Abstract
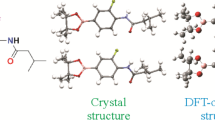
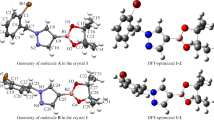
The target compound ethyl 3-(2,3-dihydrobenzofuran-5-yl)-2-propenoate was synthesized by a two-step reaction. A single crystal of this compound was grown in a suitable solvent system, the structure of this compound was confirmed by 1H and 13C NMR, MS, FT-IR, and X-ray single crystal diffraction methods. At the same time, conformational studies and crystallographic analysis of the structure were carried out. The Hirshfeld surface analysis method was used to analyze the intermolecular forces. The crystal structure of the target compound was optimized by density functional theory calculations. The crystal structure after X-ray crystal diffraction was compared with the crystal structure optimized by DFT calculation. The results showed that the single crystal structure determined by X-ray crystal diffraction was consistent with the molecular structure after DFT optimization. In addition, by further studying the molecular electrostatic potential and frontier molecular orbital of the target compound, the molecular physicochemical properties of the target compound are understood.







Similar content being viewed by others
REFERENCES
Xu, Z., Zhao, S.J., Lv, Z.S., Feng, L.S., Wang, Y.L., Zhang, F., Bai, L.Y, and Deng, J.L., Eur. J. Med. Chem., 2019, vol. 162, p. 266. https://doi.org/10.1016/j.ejmech.2018.11.025
Dong, M.Y., Zhang, Y., Jiang, H.Q., Ren, W.J., Xu, L.C., Zhang, Y.Q., and Liu, Y.H., Eur. J. Med. Chem., 2019, vol. 35, p. 5145. https://doi.org/10.1080/14786419.2020.1786827
Thanika, P., Pawinee, J., Wisanu, M., Roonglawan, R., Sumalee, K., Stephen, G.P., and Thunwadee, L., Nat. Prod. Res., 2020, vol. 34, p. 1394. https://doi.org/10.1080/14786419.2018.1512990
Kunwar, S., Lee, S.M., Kadayat, T.M., Shrestha, A., Park, P.H., and Lee, E.S., Bioorg. Med. Chem. Lett., 2022, vol. 73, p. 824. https://doi.org/10.1016/j.bmcl.2022.128921
López-Frías, G., Camacho-Dávila, A.A., ChávezFlores, D., Zaragoza-Galán, G., and RamosSánchez, V.H., Molecules, 2015, vol. 20, p. 8654. https://doi.org/10.3390/molecules20058654
Ha, H.J., Kang, D.W., Kim, H.M., Kang, J.M., Ann, J., Hyun, H.J., Lee, J.H., Kim, S.H., Kim, H., Choi, K., Hong, H.S., Kim, Y.H., Jone, D.G., and Lee, J., J. Med. Chem. 2018, vol. 61, p. 396. https://doi.org/10.1021/acs.jmedchem.7b00844
Liang, Z., Xu, Z., Tian, Y., Guo, M.B., Su, X., and Guo, C., Molecules, 2016, vol. 21, p. 732. https://doi.org/10.3390/molecules21060732
Gao, Y.T., Ma, C., Feng, X. Z., Liu, Y., and Hai, M.T.X., Chem. Biodivers., 2020, vol. 17, p. 346. https://doi.org/10.1002/cbdv.201900622
Abdelhafez, O.M., Ali, H.I., Amin, K.M., Abdalla, M.M., and Ahmed, E.Y., RSC Adv., 2015, vol. 5, p. 25312. https://doi.org/10.1039/c4ra16228e
Gao, S.Y., Qian, G.Y., Tang, H., Yang, Z., and Zhou, Q.H., ChemCatChem, 2019, vol. 11, p. 5762. https://doi.org/10.1002/cctc.201901355
Ye, W.J., Chen, D.M., Wu, Q.M., Chen, Y.M., Yang, D.Z., Liao, T.H., and Zhou, Z.X., J. Struct. Chem., 2021, vol. 62, p. 1357. https://doi.org/10.1134/s0022476621090043
Zhou, L.P., Qiu, F., Zhang, Z.F., Wang, C.M., Dong, Y.L., and Liu, J.P., J. Struct. Chem., 2020, vol. 61, p. 1111. https://doi.org/10.1134/s0022476620070148
