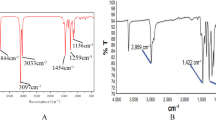
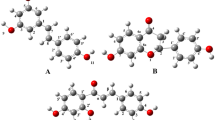
Abstract
Epigallocatechin gallate (EGCG or (2S,3R)-5,7-dihydroxy-2-(3,4,5-trihydroxyphenyl)-3,4-dihydro-2H-chromen-3-yl 3,4,5-trihydroxybenzoate) and its isomers gallocatechin gallate (GCG or (2R,3R)-2-(3,4,5-trihydroxyphenyl)-3,4-dihydro-1(2H)-benzopyran-3,5,7-triol 3-(3,4,5-trihydroxybenzoate) are the main antioxidant components in tea polyphenols and tea. In this paper, the B3LYP density functional theory (DFT) was used to optimize the EGCG and GCG molecular configuration at the 6‑311G(d,p) level of theory implemented in Gaussian software. The result is convergent and has no imaginary frequency, indicating a stable structure that reaches minimum energy value. Multiwfn wave function analysis software was used to study the frontier orbitals of EGCG and GCG. The electrophilic and nucleophilic sites of the two molecules were predicted by the contribution of each atom to the highest occupied molecular orbital (HOMO) and the lowest unoccupied molecular orbital (LUMO). The results show that the electrophilic reaction is mainly on No.1 benzene ring, and the main contribution is the C atom, especially the para‑C atom of the C atom connected by the two phenolic hydroxyl groups on the benzene ring. At the same time, the activation of the phenolic hydroxyl group on the para- and ortho-C atoms on the benzene ring was verified. When there is a phenolic hydroxyl group on the benzene ring, its para- and ortho-C atoms become more active, and they are easier to lose electrons. So substitution or other reactions occurs. This site is also the main site against oxygen free radicals which is an accelerated aging substance. The nucleophilic sites are mainly around C=O and no. 3 benzene rings, so it has certain ability to obtain electrons. Finally, the active sites of EGCG were analyzed by using the two methods of Fukui function and mean partial ionization energy, and the results were basically consistent with the above.






Similar content being viewed by others
REFERENCES
H. Mukhtar and N. Ahmad, Am. J. Clin. Nutr. 71, 1698 (2000).
S. Katiyar and H. Mukhtar, Int. J. Oncol. 8, 221 (1996).
L. Trovoa, C. Fuchsb, R. de Rosaa, I. Barbieroa, M. Tramarina, E. Cianib, L. Rusconia, and C. Kilstrup-Nielsena, Neurobiol. Disease 138, 104791 (2020)
Y. Li, S. U. Rahman, Y. Huang, Y. Zhang, P. Ming, L. Zhu, X. Chu, J. Li, S. Feng, X. Wang, and J. Wu, J. Nutr. Biochem. 78, 108324 (2020)
Q. Ru, Q. Xiong, X. Tian, L. Chen, M. Zhou, Y. Li, and C. Li, Front. Physiol. 10, 1450 (2019).
Y. Baba, J. I. Sonoda, S. Hayashi, N. Tosuji, S. Sonoda, K. Makisumi, and M. Nakajo, Exp. Ther. Med. 4, 452 (2012).
B. Pu, Y. Xu, C. Du, B. Qin, H. Cao, and Y. You, Food Ind. 38, 301 (2017).
D. Cho, H. W. Jeong, J. K. Kim, A. Y. Kim, Y. D. Hong, J. H. Lee, J. K. Choi, and D. B. Seo, J. Med. Food 22, 779 (2019).
T. Yu, B. Hu, R. Sun, Z. Jin, Y. Wang, L. Zhang, W. Xu, and G. Liu, Remote Sens. Tech. Appl. 31, 872 (2016).
G. Feng, H. Zhang, Y. Ma, Y. Zhang, K. Zhao, and Y. Shang, Nat. Prod. Res. Develop. 29, 1882 (2017).
J. Luo, J. Song, L. Liu, B. Xue, G. Tian, and Y. Yang, Poultry Sci. 97, 599 (2018).
A. Takagaki, S. Otani, and F. Nanjo, Biosci. Biotechnol. Biochem. 75, 582 (2011).
J. D. Lambert, S. Sang, J. Hong, and C. S. Yang, J. Agric. Food Chem. 58, 10016 (2010).
T. H. Peeters, L. Krissie, B. Vincent, A. M. L. Sanne, N. A. M. H. Corina, M. Remco, V. R. Arno, W. Ron, N. S. Paul, H. Arend, and P. J. L. William, Cancer Metab. 20 (7), 4 (2019).
