Abstract
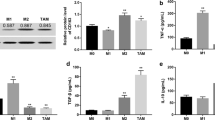
Hepatocellular carcinoma (HCC) is the most frequently diagnosed primary liver tumor worldwide. Tumor-associated macrophages (TAMs) usually have a similar phenotype to M2-like macrophages and can participate in tumor progression by secreting cytokines to suppress the immune response and activity of tumor-infiltrating lymphocytes. We investigated the role of M2 macrophages in HCC progression and explored the effects of vascular endothelial growth factor receptor 2 inhibitor—apatinib. As a cellular model of HCC, Hepb3 cell line was used. M2 macrophages were obtained by differentiation of THP-1 cells. The Transwell chamber was used to co-culture M2 macrophages and Hepb3 cells. CCK-8 and EdU assays were conducted to measure cell viability and proliferation capacity. Transwell migration assay was performed to estimate cellular metastatic potential. Cytokine expression levels were assessed by ELISA. Western blotting was used to characterize activation of the VEGFR2/STAT3/PD-L1 axis. It has been shown that co-culture with M2 macrophages increased viability, cytokine production, promoted proliferation, invasion, and migration of Hepb3 cells. The secretion of TGF-β1, IL-6, MMP-9, and VEGF was significantly increased after co-culture. In contrast apatinib suppressed M2 macrophage-induced proliferation, cell viability, invasion, and migration of Hepb3 cells. Moreover, apatinib markedly decreased expression levels of p-VEGFR2, p-STAT3, and PD-L1 in Hepb3 cells under the co-culture conditions. In conclusion, apatinib treatment can suppress TAMs-mediated malignant behavior of HCC cells via modulation of the VEGFR2/STAT3/PD-L1 signaling pathway.





Similar content being viewed by others
REFERENCES
Ferlay J., Colombet M., Soerjomataram I., Parkin D.M., Piñeros M., Znaor A., Bray F. 2021. Cancer statistics for the year 2020: an overview. Int. J. Cancer. 149, 778–789.
Caines A., Selim R., Salgia R. 2020. The Changing global epidemiology of hepatocellular carcinoma. Clin Liver Dis. 24, 535–547.
Pascual S., Herrera I., Irurzun J. 2016. New advances in hepatocellular carcinoma. World J. Hepatol. 8, 421–438.
Laplane L., Duluc D., Bikfalvi A., Larmonier N., Pradeu T. 2019. Beyond the tumour microenvironment. Int. J. Cancer. 145, 2611–2618.
Yin Y., Yao S., Hu Y., Feng Y., Li M., Bian Z., Zhang J., Qin Y., Qi X., Zhou L., Fei B., Zou J., Hua D., Huang Z. 2017. The immune-microenvironment confers chemoresistance of colorectal cancer through macrophage-derived IL6. Clin. Cancer Res. 23, 7375–7387.
Qian B.Z., Pollard J.W. 2010. Macrophage diversity enhances tumor progression and metastasis. Cell. 141, 39–51.
Pittet M.J., Michielin O., Migliorini D. 2022. Clinical relevance of tumour-associated macrophages. Nat. Rev. Clin. Oncol. 19, 402–421.
Martínez V.G., Rubio C., Martínez-Fernández M., Segovia C., López-Calderón F., Garín M.I., Teijeira A., Munera-Maravilla E., Varas A., Sacedón R., Guerrero F., Villacampa F., de la Rosa F., Castellano D., López-Collazo E., Paramio J.M., Vicente Á., Dueñas M. 2017. BMP4 induces M2 macrophage polarization and favors tumor progression in bladder cancer. Clin. Cancer Res. 23, 7388–7399.
Lu H., Clauser K.R., Tam W.L., Fröse J., Ye X., Eaton E.N., Reinhardt F., Donnenberg V.S., Bhargava R., Carr S.A., Weinberg R.A. 2014. A breast cancer stem cell niche supported by juxtacrine signalling from monocytes and macrophages. Nat. Cell Biol. 16, 1105–1117.
Daurkin I., Eruslanov E., Stoffs T., Perrin G.Q., Algood C., Gilbert S.M., Rosser C.J., Su L.M., Vieweg J., Kusmartsev S. 2011. Tumor-associated macrophages mediate immunosuppression in the renal cancer microenvironment by activating the 15-lipoxygenase-2 pathway. Cancer Res. 71, 6400–6409.
