Abstract
Background
Hepatocellular carcinoma (HCC) is highly recurrent. Cancer-associated fibroblasts (CAFs), a major component of the tumor microenvironment, promote malignancy; however, the mechanisms underlying their actions are obscure. We aimed to identify CAF-specific proteins in HCC and determine whether they could be potential therapeutic targets.
Methods
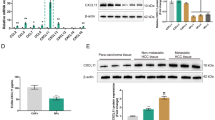
Using comprehensive proteomic analysis of CAFs and noncancerous fibroblasts (NFs) primary-cultured from resected HCC specimens from the same patients, CAF-specific proteins were identified. Immunohistochemistry for versican (VCAN) was performed on cancerous tissues obtained from 239 patients with HCC. Conditioned medium from CAFs transfected with siRNA for VCAN was analyzed in vitro.
Results
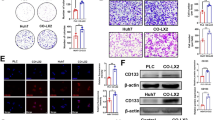
CAFs significantly promoted HCC cell proliferation, migration, and invasion (p < 0.01, 0.01, and 0.01, respectively) compared with NFs. VCAN was upregulated in CAFs, and its stromal level correlated with poor differentiation (p = 0.009) and positive vascular invasion (p = 0.003). Stromal VCAN level was also associated with significantly lower overall (p = 0.002) and relapse-free (p < 0.001) survival rates. It also independently predicted prognosis and recurrence. VCAN-knockdown CAFs significantly suppressed HCC cell migration and invasion compared with negative control.
Conclusions
VCAN secreted from CAFs promoted malignant transformation of HCC cells and has potential as a new therapeutic target in HCC.




Similar content being viewed by others
References
Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68(6):394–424.
European Association for the Study of the Liver. Electronic address EEE, European Association for the Study of the L. EASL clinical practice guidelines: management of hepatocellular carcinoma. J Hepatol. 2018;69(1):182–236.
Ioannou GN, Green PK, Berry K. HCV eradication induced by direct-acting antiviral agents reduces the risk of hepatocellular carcinoma. J Hepatol. 2018;68:25–32.
Said A, Ghufran A. Epidemic of non-alcoholic fatty liver disease and hepatocellular carcinoma. World J Clin Oncol. 2017;8(6):429–36.
Kamiyama T, Nakanishi K, Yokoo H, et al. Recurrence patterns after hepatectomy of hepatocellular carcinoma: implication of Milan criteria utilization. Ann Surg Oncol. 2009;16(6):1560–71.
Finn RS, Qin S, Ikeda M, et al. Atezolizumab plus bevacizumab in unresectable hepatocellular carcinoma. N Engl J Med. 2020;382(20):1894–905.
Quail DF, Joyce JA. Microenvironmental regulation of tumor progression and metastasis. Nat Med. 2013;19(11):1423–37.
Kalluri R, Zeisberg M. Fibroblasts in cancer. Nat Rev Cancer. 2006;6(5):392–401.
Gascard P, Tlsty TD. Carcinoma-associated fibroblasts: orchestrating the composition of malignancy. Genes Dev. 2016;30(9):1002–19.
Jena BC, Das CK, Bharadwaj D, Mandal M. Cancer associated fibroblast mediated chemoresistance: A paradigm shift in understanding the mechanism of tumor progression. Biochim Biophys Acta Rev Cancer. 2020;1874(2):188416.
Mhaidly R, Mechta-Grigoriou F. Fibroblast heterogeneity in tumor micro-environment: Role in immunosuppression and new therapies. Semin Immunol. 2020;48:101417.
Dykes SS, Hughes VS, Wiggins JM, Fasanya HO, Tanaka M, Siemann D. Stromal cells in breast cancer as a potential therapeutic target. Oncotarget. 2018;9(34):23761–79.
Sukowati CH, Anfuso B, Crocé LS, Tiribelli C. The role of multipotent cancer associated fibroblasts in hepatocarcinogenesis. BMC Cancer. 2015;15:188.
Murakami T, Takasawa A, Takasawa K, et al. Aberrant expression of junctional adhesion molecule: A contributes to the malignancy of cervical adenocarcinoma by interaction with poliovirus receptor/CD155. Cancer Sci. 2021;112(2):906–917.
Saito Y, Takasawa A, Takasawa K, et al. Aldolase A promotes epithelial-mesenchymal transition to increase malignant potentials of cervical adenocarcinoma. Cancer Sci. 2020;111(8):3071–81.
Pickup MW, Mouw JK, Weaver VM. The extracellular matrix modulates the hallmarks of cancer. EMBO Rep. 2014;15(12):1243–53.
