Abstract
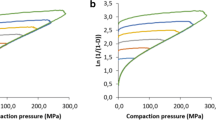
Use of higher tableting speeds is gaining increasing importance for pharmaceutical industry. There is a profound lack of new studies of mechanical properties of hypromellose, and none of them evaluate different suppliers. Thus, the objective of this study was to investigate flow and compaction properties of different grades of hypromellose (type 2208) from three different suppliers, with particular focus on the effect of the compression speed. The flow properties were determined using flow time, shear cell, Carr index, and constant B from initial part of Heckel profile. Compaction properties were quantified using “out-of-die” Heckel, Walker, and Kuentz-Leuenberger models; two tensile strength profiles (tabletability and compactibility); and elastic recovery. Compaction was performed by both an instrumented single-punch press and a high-speed rotary press simulator. Due to larger, rounder, and smoother particles, both Methocel™ DC grades together with Benecel™ K4M showed better flow properties compared with other materials, with Metolose® K100M having the worst flow. Overall, Benecel™ K100M and Metolose® K100M showed the best compaction properties, closely followed by Metolose® K4M. Heckel analysis showed the highest compressibility of Benecel™ K100M, followed by both Methocel™ DC grades. Kuentz-Leuenberger model showed to have no practical superiority in comparison with Heckel model in the compression pressure range used. Results of strain rate sensitivity showed that Methocel™ K4M DC was the least susceptible to change of tableting speed, followed by Methocel™ K100M DC and both grades of Benecel™, and in contrast, both grades of Metolose® were the most sensitive. Effect of moisture on compaction was also studied.










Similar content being viewed by others
References
Roger TL. Hypromellose. In: Rowe RC, Sheskey PJ, Quinn ME, editors. Handbook of pharmaceutical excipients. 6th ed. London: Pharmaceutical Press; 2009. p. 326–9.
Grdešič P, Vrečer F, Ilić I. Flow and compaction properties of hypromellose: new directly compressible versus the established grades. Drug Dev Ind Pharm. 2016;42(11):1877–86.
Devjak Novak S, Šporar E, Baumgartner S, Vrečer F. Characterization of physicochemical properties of hydroxypropyl methylcellulose (HPMC) type 2208 and their influence on prolonged drug release from matrix tablets. J Pharm Biomed Anal. 2012;66:136–43.
Gustafsson C, Bonferoni MC, Caramella C, Lenholm H, Nystrom C. Characterisation of particle properties and compaction behavior of hydroxypropylmethylcellulose with different deegres of methoxy/hydroxypropyl substitution. Eur J Pharm Sci. 1999;9:171–84.
Gustafsson C, Nyström C, Lennholm H, Bonferoni MC, Caramella MC. Characteristics of hydroxypropyl methylcellulose influencing compactibility and prediction of particle and tablet properties by infrared spectroscopy. J Pharm Sci. 2003;92:494–504.
Nokhodchi A, Ford JL, Rowe P, Rubinstein MH. The effects of compression rate and force on the compaction properties of different viscosity grades of hydroxypropylmethylcellulose. Int J Pharm. 1996;129:21–31.
Nokhodchi A, Ford JL, Rowe P, Rubinstein MH. The influence of moisture content on the consolidation properties of hydroxypropylmethylcellulose K4M (HPMC 2208). J Pharm Pharmacol. 1996;48:1116–21.
Nokhodchi A, Ford JL, Rubinstein MH. Studies on the interaction between water and (hydroxypropyl)methylcellulose. J Pharm Sci. 1997;86:608–15.
Malamataris S, Karidas T, Goidas P. Effect of particle size and sorbed moisture on the compression behaviour of some hydroxypropyl methylcellulose (HPMC) polymers. Int J Pharm. 1994;103:205–15.
Sinko CM, Smith DP, Nixon PR. Mechanical characterization of hydroxypropyl methylcellulose: modulus determination from indentation loading profiles. Int J Pharm. 1992;81:243–52.
European Pharmacopoeia. Edition 9, 2017.
Heckel RW. Density-pressure relationship in powder compaction. Trans Metall Soc AIME. 1961;221:671–5.
Walker EE. The properties of powder VI: the compressibility of powders. Trans Faraday Soc. 1923;19:73–82.
Sonnergaard JM. Quantifiaction of the compactibility of pharmaceutical powders. Eur J Pharm Biopharm. 2006;63:270–7.
Kuentz M, Leuenberger H. Pressure susceptibility of polymer tablets as a critical property: a modified Heckel equation. J Pharm Sci. 1999;88:174–9.
Fell JT, Newton JM. Determination of tablet strength by the diametral compression test. J Pharm Sci. 1970;59:688–91.
Armstrong NA, Haines-Nutt RF. Elastic recovery and surface area changes in compacted powder systems. J Pharm Pharmacol. 1972;24:135–6.
Malamataris S, BibBaie S, Pilpel N. Plasto-elasticity and tableting of paracetamol. J Pharm Pharmacol. 1984;36:616–7.
Sonnergaard JM. Investigation of a new mathematical model for compression of pharmaceutical powders. Eur J Pharm Sci. 2001;14:149–57.
Sonnergaard JM. A critical evaluation of the Heckel equation. Int J Pharm. 1999;193:63–71.
Ilić I, Govedarica B, Šibanc R, Dreu R, Srčič S. Deformation properties of pharmaceutical excipients determined using an in-die and out-die method. Int J Pharm. 2013;446:6–15.
Egart M, Ilić I, Janković B, Lah N, Srčič S. Compaction properties of crystalline pharmaceutical ingredients according to the Walker model and nanomechanical attributes. Int J Pharm. 2014;472:347–55.
Ilić I, Kasa P Jr, Dreu R, Pintye-Hodi K, Srčič S. The compressibility and compactibility of different types of lactose. Drug Dev Ind Pharm. 2009;35:1271–80.
Šantl M, Ilić I, Vrečer F, Baumgartner S. A compressibility and compactibility study of real tableting mixtures: the impact of wet and dry granulation versus direct tableting mixture. Int J Pharm. 2011;414:131–9.
Nokhodchi A, Ford JL, Rubinstein MH. The effect of particle size and viscosity grade on the compaction properties of K grade of hydroxypropylmethylcellulose. Int J Pharm. 1995;126:189–97.
Roberts RJ, Rowe RC. The effect of punch velocity on the compaction of a variety of materials. J Pharm Pharmacol. 1985;37(6):377–84.
Tye CK, Sun C, Amidon GE. Evaluation of the effects of tableting speed on the relationships between compaction pressure, tablet tensile strength, and tablet solid fraction. J Pharm Sci. 2004;94:465–72.
Natoli D, Liu L, Levin M, Tsygan L. Chapter 32 – development, optimization, and scale-up of process parameters: tablet compression. In: Developing Solid Oral Dosage Forms. 2009; 725–759.
Acknowledgments
The authors wish to express sincere gratitude to KRKA, d.d., Novo mesto and Faculty of Pharmacy, University of Ljubljana, for providing us the opportunity and resources to develop and complete this work. The authors would like to thank Prof. Odon Planinšek from the Faculty of Pharmacy, University of Ljubljana, for his assistance in conducting the SEM analysis.
Funding
The research was supported by Krka, d.d., Novo mesto.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Grdešič, P., Paudel, A. & German Ilić, I. High-Molecular-Weight Hypromellose from Three Different Suppliers: Effects of Compression Speed, Tableting Equipment, and Moisture on the Compaction. AAPS PharmSciTech 21, 203 (2020). https://doi.org/10.1208/s12249-020-01688-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1208/s12249-020-01688-y