Xiao P., Long X., Zhang L., Ye Y., Guo J., Liu P., Zhang R., Ning J., Yu W., Wei F., Yu J. 2018. Neurotensin/IL-8 pathway orchestrates local inflammatory response and tumor invasion by inducing M2 polarization of tumor-associated macrophages and epithelial-mesenchymal transition of hepatocellular carcinoma cells. Oncoimmunology. 7, e1440166.
Bellmunt J., Powles T., Vogelzang N.J. 2017. A review on the evolution of PD-1/PD-L1 immunotherapy for bladder cancer: The future is now. Cancer Treat. Rev. 54, 58–67.
Llovet J.M., Castet F., Heikenwalder M., Maini M.K., Mazzaferro V., Pinato D.J., Pikarsky E., Zhu A.X., Finn R.S. 2022. Immunotherapies for hepatocellular carcinoma. Nat. Rev. Clin. Oncol. 19, 151–172.
Zhang H. 2015. Apatinib for molecular targeted therapy in tumor. Drug. Des. Dev. Ther. 9, 6075-6081.
Scott A.J., Messersmith W.A., Jimeno A. 2015. Apatinib: a promising oral antiangiogenic agent in the treatment of multiple solid tumors. Drugs Today (Barc.). 51, 223–229.
Chen X., Qiu T., Zhu Y., Sun J., Li P., Wang B., Lin P., Cai X., Han X., Zhao F., Shu Y., Chang L., Jiang H., Gu Y. 2019. A single-arm, phase II study of apatinib in refractory metastatic colorectal cancer. Oncologist. 24, 883–e407.
Zhao S., Ren S., Jiang T., Zhu B., Li X., Zhao C., Jia Y., Shi J., Zhang L., Liu X., Qiao M., Chen X., Su C., Yu H., Zhou C., Zhang J., Camidge D.R., Hirsch F.R. 2019. Low-dose apatinib optimizes tumor microenvironment and potentiates antitumor effect of PD-1/PD-L1 blockade in lung cancer. Cancer Immunol. Res. 7, 630–643.
Li L, Yu R, Cai T, Chen Z, Lan M, Zou T, Wang B, Wang Q, Zhao Y, Cai Y. 2020. Effects of immune cells and cytokines on inflammation and immunosuppression in the tumor microenvironment. Int. Immunopharmacol. 88, 106939.
Saraswati S., Alhaider A., Abdelgadir A.M., Tanwer P., Korashy H.M. 2019. Phloretin attenuates STAT-3 activity and overcomes sorafenib resistance targeting SHP-1-mediated inhibition of STAT3 and Akt/VEGFR2 pathway in hepatocellular carcinoma. Cell Commun. Signal. 17, 127.
Wen S., Shao G., Zheng J., Zeng H., Luo J., Gu D. 2019. Apatinib regulates the cell proliferation and apoptosis of liver cancer by regulation of VEGFR2/STAT3 signaling. Pathol. Res. Pract. 215, 816–821.
Anderson N.M., Simon M.C. 2020. The tumor microenvironment. Curr. Biol. 30, R921–r925.
Ngambenjawong C., Gustafson H.H., Pun S.H. 2017. Progress in tumor-associated macrophage (TAM)-targeted therapeutics. Adv. Drug Delivery Rev. 114, 206–221.
DeNardo D.G., Ruffell B. 2019. Macrophages as regulators of tumour immunity and immunotherapy. Nat. Rev. Immunol. 19, 369–382.
Mao X., Xu J., Wang W., Liang C., Hua J., Liu J., Zhang B., Meng Q., Yu X., Shi S. 2021. Crosstalk between cancer-associated fibroblasts and immune cells in the tumor microenvironment: new findings and future perspectives. Mol. Cancer. 20, 131.
Laoui D., Van Overmeire E., De Baetselier P., Van Ginderachter J.A., Raes G. 2014. Functional relationship between tumor-associated macrophages and macrophage colony-stimulating factor as contributors to cancer progression. Front Immunol. 5, 489.
Fantuzzi L., Tagliamonte M., Gauzzi M.C., Lopalco L. 2019. Dual CCR5/CCR2 targeting: opportunities for the cure of complex disorders. Cell. Mol. Life Sci. 76, 4869–4886.
Campagnolo L., Telesca C., Massimiani M., Stuhlmann H., Angelico M., Lenci I., Manzia T.M., Tariciotti L., Lehmann G., Baiocchi L. 2016. Different expression of VEGF and EGFL7 in human hepatocellular carcinoma. Dig. Liver Dis. 48, 76–80.
Bhoori S., Mazzaferro V. 2020. Combined immunotherapy and VEGF-antagonist in hepatocellular carcinoma: a step forward. Lancet Oncol. 21, 740–741.