Sahai E, Astsaturov I, Cukierman E, et al. A framework for advancing our understanding of cancer-associated fibroblasts. Nat Rev Cancer. 2020;20(3):174–86.
Iozzo RV. Matrix proteoglycans: from molecular design to cellular function. Annu Rev Biochem. 1998;67:609–52.
Papadas A, Arauz G, Cicala A, Wiesner J, Asimakopoulos F. Versican and Versican-matrikines in Cancer Progression, Inflammation, and Immunity. J Histochem Cytochem. 2020;68(12):871–85.
Hernandez D, Miquel-Serra L, Docampo MJ, Marco-Ramell A, Bassols A. Role of versican V0/V1 and CD44 in the regulation of human melanoma cell behavior. Int J Mol Med. 2011;27(2):269–75.
Papadas A, Asimakopoulos F. Versican in the Tumor Microenvironment. Adv Exp Med Biol. 2020;1272:55–72.
Bissell MJ, Hines WC. Why don’t we get more cancer? A proposed role of the microenvironment in restraining cancer progression. Nat Med. 2011;17(3):320–9.
Xia L, Huang W, Tian D, et al. Forkhead box Q1 promotes hepatocellular carcinoma metastasis by transactivating ZEB2 and VersicanV1 expression. Hepatology. 2014;59(3):958–73.
Wight TN, Kang I, Evanko SP, et al. Versican: a critical extracellular matrix regulator of immunity and inflammation. Front Immunol. 2020;11:512.
Kodama J, Hasengaowa KT, et al. Versican expression in human cervical cancer. Eur J Cancer. 2007;43(9):1460–6.
Kodama J, Hasengaowa Kusumoto T, et al. Prognostic significance of stromal versican expression in human endometrial cancer. Ann Oncol. 2007;18(2):269–74.
Kusumoto T, Kodama J, Seki N, Nakamura K, Hongo A, Hiramatsu Y. Clinical significance of syndecan-1 and versican expression in human epithelial ovarian cancer. Oncol Rep. 2010;23(4):917–25.
Voutilainen K, Anttila M, Sillanpaa S, et al. Versican in epithelial ovarian cancer: relation to hyaluronan, clinicopathologic factors and prognosis. Int J Cancer. 2003;107(3):359–64.
Said N, Sanchez-Carbayo M, Smith SC, Theodorescu D. RhoGDI2 suppresses lung metastasis in mice by reducing tumor versican expression and macrophage infiltration. J Clin Invest. 2012;122(4):1503–18.
Cox TR. The matrix in cancer. Nat Rev Cancer. 2021;21:217–238.
Liu T, Zhou L, Li D, Andl T, Zhang Y. Cancer-associated fibroblasts build and secure the tumor microenvironment. Front Cell Dev Biol. 2019;7:60.
Yeung TL, Leung CS, Wong KK, et al. TGF-beta modulates ovarian cancer invasion by upregulating CAF-derived versican in the tumor microenvironment. Cancer Res. 2013;73(16):5016–28.
Aspberg A, Miura R, Bourdoulous S, et al. The C-type lectin domains of lecticans, a family of aggregating chondroitin sulfate proteoglycans, bind tenascin-R by protein-protein interactions independent of carbohydrate moiety. Proc Natl Acad Sci U S A. 1997;94(19):10116–21.
Isogai Z, Aspberg A, Keene DR, Ono RN, Reinhardt DP, Sakai LY. Versican interacts with fibrillin-1 and links extracellular microfibrils to other connective tissue networks. J Biol Chem. 2002;277(6):4565–72.
Olin AI, Morgelin M, Sasaki T, Timpl R, Heinegard D, Aspberg A. The proteoglycans aggrecan and Versican form networks with fibulin-2 through their lectin domain binding. J Biol Chem. 2001;276(2):1253–61.
Acknowledgment
The authors thank all the patients who provided specimens for this study and the Department of Gastroenterological Surgery I at Hokkaido University Hospital for sample collection. They also thank Naoki Okada for preparing the tissue microarrays and Nozomi Kobayashi, Ayae Nange, and Asami Okumura for their technical assistance on the experiments. Thanks to Editage (www.editage.jp) for the English language review.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosures
This study was supported by the Japan Agency for Medical Research and Development (AMED) under the Grant number JP20fk0210041. The authors declare no conflict of interest
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kato, K., Fukai, M., Hatanaka, K.C. et al. Versican Secreted by Cancer-Associated Fibroblasts is a Poor Prognostic Factor in Hepatocellular Carcinoma. Ann Surg Oncol 29, 7135–7146 (2022). https://doi.org/10.1245/s10434-022-11862-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-022-11862-0