DAA HCV therapy in a national analysis of >20,000 patients. HPB (Oxford). 2021. https://doi.org/10.1016/j.hpb.2021.11.018.
von Elm E, Altman DG, Egger M, et al. The strengthening the reporting of observational studies in epidemiology (STROBE) statement: guidelines for reporting observational studies. Int J Surg. 2014;12(12):1495–9. https://doi.org/10.1016/j.ijsu.2014.07.013.
Ingraham AM, Richards KE, Hall BL, Ko CY. Quality improvement in surgery: the American college of surgeons national surgical quality improvement program approach. Adv Surg. 2010;44:251–67. https://doi.org/10.1016/j.yasu.2010.05.003.
Clavien PA, Barkun J, de Oliveira ML, et al. The Clavien-Dindo classification of surgical complications: five-year experience. Ann Surg. 2009;250(2):187–96. https://doi.org/10.1097/SLA.0b013e3181b13ca2.
Sood A, Meyer CP, Abdollah F, et al. Minimally invasive surgery and its impact on 30-day postoperative complications, unplanned readmissions and mortality. Br J Surg. 2017;104(10):1372–81. https://doi.org/10.1002/bjs.10561.
Gurien LA, Ra JH, Crandall M, Kerwin AJ, Tepas JJ 3rd. Clavien-Dindo analysis of NSQIP data objectively measures patient-focused quality. Am Surg. 2019;85(8):789–93.
Pathak P, Tsilimigras DI, Hyer JM, Diaz A, Pawlik TM. Timing and severity of postoperative complications and associated 30-day mortality following hepatic resection: a national surgical quality improvement project study. J Gastrointest Surg. 2022;26(2):314–22. https://doi.org/10.1007/s11605-021-05088-w.
Surgeons ACo. User guide for the 2018 ACS NSQIP participant use data file (PUF). PDF. October 2019, 2019. https://www.facs.org/-/media/files/quality-programs/nsqipnsqip_puf_userguide_2018.ashx
Ye JZ, Mai RY, Guo WX, et al. Nomogram for prediction of the international study Group of Liver Surgery (ISGLS) grade B/C Posthepatectomy liver failure in HBV-related hepatocellular carcinoma patients: an external validation and prospective application study. BMC Cancer. 2020;20(1):1036. https://doi.org/10.1186/s12885-020-07480-2.
Hoffmann K, Hinz U, Stravodimos C, et al. Risk assessment for liver resection. Surgery. 2018;164(5):998–1005. https://doi.org/10.1016/j.surg.2018.06.024.
Dhir M, Samson KK, Yepuri N, Yanala UR, Smith LM, Are C. Preoperative nomogram to predict posthepatectomy liver failure. J Surg Oncol. 2021;123(8):1750–6. https://doi.org/10.1002/jso.26463.
Etra JW, Squires MH 3rd, Fisher SB, et al. Early identification of patients at increased risk for hepatic insufficiency, complications and mortality after major hepatectomy. HPB (Oxford). 2014;16(10):875–83. https://doi.org/10.1111/hpb.12270.
FE Harrell Jr MHJ, D Hmisc. Package ‘rms.’ 2017; 299
Harrell F. Missing data. In: Regression modeling strategies. Springer; 2015. P. 45-61.
Rubin D. Multiple imputation after 18+ years. J Am Stat Assoc. 1996;91(434):473–89.
Team RC. A language and environment for statistical computing. 2021. https//www R-project org/[Google Sch
Perisetti A, Goyal H, Yendala R, Thandassery RB, Giorgakis E. Non-cirrhotic hepatocellular carcinoma in chronic viral hepatitis: Current insights and advancements. World J Gastroenterol. 2021;27(24):3466–82. https://doi.org/10.3748/wjg.v27.i24.3466.
Singal AG, Rich NE, Mehta N, et al. Direct-acting antiviral therapy for hepatitis C virus infection is associated with increased survival in patients with a history of hepatocellular carcinoma. Gastroenterology. 2019;157(5):1253-1263 e2. https://doi.org/10.1053/j.gastro.2019.07.040.
Hsu PY, Hsu CT, Yeh ML, et al. Early fibrosis but late tumor stage and worse outcomes in hepatocellular carcinoma patients without hepatitis B or hepatitis C. Dig Dis Sci. 2020;65(7):2120–9. https://doi.org/10.1007/s10620-019-05938-3.
