Abstract
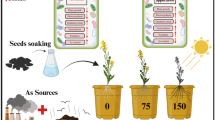
The present study was conducted to investigate the effect of titanium dioxide nanoparticles (TiO2 NPs) in the regulation of germination and seedling parameters of Bitter vetch (Vicia ervilia L.). Plants under salinity stress [sodium chloride (NaCl)] and drought stress [polyethylene glycol 6000, H(OCH2CH2)nOH synonyms: (PEG)]. Experimental salinity was controlled in terms of dS/m at levels of 0 (control), 4 and 12 dS/m and PEG induced drought stress (0, –0.4, and –0.8 MPa). TiO2 NPs (0, 20, and 40 ppm) was applied to Bitter vetch seed at germination and seedling growth at 21 days. The experiment was a three (TiO2 NPs) × three (salt) × three (drought) factorial combination (for 27 treatments) with three replicates. After 21 day cultivation, plants were harvested to determine the germination parameters, root length, shoot length, root and shoot fresh weight, root and shoot dry weight, and the proline content. Exposure of salinity stress and drought stress alone reduced the germination parameter in the plant. However, the application of TiO2 NPs protects Bitter vetch plants against salinity and drought stress as well as improves the germination parameters, root length, shoot length, root fresh weight, shoot dry weight, root dry weight and shoot fresh weight compared to control. The application of TiO2 NPs significantly enhanced germination percentage (GP), germination rate (GR), germination index (GI), seed vigor index (SVI), root length (RL), shoot length (SL), root and shoot fresh weight (RSFW), root and shoot dry weight (RSDW) up to 19, 23, 38, 16, 85, 45, 59, and 14%, respectively, under severe drought stress. And the application of TiO2 NPs significantly enhanced (GP), (GR), (GI), (SVI), (RL), (SL), (RSFW), and (RSDW) up to 5.4, 46.5, 61.6, 8.5, 3.9, 33, 44, and 11%, respectively, under severe salinity stress. Based on biomass assay, it was found that the seedlings displayed good growth over control, demonstrating a positive effect of the TiO2 NPs treatment. In high concentrations of TiO2 NPs, no positive effects were observed on the germination characteristics of seed Bitter vetch. To conclude the use of TiO2 NPs can be improved by improving the seed germination properties of the plant Bitter vetch that cause increases plant’s establishment in natural areas.





Similar content being viewed by others
REFERENCES
D. Bartels and R. Sunkar, Crit. Rev. Plant. Sci. 21, 1–36 (2005).
C. Wu, Q. Wang, B. Xie, et al., Afric. J. Biotechnol. 10, 17954–17961 (2011).
D. B. Egli and W. P. Bruening, Agronomy J. 92, 532–537 (2000).
T. J. Flowers, J. Exp. Bot. 55, 307–319 (2004).
H. C. J. Godfray, J. R. Beddington, I. R. Crute, et al., Am. Assoc. Advancem. Sci. 327, 812–818 (2010).
M. Tester and P. Langridge, Science (Washington, DC, U. S.) 327, 818–22 (2010).
W. Zhuang, J. Li, M. Cao, et al., J. Wuhan Bot. Res. 28, 730–736 (2010).
P. Ahmad, A. A. Abdel Latef, A. Hashem, et al., Front. Plant Sci. 7, 347 (2016).
R. Munns and M. Tester, Ann. Rev. Plant Biol. 59, 651–681 (2008).
FAO, FAO Land and Plant Nutrition Management Service (2010).
S. S. Pourdad and A. Beg, “Safflower: A suitable oilseed crop for dry-land areas of Iran,” in Proceedings of the 7th International Conference on Development of Drylands (2003), pp. 32, 33.
M. D. Kaya, G. Okçu, M. Atak, et al., Eur. J. Agron. 24, 291–295 (2006).
M. Ashraf and H. Rauf, Acta Physiol. Plantarum. 23, 407–414 (2001).
S. Ahmad, R. Ahmad, M. Y. Ashraf, et al., Pakist. J. Bot. 41, 647–654 (2009).
A. Maleki, A. Naderi, R. Naseri, et al., Bull. Env. Pharmacol. Life Sci. 2, 38–44 (2013).
M. D. F. Albuquerque and N. D. Carvalho, Seed Sci. Technol. (Switzerland) 31, 465–479 (2003).
A. Soleymani, M. Hesam, and H. Shahrajabian, Int. J. Biol. Macromol. 4, 2330 (2012).
H. Etesami, H. Fatemi, and M. Rizwan, “Interactions of nanoparticles and salinity stress at physiological, biochemical and molecular levels in plants: A review,” Ecotoxicol. Environ. Safety 225, 112769 (2021).