Chamberland, S., Ziller, J.W., and Woerpel, K.A., J. Am. Chem. Soc, 2005, vol. 127, p. 755. https://doi.org/10.1107/s2053229614024218
Dai, H.Y., Yang, D.Z., Liao, W.P., Wu, F., Zhou, Z.X., and Huang, Z.Y., Russ. J. Gen. Chem., 2022, vol. 92, p. 438. https://doi.org/10.1134/s1070363222030112
Wu, Q.M., Zheng, Z.P., Ye, W.J., Guo, Q., Liao, T.H., Yang, D.Z., and Zhou, Z.X., J. Struct. Chem., 2022, vol. 52, p. 1247. https://doi.org/10.1016/j.molstruc.2021.131367
Im, G.Y.J., Bronner, S.M., Goetz, A.E., Paton, R.S., Cheong, P.H Y, Houk, K.N., and Garg, N.K., J. Am. Chem. Soc., 2010, vol. 132, p. 17933. https://doi.org/10.1021/ja1086485
Deng, L.Y., Liao, T.H., Sun, H., Hu, W.Y., Zhou, Z.X., Zhao, C.S., and Pan, H.Y., Mol. Cryst. Liq. Cryst., 2022, vol. 737, p. 30. https://doi.org/10.1080/15421406.2021.2003952
Wu, Q., Li, J.D., Liu, F.X., Xiao, J.C., Tang, Y.F., and Zi, Q.L., Russ. J. Coord. Chem., 2020, vol. 46, p. 37. https://doi.org/10.1134/s1070328420020086
Liu, Y., Zheng, Z., Deng, H., Ren, Q., Zhou, Z., Zhao, C., and Chai, H., J. Struct. Chem., 2021, vol. 62, p. 1285. https://doi.org/10.1134/s0022476621080163
Liang, S.Z., Hammond, G.B., and Bo, X., Eur. J. Med. Chem., 2017, vol. 23, p. 17850. https://doi.org/10.1080/15421406.2021.2003952
Daoui, S., Çınar, E.B., El Kalai, F., Saddik, R., Dege, N., Karrouchi, K., and Benchat, N., Acta Cryst., 2019, vol. 75, p. 1880. https://doi.org/10.1107/s2056989019015147
Fukui, K., Yonezawa, T., and Shingu, H.J., Polym. Sci., 1952, vol. 20, p. 722. https://doi.org/10.1021/ja1086485
Dyachenko, I.V., Dyachenko, V.D., Yakushev, I.A., Khrustalev, V.N., and Nenaidenko, V.G., Russ. J. Org. Chem., 2020, vol. 56, p. 1669. https://doi.org/10.1134/s1070428020090262
Pearson, R.G., Proc. Natl. Acad. Sci. U.S.A., 1986, vol. 83, p. 8440. https://doi.org/10.1073/pnas.83.22.8440
Mckinnon, J.J., Fabbiani, F., and Spackman, M.A., Cryst. Growth Des, 2007, vol. 7, p. 755. https://doi.org/10.1021/cg060773k
Roberto, G.S.B., J. Nat. Prod. 2005, vol. 66, p. 1577. https://doi.org/10.1021/np058254r
Lin, W.Y., Yang, F., Duan, A.N.W., You, W., and Zhao, P.L., J. Struct. Chem, 2018, vol. 37, p. 1557. https://doi.org/10.14102/j.cnki.0254-5861
Sheldrick, G.M., Acta Cryst. (C), 2015, vol. 71, p. 467. https://doi.org/10.1107/s2053229614024218
Qin, Y., Long, D., Zhu, X., Zhou, Z., Chai, H., and Zhao, J. Struct. Chem., 2019, vol. 11, p. 1917. https://doi.org/10.1134/s0022476619120072
Vieweg, N., Celik, M.A., Zakel, S., Gupta, V., Frenking, G., and Koch, M., J. Infrared Millim. Terahertz Waves, 2014, vol. 35, p. 478. https://doi.org/10.1007/s10762-014-0062-3
Wandas, M., Lorenc, J., Kucharska, E., Ma̧czka, M., and Hanuza, J., J. Raman Spectrosc., 2008, vol. 39, p. 832. https://doi.org/10.1002/jrs.1922
ACKNOWLEDGMENTS
This work has been awarded the Guizhou Provincial Natural Science Foundation ([2020]1Y393).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
No conflict of interest was declared by the authors.
Supplementary information
Rights and permissions
About this article
Cite this article
Ma, LX., Shi, Y., Li, JL. et al. Synthesis, Crystal Structure, and DFT Study of Ethyl 3-(2,3-Dihydrobenzofuran-5-yl)-2-propenoate. Russ J Gen Chem 92, 2880–2887 (2022). https://doi.org/10.1134/S1070363222120416
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070363222120416