Abstract—
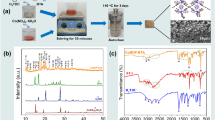
In this study, Ni–Co–Al2O3–SiC nanocomposite coatings with varying concentrations of Co were electrodeposited in a modified watts bath using conventional electrodeposition technique. The microhardness, surface morphology and phase structure of the coatings were characterized using Vickers microhardness tester, scanning electron microscopy and X-ray diffraction, respectively. The corrosion resistance of the composite coating was evaluated in 3.5% NaCl solution using an electrochemical work station. Increase in Co concentration in the electrolyte resulted in an increase in the microhardness of the nanocomposite coating. Corrosion resistance of coatings was also seen to increase considerably with increase in Co concentration in electrolyte. This was attributed mainly to the decrease in grain size, chemical composition, phase structure and preferred orientation of the coatings. The increase in Co in the electrolyte had a significant effect on the crystallite size of the Ni–Co–Al2O3–SiC coatings.







Similar content being viewed by others
REFERENCES
Srivastava, M., et al., Surf. Coat. Technol., 2006, vol. 201, pp. 3051–3060.
Ghaemi, M. and Binder, L.,J. Power Sources, 2002, vol. 111, pp. 248–254.
Chiu, S.-Y., et al., Surf. Coat. Technol., 2014, vol. 247, pp. 68–73.
Schlesinger, M. and M. Paunovic, Modern Electroplating, Hoboken, NJ: Wiley, 2011.
Fayomi, O.S.I. and Popoola, A.P.I., Egypt.J. Basic Appl. Sci., 2014, vol. 1, pp. 120–125.
Popov, K., Grgur, B., and Djokić, S.S., Fundamental Aspects of Electrometallurgy, New York: Kluwer Academic Publ., 2007.
Shi, L., et al., Appl. Surf. Sci., 2006, vol. 252, pp. 3591–3599.
Chang, L.M., Guo, H.F., and An, M.Z., Mater. Lett., 2008, vol. 62, pp. 3313–3315.
Ghazanlou, S.I., et al., Bull. Mater. Sci., 2016, vol. 39, pp. 1185–1195.
Srivastava, M., Srinivasan, A., and Grips, V.K.W., Am. J. Mater. Sci., 2011, vol. 1, pp. 113–122.
Srivastava, M., et al., Surf. Coat. Technol., 2010, vol. 205, pp. 66–75.
Shi, L., et al., Mater. Sci. Eng., A, 2005, vol. 397, pp. 190–194.
Ranjith, B. and Kalaignan, G.P., Appl. Surf. Sci., 2010, vol. 257, pp. 42–47.
Novitskaya, E., Khalifa, H.E., and Graeve, O.A., Mater. Lett., 2018, vol. 213, pp. 286–289.
Xu, S., et al., Ceram. Int., 2017, vol. 43, pp. 15060–15067.
Parchovianský, M., et al., J. Eur. Ceram. Soc., 2017, vol. 37, pp. 4297–4306.
Wu, Z., et al., Mater. Sci. Eng., A, 2012, vol. 556, pp. 767–774.
Bakhit, B. and Akbari, A., J. Coat. Technol. Res., 2013, vol. 10, pp. 285–295.
Wang, L., et al., Appl. Surf. Sci., 2005, vol. 242, pp. 326–332.
Altamirano-Garcia, L., et al., J. Solid State Electrochem., 2015, vol. 19, pp. 423–433.
Weil, R. and Cook, H.C., J. Electrochem. Soc., 1962, vol. 109, pp. 295–301.
Monshi, A., et al., World J. Nano Sci. Eng., 2012, vol. 2, pp. 154–160.
Zamani, M., Amadeh, A., and Baghal, S.M.L., Trans. Nonferrous Met. Soc. China, 2016, vol. 26, pp. 484–491.
Li, Y., et al., Surf. Coat. Technol., 2008, vol. 202, pp. 4952–4956.
Qiao, G., et al., Electrochim. Acta, 2005, vol. 51, pp. 85–92.
Gómez, E. and Valles, E., J. Appl. Electrochem., 1999, vol. 29, pp. 803–810.
Dehgahi, S., et al., J. Alloys Compd., 2017, vol. 692, pp. 622–628.
Hassani, S., Raeissi, K., and Golozar, M.A., J. Appl. Electrochem., 2008, vol. 38, pp. 689–694.
Shim, H.Y., et al., J. Agric. Chem. Environ., 2014, vol. 3, pp. 130–138.
Hanaor, D., et al., J. Eur. Ceram. Soc., 2012, vol. 32, pp. 235–244.
Cai, F., et al., Appl. Surf. Sci., 2015, vol. 324, pp. 482–489.
Bakhit, B., Surf. Coat. Technol., 2015, vol. 275, pp. 324–331.
Low, C., et al., Surf. Coat. Technol., 2006, vol. 201, pp. 371–383.
Van Tassel, J.J. and C.A. Randall, Key Eng. Mater., 2006, vol. 314, pp. 167–174.
Besra, L. and Liu, M., Prog. Mater. Sci., 2007, vol. 52, pp. 1–61.
Bakhit, B. and Akbari, A., Surf. Coat. Technol., 2012, vol. 206, pp. 4964–4975.
Prabu, S. and Wang, H.W., J. Chin. Chem. Soc., 2017, vol. 64, pp. 1467–1477.
Goto, Y., et al., Coatings, 2018, vol. 8, p. 5.
Atapattu, H., et al., J. Mater. Sci.: Mater. Electron., 2016, vol. 27, pp. 5415–5421.
Wang, P., et al., J. Coat. Technol. Res., 2011, vol. 8, pp. 409–417.
Schuh, C., Nieh, T., and Iwasaki, H.J.A.M., Acta Mater., 2003, vol. 51, pp. 431–443.
Pande, C. and Cooper, K., Prog. Mater. Sci., 2009, vol. 54, pp. 689–706.
Tury, B., et al., Surf. Coat. Technol., 2006, vol. 200, pp. 6713–6717.
Bakhit, B., et al., Appl. Surf. Sci., 2014, vol. 307, pp. 351–359.
Dheeraj, P., et al., J. Alloys Compd., 2017, vol. 729, pp. 1093–1107.
ACKNOWLEDGMENTS
EDS spectra of the Ni–Al2O3–SiC nanocomposite coatings with varying contents of Co was analysed by Research Institute of Nanjing Chemical Industry Group, Sinopec, thanks to senior engineer Jianguo Lu.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflicts of interest.
Rights and permissions
About this article
Cite this article
Nyambura Samuel Mbugua, Kang, M., Li, H. et al. The Influence of Co Concentration on the Properties of Conventionally Electrodeposited Ni–Co–Al2O3–SiC Nanocomposite Coatings. Prot Met Phys Chem Surf 56, 94–102 (2020). https://doi.org/10.1134/S2070205120010165
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S2070205120010165