F. de Amicis, A. Perri, D. Vizza, A. Russo, M. L. Panno, D. Bonofiglio, C. Giordano, L. Mauro, S. Aquila, D. Tramontano, and S. Andò, J. Cell Physiol. 228, 2054 (2013).
H. Jin, W. Gong, C. Zhang, and S. Wang, Onco Targets Ther. 6, 145 (2013).
G. Castellano-González, P. Nicolas, O. B. J. William, B. Alban, M. Helder, and J. G. Gilles, Oncotarget 7, 7426 (2016).
T. Li, N. Zhao, J. Lu, Q. Zhu, X. Liu, F. Hao, and X. Jiao, Bioengineered 10, 282 (2019).
Q. Q. Wu, Y. F. Liang, S. B. Ma, H. Li, and W. Y. Gao, J. Sci. Food Agric. 99, 5984 (2019).
C. S. Yang, J. D. Lambert, J. Ju, G. Lu, and S. Sang, Toxicol. Appl. Pharm. 224, 265 (2007).
X. A. Hui, S. H. Hua, Q. Q. Wu, H. Li, and W. Y. Gao, Arch. Biochem. Biophys. 622, 1 (2017).
J. Chen, Z. Luo, and J. Yao, Phys. Chem. Chem. Phys. 19, 21777 (2017).
X. Li, X. Cao, N. Heinz, and M. Dolg, Mol. Phys. 2020, e1736676 (2020)
Y. Su, L. Zhou, Z. Zuo, and C. Zhao, Nat. Prod. Res. Develop. 13 (6), 19 (2001).
S. Anitha, S. Krishnan, K. Senthilkumar, and V. Sasirekha, Mol. Phys. 2020, e1745917 (2020)
T. Lu and F. W. Chen, Acta Phys.-Chim. Sin. 28, 1 (2012).
K. Fukui. T, Yonezawa, C. Nagata, and H. Shingu, J. Chem. Phys. 22, 1433 (1954).
T. Li, Y. L.Tang, Z. G. Ling, and Z. W. Long, Spectrosc. Spec. Anal. 34, 2122 (2014).
H. J. Forman, K. J. A. Davies, and F. Ursini, Free Radic. Bio. Med. 66, 24 (2014).
F. Halgand, Ch. Houée-Lévin, M. Weik, and D. Madern, J. Struct. Biol. 210, 107478 (2020).
A. Warnholtz and T. Munzel, Curr. Control. Trials. Cardiovasc. Med. 1, 38 (2000).
W. Liu, Z. Xu, T. Yang, Y. Deng, B. Xu, S. Feng, and Y. Li, Free Rad. Res. 48, 849 (2014).
D. Cho, H. W. Jeong, J. K. Kim, A. Y. Kim, Y. D. Hong, J. H. Lee, J. K. Choi, and D. B. Seo, J. Med. Food 22, 779 (2019).
P. Monira, U. Keiko, T. Akiko, I. Mamoru, and N. Yoriyuki, Int. J. Mol. Sci. 20, 3630 (2019).
T. Kohri, N. Matsumoto, M. Yamakawa, M. Suzuki, F. Nanjo, Y. Hara, and N. Oku, J. Agric. Food Chem. 49, 4102 (2001).
K. Unno, M. Pervin, A. Nakagawa, K. Iguchi, A. Hara, A. Takagaki, F. Nanjo, A. Minami, and Y. Nakamura, Mol. Nutr. Food Res. 61, 1700294 (2017).
J. D. Lambert, S. Sang, J. Hong, and C. S. Yang, J. Agric. Food Chem. 58, 10016 (2010).
ACKNOWLEDGMENTS
This work is supported by the National Natural Science Foundation of China (grant no. 11164004), Guizhou industrial breakthrough project (grant no. GY[2012]3060), and Guizhou Provincial Photonic Science and Technology Innovation team (Qianke Joint talents team [2015]4017).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Tang, T., Yu, J., Yuan, L. et al. Frontier Orbitals and Аctive Site of Тea Рolyphenol Мolecules Epigallocatechin Gallate and Gallocatechin Gallate. Russ. J. Phys. Chem. 95, 1857–1863 (2021). https://doi.org/10.1134/S0036024421090284
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0036024421090284