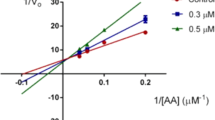
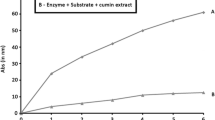
Abstract
New potential inhibitors of 5-lipoxygenase (5-LOX) based on the structure of 2-(3-benzoylphenyl)propanoic acid (an active component of the nonsteroidal antiinflammatory drug Ketoprofen) were designed using the SARD-21 program. The docking of these compounds in the active site of 5-LOX suggested that seven compounds can interact with this enzyme. Two of them can also be dual inhibitors of 5-LOX and two isoforms of cyclooxygenase.
Similar content being viewed by others
References
Tsuji, F., Aono H., Tsuboi, T., Murakami, T., Enomoto, H., Mizutani, K., and Inagaki, N. (2010) Biol. Pharm. Bull., 33, 1050–1053.
Poeckel, D., and Funk, C. D. (2010) Cardiovasc. Res., 86, 243–253.
Szczeklik, A. (1997) Allergy, 52, 613–619.
Barranco, P., Bobolea, I., Larco, J. I., Prior, N., Lopez-Serrano, M. C., and Quirce, S. (2009) J. Invest. Allergol. Clin. Immunol., 19, 446–452.
Barnes, N. C., and Smith, L. J. (1998) in Five Lipoxygenase Products in Asthma. Lung Biology in Health Disease (Drazen, J. M., Dahlen, S. E., and Lee, T. H., eds.) N. Y., pp. 175–188.
Carraway, R. E., Cochrane, D. E., Salmonsen, R., Muraki, K., and Boucher, W. (1991) Peptides, 12, 1105–1111.
Rajic, Z., Hadjipavlou-Litina, D., Pontiki, E., Kralj, M., Suman, L., and Zorc, B. (2010) Chem. Biol. Drug Des., 75, 641–652.
Miyazawa, K., Iimori, Y., Makino, M., Mikami, T., and Miyasaka, K. (1985) Jpn. J. Pharmacol., 38, 199–205.
Flynn, D. L., Capiris, T., Cetenko, W. J., Connor, D. T., Dyer, R. D., Kostlan, C. R., Nies, D. E., Schrier, D. J., and Sircar, J. C. (1990) J. Med. Chem., 33, 2070–2072.
Muri, E. M. F., Nieto, M. J., Sindelar, R. D., and Williamson, J. S. (2002) Curr. Med. Chem., 9, 1631–1653.
Tyurina, L. A., Tyurina, O. V., and Kolbin, A. M. (2007) Methods and Results of Design and Prediction of Biologically Active Compounds [in Russian], Gilem, Ufa.
Khayrullina, V. R., Taipov, I. A., Gerchikov, A. Ya., and Zarudij, F. S. (2012) Khim.-Farm. Zh., 46, 33–43.
Khayrullina, V. R., Muhametov, A. D., Tyurina, L. A., Garifullina, G. G., Gerchikov, A. Ya., and Zarudij, F. S. (2007) Biomed. Khim., 53, 266–275.
Gerchikov, A. Ya., Monakov, Yu. B., Khayrullina, V. R., Muhametov, A. D., Garifullina, G. G., Tarasov, G. P., and Tyurina, L. A. (2008) Izv. Vysshikh Uchebn. Zaved., Ser. Khim. Khim. Tekhnol., 51, 87–92.
Pettersen, E. F., Goddard, T. D., Huang, C. C., Couch, G. S., Greenblatt, D. M., Meng, E. C., and Ferrin, T. E. (2004) J. Comput. Chem., 25, 1605–1612.
Gasteiger, J., and Marsili, M. (1980) Tetrahedron, 36, 3219–3228.
Gilbert, N. C., Bartlett, S. G., Waight, M. T., Neau, D. B., Boeglin, W. E., Brash, A. R., and Newcomer, M. E. (2011) Science, 331, 217–219.
Werz, O. (2007) Planta Med., 73, 1331–1357.
Butovich, I., and Reddy C. C. (2002) Biochem. J., 365, 865–871.
Karelina, T. A., Zhudenkov, K. V., Demin, O. O., Svetlichny, D. V., Agoram, B., Fairman D., and Demin, O. V. (2012) Systems Biol., 6, 141–154.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Original Russian Text © V. R. Khayrullina, I. A. Taipov, A. V. Veselovsky, D. S. Shcherbinin, A. Ya. Gerchikov, 2014, published in Biokhimiya, 2014, Vol. 79, No. 4, pp. 476–484.
Rights and permissions
About this article
Cite this article
Khayrullina, V.R., Taipov, I.A., Veselovsky, A.V. et al. New inhibitors of 5-lipoxygenase catalytic activity based on 2-(3-methylphenyl)propanoic acid and 4-substituted morpholine derivatives. Biochemistry Moscow 79, 376–384 (2014). https://doi.org/10.1134/S0006297914040063
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0006297914040063