Li Q, Li H, Qin Y, Wang PP, Hao X. Comparison of surgical outcomes for small hepatocellular carcinoma in patients with hepatitis B versus hepatitis C: a Chinese experience. J Gastroenterol Hepatol. 2007;22(11):1936–41. https://doi.org/10.1111/j.1440-1746.2006.04619.x.
Wakiyama S, Matsumoto M, Haruki K, et al. Clinical features and outcome of surgical patients with non-B non-C hepatocellular carcinoma. Anticancer Res. 2017;37(6):3207–13. https://doi.org/10.21873/anticanres.11682.
Zhang EL, Li J, Li J, Wang WQ, Gu J, Huang ZY. Sub-classification of cirrhosis affects surgical outcomes for early hepatocellular carcinoma independent of portal hypertension. Front Oncol. 2021;11:671313. https://doi.org/10.3389/fonc.2021.671313.
Shehta A, Farouk A, Fouad A, et al. Post-hepatectomy liver failure after hepatic resection for hepatocellular carcinoma: a single center experience. Langenbecks Arch Surg. 2021;406(1):87–98. https://doi.org/10.1007/s00423-020-01956-2.
Kim JM, Rhu J, Ha SY, Choi GS, Kwon CHD, Joh JW. Hepatectomy outcomes in patients with hepatitis C virus-related hepatocellular carcinoma with or without cirrhosis. Ann Surg Treat Res. 2022;102(1):1–9. https://doi.org/10.4174/astr.2022.102.1.1.
Elshaarawy O, Aman A, Zakaria HM, et al. Outcomes of curative liver resection for hepatocellular carcinoma in patients with cirrhosis. World J Gastrointest Oncol. 2021;13(5):424–39. https://doi.org/10.4251/wjgo.v13.i5.424.
Sultana A, Brooke-Smith M, Ullah S, et al. Prospective evaluation of the International Study Group for Liver Surgery definition of post hepatectomy liver failure after liver resection: an international multicentre study. HPB (Oxford). 2018;20(5):462–9. https://doi.org/10.1016/j.hpb.2017.11.007.
Aragon RJ, Solomon NL. Techniques of hepatic resection. J Gastrointest Oncol. 2012;3(1):28–40. https://doi.org/10.3978/j.issn.2078-6891.2012.006.
Acknowledgement
None.
Author information
Authors and Affiliations
Contributions
L.R.: conception of the project, literature review, data synthesis, interpretation of results, statistical analysis, and writing of the manuscript. W.C.: conception of the project, data synthesis, interpretation of results, statistical analysis, and writing of the manuscript. H.M.: conception of the project, interpretation of results, statistical analysis, and writing of the manuscript. T.I.: conception of the project, interpretation of results, statistical analysis, and writing of the manuscript. J.J.F.: conception of the project, interpretation of results, statistical analysis, and writing of the manuscript. M.P.A.W.C.: conception of the project, interpretation of results, statistical analysis, and writing of the manuscript. M.C.: data synthesis, interpretation of results, statistical analysis, and writing of the manuscript. G.S.: conception of the project, literature review, interpretation of results, statistical analysis, and writing of the manuscript.
Corresponding author
Ethics declarations
DISCLOSURES
Gonzalo Sapisochin discloses consultancy for Astra-Zeneca, Roche, Novartis, and Integra. Gonzalo Sapisochin has received financial compensation for talks for Roche, Astra-Zeneca, Chiesi, Evidera and Integra. Gonzalo Sapisochin has received a grant from Roche. Jordan Feld discloses research support from Abbvie, Gilead, GSK, Janssen, Eiger, Enanta, and Roche and consulting for Abbvie, GSK, Gilead, Roche. None of the other authors have any conflicts of interest to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Disclaimer: The American College of Surgeons National Surgical Quality Improvement Program and the hospitals participating in this program are the sources of the data used herein; they have not verified and are not responsible for the statistical validity of the data analysis or the conclusions derived by the authors.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Rajendran, L., Choi, W.J., Muaddi, H. et al. Association of Viral Hepatitis Status and Post-hepatectomy Outcomes in the Era of Direct-Acting Antivirals. Ann Surg Oncol 30, 2793–2802 (2023). https://doi.org/10.1245/s10434-022-12937-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-022-12937-8