Abstract
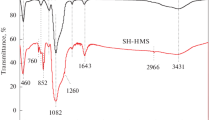
Thiol-functionalized organosilica (OS-SH) was synthesized via an ultra-fast sol–gel procedure without using any templates. The OS-SH was then employed as an adsorbent to remove Hg(II) ions from aqueous solution. Pore structure and material morphology were characterized by FTIR, TGA, SEM, TEM, and N2 sorption measurements. The OS-SH adsorbents demonstrated an outstanding adsorption performance (72.48 mg g–1 at pH 3). The adsorption isotherm can be best described by the Langmuir model, suggesting a monolayer adsorption behavior by chemical mechanism. The adsorption kinetics of Hg(II) on OS‑SH was well fitted by the pseudo-second-order model. The synthesis approach proposed in this study provides a much simple, tender as well as highly efficient way to design functional porous adsorbents for the removal of heavy metal ions.








Similar content being viewed by others
REFERENCES
J. Wang, X. Feng, C. W. N. Anderson, et al., J. Hazard. Mater. 221, 1 (2012).
P. Kampalanonwat and P. Supaphol, ACS Appl. Mater. Interfaces 2, 3619 (2010).
C. Sun, C. Li, C. Wang, et al., Chem. End. J. 200, 291 (2012).
Y. Niu, H. Liu, R. Qu, et al., Ind. End. Chem. Res. 54, 1656 (2015).
J. L. Gong, B. Wang, G. M. Zeng, et al., J. Hazard. Mater. 164, 1517 (2009).
Y. Li, M. Xia, F. An, et al., J. Hazard. Mater. 271, 33 (2019).
H. Takahashi, B. Li, T. Sasaki, et al., Chem. Mater. 12, 3301 (2000).
E. de Canck, L. Lapeire, J. de Clercq, et al., Langmuir 26, 10076 (2010).
T. J. Pinnavaia, Chem. Commun. 1, 69 (1999).
Y. Niu, R. Qu, H. Chen, et al., J. Hazard. Mater. 278, 267 (2014).
B. Samiey, C. H. Cheng, and J. Wu, Materials 7, 673 (2014).
T. Velempini and K. Pillay, J. Environ. Chem. End. 7, 103350 (2019).
L. Mercier and T. J. Pinnavaia, Environ. Sci. Technol. 32, 2749 (1998).
J. Ryu, M. Y. Lee, M. G. Song, et al., Sep. Purif. Technol. 250, 117120 (2020).
R. Xu, P.Lin, Q. Zhang, et al., Ind. End. Chem. Res. 55, 2183 (2016).
P. H. T. Ngamou, J. P. Overbeek, R. Kreiter, et al., J. Mater. Chem. A 1, 5567 (2013).
X. Yan, J. Meng, X. Hu, et al., J. Sol–Gel Sci. Technol. 89, 617 (2019).
J. Mosa, A. Duran, and M. Aparicio, J. Power Sources 297, 208 (2015).
X. Wang, Q. Kang, D. Shen, et al., Talanta 124, 7 (2014).
M. Puanngam and F. Unob, J. Hazard. Mater. 154, 578 (2008).
J. Li, X. Li, A. Alsaedi, et al., J. Colloid Interface Sci. 517, 61 (2018).
L. Huang, M. He, B. Chen, et al., J. Mater. Chem. A 4, 5159 (2016).
L. Mercier and T. J. Pinnavaia, Environ. Sci. Technol. 32, 2749 (1998).
J. Chen, R. Qu, Y. Zhang, et al., Chem. End. J. 209, 235 (2012).
T. A. Saleh, Environ. Sci. Pollut. R 22, 16721 (2015).
A. K. Meena, G. K. Mishra, P. K. Rai, et al., J. Hazard. Mater. 122, 161 (2005).
M. Naushad, T. Ahamad, G. Sharma, et al., Chem. End. J. 300, 306 (2016).
ACKNOWLEDGMENTS
This work was supported by Jiangsu Key Laboratory of Advanced Catalytic Materials and Technology (BM2012110) and Advanced Catalysis and Green Manufacturing Collaborative Innovation Center, Changzhou University.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
The author declares that he (she) has no conflicts of interest.
Rights and permissions
About this article
Cite this article
Xu, R., Tan, H., Guo, M. et al. Ultra-Fast Synthesis of Thiol-Functionalized Organosilica (OS-SH) for Adsorption of Hg(II) from Aqueous Solution. Russ. J. Phys. Chem. 96, 1561–1566 (2022). https://doi.org/10.1134/S0036024422070263
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0036024422070263