A. Mattiello, A. Filippi, F. Pošcic, et al., Front. Plant Sci. 6, 1043 (2015).
S. S. Hojjat and M. Kamyab, Russ. Agricult. Sci. 43, 61–65 (2017).
S. S. Hojjat, J. Nabati, S. M. Mirmiran, and H. Hojjat, Azar. J. Agricult. 7, 17–25 (2020).
S. S. Hojjat and H. Hojjat, Int. J. Food Eng. 1, 106–110 (2015).
S. S. Hojjat, Nanotechnol. Russ. 15, 204–211 (2020).
S. S. Hojjat, J. Environ. Soil Sci. 2, 186–191 (2019).
J. Moll, A. Gogos, T. D. Bucheli, et al., J. Nano Biotechnol. 14, 1–8 (2016).
H. Shi, R. Magaye, V. Castranova, and J. Zhao, Part. Fibre Toxicol. 10, 1–33 (2013).
E. Morteza, P. Moaveni, H. A. Farahani, and M. Kiyani, Springer Plus 2, 1–5 (2013).
H. Mahmoodzadeh, R. Aghili, and Nabavi, Tech. J. Eng. Appl. Sci. 3, 1365–1370 (2013).
M. Hrubý, P. Cígler, and S. Kuzel, J. Plant Nutrit. 25, 577–598 (2002).
L. Clément, C. Hurel, and N. Marmier, Chemosphere 90, 1083–1090 (2013).
J. Faraji and A. Sepehri, J. Seed Sci. 41, 309–317 (2019).
United States Environmental Protection Agency, Ecological Effects Test Guidelines Terrestrial Plant Toxicity, Vol. 1: Seedling Emergence (U.S. Environ. Protect. Agency, 1996).
Int. Seed Testing Assoc., “International rules for seed testing,” Seed Sci. Technol. 4, 51–177 (1976).
Int. Seed Testing Assoc., ISTA Rules (Int. Seed Test. Assoc., Zurich, Switzerland, 2009).
I. D. Maguire, Crop Sci. 22, 176–177 (1982).
L. S. Bates, R. P. Waldren, and I. D. Teare, Plant Soil 39, 205–207 (1973).
B. E. Michel and M. R. Kaufmann, Plant Physiol. 51, 914–916 (1973).
H. Feizi, P. R. Moghaddam, N. Shahtahmassebi, and A. Fotovat, Biol. Trace Element Res. 146, 101–106 (2012).
H. Feizi, M. Kamali, L. Jafari, and P. R. Moghaddam, Chemosphere 91, 506–511 (2013).
E. H. Dehkourdi and M. Mosavi, Biol. Trace Element Res. 155, 283–286 (2013).
A. Jaberzadeh, P. Moaveni, H. R. T. Moghadam, and H. Zahedi, Not. Botan. Horti Agrobot. Cluj-Napoca 41, 201–207 (2013).
J. Moll, F. Klingenfuss, Widmer, et al., Soil Biol. Biochem. 111, 85–93 (2017).
B. E. Michel and M. R. Kaufmann, Plant Physiol. 51, 914–916 (1973).
F. Kizilağaç and U. Tural, Klin. Psikofarmakol. Bul. 27, 172 (2017).
S. S. Hojjat, C. Mozumder, T. Bora, and G. L. Hornyak, Nanotechnol. Russ. 14, 582–587 (2019).
A. K. Parida and A. B. Das, Ecotoxicol. Environ. Safety 60, 324–349 (2005).
A. M. Abbasi, M. H. Shah, T. Li, et al., J. Ethno Pharmacol. 162, 333–345 (2015).
M. Farooq, M. Irfan, T. Aziz, et al., J. Agron. Crop Sci. 199, 12–22 (2013).
H. Mahmoodzadeh, M. Nabavi, and H. Kashefi, “Effect of nanoscale titanium dioxide particles on the germination and growth of canola (Brassica napus),” J. Ornament. Plants 3, 25–32 (2013).
A. Jaberzadeh, P. Moaveni, H. R. T. Moghadam, and H. Zahedi, Not. Botan. Horti Agrobot. Cluj-Napoca 41, 201–207 (2013).
F. L. Li, W. K. Bao, and N. Wu, Sci. Horticult. 127, 436–443 (2011).
H. Li, X. Li, D. Zhang, et al., Excli J. 12, 89 (2013).
C. Laura, H. Charlotte, and M. Nicolas, Chemosphere 1, 1083–1090 (2013).
H. Feizi, P. R. Moghaddam, N. Shahtahmassebi, and A. Fotovat, Biol. Trace Element Res. 146, 101–106 (2012).
ACKNOWLEDGMENTS
In addition, authors are thankful to all faculty members of the research center for plant sciences, Ferdowsi University of Mashhad, Iran, for their unconditional support to execute the present research work.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflicts of interest.
Rights and permissions
About this article
Cite this article
Seyed Saeid Hojjat, Louis Hornyak, G. TiO2 Nanoparticle Improve Germination and Seedling Parameters and Enhance Tolerance of Bitter Vetch (Vicia ervilia L.) Plants under Salinity and Drought Stress. Nanotechnol Russia 17, 411–419 (2022). https://doi.org/10.1134/S2635167622030168
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S2635167622030168