Abstract
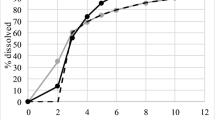
Bioequivalence (BE) is usually hard to achieve for extended-release (ER) dosage form products due to not only its complicated formulation but also to the BCS classification of the investigated drugs. Considering the difficulties in establishing full-scale IVIVC and limited in vivo pharmacokinetics data in the early stage of formulation development, we have selected BCS III drug metformin as a model drug to demonstrate a novel approach for the selection of BE formulations. Firstly, dissolution tests in both standard and biorelevant media were performed followed by identification of the most similar formulation WM to the reference product (GXR) based on principal component analysis (PCA) of the dissolution data. Then, we developed an IVIVC model using the reported GXR pharmacokinetics profiles via a convolution-based approach. Based on our established IVIVC and in vitro dissolution profiles of generic metformin ER products, we were able to predict their in vivo pharmacokinetic profiles and quantitatively compare the differences in AUC and Cmax to ensure the correct selection of BE product. Finally, the selection of WM as the BE formulation of GXR was confirmed with a pilot BE study in healthy volunteers under fasting state. Moreover, the in vivo data from the fed state study were further integrated into our IVIVC model to identify FeSSIF-V2 as the biorelevant media for WM. Our novel integrative approach of PCA with a convolution-based IVIVC was successfully adopted for the screening of the BE metformin ER formulation and such an approach could be further utilized for the effective selection of BE formulation for other drugs/formulations with complex in vivo absorption processes.







Similar content being viewed by others
References
Center for Drug Evaluation and Research. Guidance for industry: bioavailability and bioequivalence studies submitted in NDAs or INDs - General Considerations (DRAFT). US Food and Drug Administration; 2014.
Center for Drug Evaluation and Research. Guidance for industry: extended release oral dosage forms: development, evaluation, and application of in vitro/in vivo correlations. US Food and Drug Administration; 1997.
Le Guellec S, Ehrmann S, Vecellio L. In vitro - in vivo correlation of intranasal drug deposition. Adv Drug Deliv Rev. 2020. https://doi.org/10.1016/j.addr.2020.09.002.
Jacob S, Nair AB. An updated overview with simple and practical approach for developing in vitro-in vivo correlation. Drug Dev Res. 2018;79(3):97–110. https://doi.org/10.1002/ddr.21427.
Kazi M, Al Amri R, Alanazi FK, Hussain MD. In vitro methods for in vitro-in vivo correlation (IVIVC) for poorly water soluble drugs: lipid based formulation perspective. Curr Drug Deliv. 2018;15(7):918–29. https://doi.org/10.2174/1567201815666180116090910.
Choi SM, Kang CY, Lee BJ, Park JB. In vitro-in vivo correlation using in silico modeling of physiological properties, metabolites, and intestinal metabolism. Curr Drug Metab. 2017;18(11):973–82. https://doi.org/10.2174/1389200218666171031124347.
Shen J, Burgess DJ. In vitro-in vivo correlation for complex non-oral drug products: where do we stand? J Control Release. 2015;219:644–51. https://doi.org/10.1016/j.jconrel.2015.09.052.
Kollipara S, Gandhi RK. Pharmacokinetic aspects and in vitro-in vivo correlation potential for lipid-based formulations. Acta Pharm Sin B. 2014;4(5):333–49. https://doi.org/10.1016/j.apsb.2014.09.001.
Lin Z, Zhou D, Hoag S, Qiu Y. Influence of drug properties and formulation on in vitro drug release and biowaiver regulation of oral extended release dosage forms. AAPS J. 2016;18(2):333–45. https://doi.org/10.1208/s12248-015-9861-2.
Giuliani A. The application of principal component analysis to drug discovery and biomedical data. Drug Discov Today. 2017;22(7):1069–76. https://doi.org/10.1016/j.drudis.2017.01.005.
Jolliffe IT, Cadima J. Principal component analysis: a review and recent developments. Philos Trans A Math Phys Eng Sci. 2016;374(2065):20150202. https://doi.org/10.1098/rsta.2015.0202.
Bauer RJ. NONMEM tutorial part I: description of commands and options, with simple examples of population analysis. CPT Pharmacometrics Syst Pharmacol. 2019;8:525–37. https://doi.org/10.1002/psp4.12404.
Gomeni R, Bressolle-Gomeni F. Comparison of alternative population modeling approaches for implementing a level a IVIVC and for assessing the time-scaling factor using deconvolution and convolution-based methods. AAPS J. 2020;22(3):67. https://doi.org/10.1208/s12248-020-00445-0.
Gomeni R, Bressolle-Gomeni F. Deconvolution analysis by non-linear regression using a convolution-based model: comparison of nonparametric and parametric approaches. AAPS J. 2019;22(1):9. https://doi.org/10.1208/s12248-019-0389-8.
