Abstract
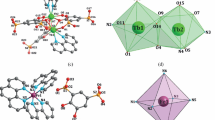
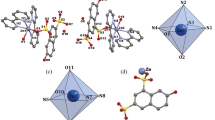
Two coordination compounds, Dy2(H2L)3(phen)2·2(phen) (I) and [Dy(L)(H2O)2·Hbipy]n (II) (Na2H2L is 4,5-dihydroxy-1,3-benzenedisulfonic acid disodium; phen is 1,10-phenanthroline; bipy is 2,2'-bipyridine), were hydrothermally synthesized. They have been characterized by single crystal X-ray diffraction and elemental analyses. In compound I, introduction of the auxiliary chelating ligand phen led to a new Dy phenol-sulfonate with dinuclear structure bridging by the three H2L2– ligands. The interconnection of the dinuclear compounds via the π–π stacking interactions between phen molecules resulted in a one dimensional chain structure. Compound II features a novel one-dimensional chain in which Dy atoms are bridged by the phenol-sulfonate ligands in pentadentate fashion. Dy atom is eight-coordinated by one sulfonate group bidentately, one phenol group unidentately and two phenol groups from one L4– ligand. Compounds I and II exhibit photoluminescence with emissions maximum at 408 and 438 nm, which can be attributed to the intraligand emission state, furthermore, they also display the characteristic emissions of Dy ions, correspond to 4F5/2–6H15/2, 4F5/2–6H13/2 and 4F5/2–6H11/2 transitions, respectively.





Similar content being viewed by others
REFERENCES
M. Y. Masoomi and A. Morsali, Coord. Chem. Rev. 256, 2921(2012). https://doi.org/10.1016/j.ccr.2012.05.032
C. Janiak, Cheminform 35, 2781(2003). https://doi.org/10.1002/chin.200406292
V. S. Gusarov, A. M. Cheplakova, D. G. Samsonenko, et al., Russ. J. Inorg. Chem. 66, 1374 (2021). https://doi.org/10.1134/S0036023621090035
Y. Liu, W. Li, Y. Q. Yang, et al., J. Struct. Chem. 62, 740(2021). https://doi.org/10.1134/S0022476621050103
D. Ureche, A. Rija, I. Bulhac, et al., Russ. J. Inorg. Chem. 65, 1838(2021). https://doi.org/10.1134/S0036023620120189
J. A. Hua, Z. P. Duan, C. K. Gao, et al., Russ. J. Coord. Chem. 47, 646(2021). https://doi.org/10.1134/S1070328421090049
R. S. Chemistry, Dalton Trans. 9, 1447(2000). https://doi.org/10.1039/b000871k
B. Chand, U. Ray, G. Mostafa, et al., Polyhedron 23, 1669 (2004). https://doi.org/10.1016/j.poly.2004.02.029
R. P. Ye, X. Zhang, Y. Y. Qin, et al., CrystEngComm 19, 1658 (2017). https://doi.org/10.1039/C7CE00004A
P. Thuéry, Inorg. Chem. 52, 435 (2013). https://doi.org/10.1021/ic3022744
H. Huang, M. Payehghadr, J. Wang, et al., Helv. Chim. Acta 97, 345 (2014). https://doi.org/10.1002/hlca.201300156
B. Yhxa, A. Zpd, A. Zbz, et al., J. Solid State Chem. 271, 273 (2019). https://doi.org/10.1016/j.jssc.2019.01.007
W. Min and Z. Lian, J. Cluster Sci. 24, 989 (2013). https://doi.org/10.1007/s10876-013-0591-2
F. Y. Yi, Q. P. Lin, T. H. Zhou, et al., Crystal Growth Des. 10, 1788 (2010). https://doi.org/10.1021/cg901463f
L. L. Wei and J. J. Vittal, J. Inclusion Phenom. Macrocyclic Chem. 71, 557(2011). https://doi.org/10.1007/s10847-011-0015-6
P. K. Baker, A. E. Jenkins, and M. M. Meehan, Polyhedron 19, 2171(2000). https://doi.org/10.1016/S0277-5387(00)00518-0
Z. Y. Du, H. B. Xu, and J. G. Mao, Inorg. Chem. 45, 9780 (2006). https://doi.org/10.1021/ic0613255
Z. Y. Du, H. B. Xu, X. L. Li, et al., Eur. J. Inorg. Chem. 4520, 4520 (2007). https://doi.org/10.1002/ejic.200700463
H. Y. Cao, Q. Y. Liu, L. Q. Li, et al., Z. Anorg. Allg. Chem. 640, 1420 (2014). https://doi.org/10.1002/zaac.201300681
G. M. Sheldrick, SHELXT-2018, Program for X-ray Crystal Structure Solution (University of Göttingen, Göttingen, 2018).
G. M. Sheldrick, SHELXL-2018, Program for X-ray Crystal Structure Refinement (University of Göttingen, Göttingen, 2018).
Y. G. Huang, X. T. Wang, F. L. Jiang, et al., Chemistry 14, 10340 (2008). https://doi.org/10.1002/chem.200800344
Y. X. Chi, S. Y. Niu, and J. Jin, Inorg. Chim. Acta 362, 3821 (2009). https://doi.org/10.1016/j.ica.2009.05.001
Y. Chen, Y. Cao, W. Chen, et al., Dalton Trans. 42, 10011 (2013). https://doi.org/10.1039/c3dt50453k
N. Phukan, S. Goswami, S. Lipstman, et al., Cryst. Growth Des. 20, 2973 (2020). https://doi.org/10.1021/acs.cgd.9b01599
X. Li, R. Cao, W. H. Bi, et al., Eur. J. Inorg. Chem. 3156 (2005). https://doi.org/10.1002/ejic.200400983
G. Wu, F. J. Yin, H. Wei, et al., Z. Anorg. Allg. Chem. 637, 602 (2011). https://doi.org/10.1002/zaac.201000372
Funding
This work was supported by the Scientific Research Fund Project of Liaoning Provincial Education Department no. LJKZ0371.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflicts of interest.
Rights and permissions
About this article
Cite this article
Yin, P., Xue, C., Yan, Y. et al. Syntheses, Crystal Structures, and Properties of Two Dysprosium Coordination Compounds Based on Na2H2L Ligands. Russ. J. Inorg. Chem. 67, 456–463 (2022). https://doi.org/10.1134/S0036023622040222
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0036023622040222