Hu X., Zhang J., Xu B., Jiang Z., Ragaz J., Tong Z., Zhang Q., Wang X., Feng J., Pang D., Fan M., Li J., Wang B., Wang Z., Zhang Q., Sun S., Liao C. 2014. Multicenter phase II study of apatinib, a novel VEGFR inhibitor in heavily pretreated patients with metastatic triple-negative breast cancer. Int. J. Cancer. 135, 1961–1969.
Hu X., Cao J., Hu W., Wu C., Pan Y., Cai L., Tong Z., Wang S., Li J., Wang Z., Wang B., Chen X., Yu H. 2014. Multicenter phase II study of apatinib in non-triple-negative metastatic breast cancer. BMC Cancer. 14, 820.
Wang Q., Gao J., Di W., Wu X. 2020. Anti-angiogenesis therapy overcomes the innate resistance to PD-1/PD-L1 blockade in VEGFA-overexpressed mouse tumor models. Cancer Immunol. Immunother. 69, 1781–1799.
Zheng B., Ren T., Huang Y., Guo W. 2018. Apatinib inhibits migration and invasion as well as PD-L1 expression in osteosarcoma by targeting STAT3. Biochem. Biophys. Res. Commun. 495, 1695–1701.
Cai X., Wei B., Li L., Chen X., Liu W., Cui J., Lin Y., Sun Y., Xu Q., Guo W., Gu Y. 2020. Apatinib enhanced anti-PD-1 therapy for colon cancer in mice via promoting PD-L1 expression. Int. Immunopharmacol. 88, 106858.
Kambhampati S., Bauer K.E., Bracci P.M., Keenan B.P., Behr S.C., Gordan J.D., Kelley R.K. 2019. Nivolumab in patients with advanced hepatocellular carcinoma and Child-Pugh class B cirrhosis: Safety and clinical outcomes in a retrospective case series. Cancer. 125, 3234–3241.
El-Khoueiry A.B., Sangro B., Yau T., Crocenzi T.S., Kudo M., Hsu C., Kim T.Y., Choo S.P., Trojan J., Welling T.H.R., Meyer T., Kang Y.K., Yeo W., Chopra A., Anderson J., Dela Cruz C., Lang L., Neely J., Tang H., Dastani H.B., Melero I. 2017. Nivolumab in patients with advanced hepatocellular carcinoma (CheckMate 040): an open-label, non-comparative, phase 1/2 dose escalation and expansion trial. Lancet. 389, 2492–2502.
Pardoll D.M. 2012. The blockade of immune checkpoints in cancer immunotherapy. Nat. Rev. Cancer. 12, 252–264.
Bu L.L., Yu G.T., Wu L., Mao L., Deng W.W., Liu J.F., Kulkarni A.B., Zhang W.F., Zhang L., Sun Z.J. 2017. STAT3 induces immunosuppression by upregulating PD-1/PD-L1 in HNSCC. J. Dent. Res. 96, 1027–1034.
Xie C., Zhou X., Liang C., Li X., Ge M., Chen Y., Yin J., Zhu J., Zhong C. 2021. Apatinib triggers autophagic and apoptotic cell death via VEGFR2/STAT3/PD-L1 and ROS/Nrf2/p62 signaling in lung cancer. J. Exp. Clin. Cancer Res. 40, 266.
ADDITIONAL INFORMATION
The text was submitted by the author(s) in English.
Funding
This research was funded by Chen Xiao-Ping Foundation for the Development of Science and Technology of Hubei Province (no. CXPJJH12000001-2020206).
Author information
Authors and Affiliations
Contributions
Tao Yin and Changbo Fu are co-first authors. Tao Yin, Changbo Fu and Dongde Wu designed the research plan. Tao Yin, Changbo Fu, Dongde Wu, Lei Nie, Hu Chen and Yang Wang performed the experiments. Tao Yin and Changbo Fu wrote the manuscript.
Corresponding author
Ethics declarations
Conflict of interests. The authors declare that they have no conflict of interest.
Ethical approval. This article does not contain any studies with human participants or animals performed by any of the authors.
Informed consent. Not applicable.
Data availability statement. The data used to support the findings of this study are available from the corresponding author upon request.
Rights and permissions
About this article
Cite this article
Yin, T., Fu, C.B., Wu, D.D. et al. Apatinib Suppressed Macrophage-Mediated Malignant Behavior of Hepatocellular Carcinoma Cells via Modulation of VEGFR2/STAT3/PD-L1 Signaling. Mol Biol 57, 714–723 (2023). https://doi.org/10.1134/S0026893323040180
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0026893323040180