Cheng CL, Yu LX, Lee HL, Yang CY, Lue CS, Chou CH. Biowaiver extension potential to BCS class III high solubility-low permeability drugs: bridging evidence for metformin immediate-release tablet. Eur J Pharm Sci. 2004;22(4):297–304. https://doi.org/10.1016/j.ejps.2004.03.016.
Najib N, Idkaidek N, Beshtawi M, Bader M, Admour I, Alam SM, et al. Bioequivalence evaluation of two brands of metformin 500 mg tablets (Dialon & Glucophage)--in healthy human volunteers. Biopharm Drug Dispos. 2002;23(7):301–6. https://doi.org/10.1002/bdd.326.
Center for Drug Evaluation and Research. Guidance for industry: bioanalytical method validation. US Food and Drug Administration; 2018.
Di Maio S, Carrier RL. Gastrointestinal contents in fasted state and post-lipid ingestion: in vivo measurements and in vitro models for studying oral drug delivery. J Control Release. 2011;151(2):110–22. https://doi.org/10.1016/j.jconrel.2010.11.034.
Costa P, Sousa Lobo JM. Modeling and comparison of dissolution profiles. Eur J Pharm Sci. 2001;13(2):123–33. https://doi.org/10.1016/s0928-0987(01)00095-1.
Gaynor C, Dunne A, Davis J. A comparison of the prediction accuracy of two IVIVC modelling techniques. J Pharm Sci. 2008;97(8):3422–32. https://doi.org/10.1002/jps.21220.
Gillespie WR. Convolution-based approaches for in vivo-in vitro correlation modeling. Adv Exp Med Biol. 1997;423:53–65. https://doi.org/10.1007/978-1-4684-6036-0_5.
Buchwald P. Direct, differential-equation-based in-vitro-in-vivo correlation (IVIVC) method. J Pharm Pharmacol. 2003;55(4):495–504. https://doi.org/10.1211/002235702847.
Costello C, Rossenu S, Vermeulen A, Cleton A, Dunne A. A time scaling approach to develop an in vitro-in vivo correlation (IVIVC) model using a convolution-based technique. J Pharmacokinet Pharmacodyn. 2011;38(5):519–39. https://doi.org/10.1007/s10928-011-9206-4.
Margolskee A, Darwich AS, Galetin A, Rostami-Hodjegan A, Aarons L. Deconvolution and IVIVC: exploring the role of rate-limiting conditions. AAPS J. 2016;18(2):321–32. https://doi.org/10.1208/s12248-015-9849-y.
WAGNER JG, NELSON E. Per cent absorbed time plots derived from blood level and/or urinary excretion data. J Pharm Sci. 1963;52:610–1. https://doi.org/10.1002/jps.2600520629.
Loo JC, Riegelman S. New method for calculating the intrinsic absorption rate of drugs. J Pharm Sci. 1968;57(6):918–28. https://doi.org/10.1002/jps.2600570602.
Gomeni R, Fang LL, Bressolle-Gomeni F, Spencer TJ, Faraone SV, Babiskin A. A general framework for assessing in vitro/in vivo correlation as a tool for maximizing the benefit-risk ratio of a treatment using a convolution-based modeling approach. CPT Pharmacometrics Syst Pharmacol. 2019;8(2):97–106. https://doi.org/10.1002/psp4.12378.
Hovorka R, Chappell MJ, Godfrey KR, Madden FN, Rouse MK, Soons PA. CODE: a deconvolution program implementing a regularization method of deconvolution constrained to non-negative values. Description and pilot evaluation. Biopharm Drug Dispos. 1998;19(1):39–53. https://doi.org/10.1002/(sici)1099-081x(199801)19:1<39::aid-bdd73>3.0.co;2-m.
Center for Drug Evaluation and Research. Guidance for industry: the use of physiologically based pharmacokinetic analyses — Biopharmaceutics Applications for Oral Drug Product Development, Manufacturing Changes, and Controls. US Food and Drug Administration; 2020.
Balan G, Timmins P, Greene DS, Marathe PH. In vitro-in vivo correlation (IVIVC) models for metformin after administration of modified-release (MR) oral dosage forms to healthy human volunteers. J Pharm Sci. 2001;90(8):1176–85. https://doi.org/10.1002/jps.1071.
Fuchs A, Leigh M, Kloefer B, Dressman JB. Advances in the design of fasted state simulating intestinal fluids: FaSSIF-V3. Eur J Pharm Biopharm. 2015;94:229–40. https://doi.org/10.1016/j.ejpb.2015.05.015.
Funding
This work is financially supported by the Innovation Technology and Commission of the Hong Kong Special Administrative Region, China (reference number: ITS/165/16FX).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
Guest Editors: Diane Burgess, Marilyn Morris and Meena Subramanyam
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
ESM 1
(DOCX 10933 kb)
Rights and permissions
About this article
Cite this article
Zhang, Y., Liu, H., Tang, M.J. et al. Screening of Bioequivalent Extended-Release Formulations for Metformin by Principal Component Analysis and Convolution-Based IVIVC Approach. AAPS J 23, 38 (2021). https://doi.org/10.1208/s12248-021-00559-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1208/s12248-021-00559-z