Abstract
It has great significance in profiling specific proteins throughout for better understanding of complex pathological processes and in-depth pharmacological studies. In this work, an efficient protein profiling strategy was developed based on the specific protein–drug interaction. Sofosbuvir (SOF), as a first-line drug for the treatment of hepatitis C, was modified onto the surface of nanoparticles through stable chemical bonds to fabricate a novel magnetic separation material denoted as Fe3O4@SiO2@PAA@SOF. With sequence coverage as the screening parameter, nine proteins were profiled from fetal bovine serum (FBS) of which eight were liver related. Similarly, the strategy was applied to hepatocellular carcinoma (HCC) patient serum. Eight proteins were profiled and all of them were liver related, demonstrating the superb specificity and selectivity of this strategy for profiling liver-related proteins by virtue of protein–SOF interaction. When serum proteins from HCC patients were compared to those from healthy people, one unique differential protein (D3DQX7) was profiled, which was liver related and was a potential target for ameliorating liver diseases. For further research, this material design concept and protein profiling strategy can be extended to employ other drugs for corresponding studies.
Graphical abstract
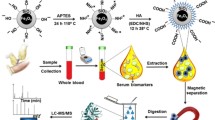
Sofosbuvir, as a therapeutic drug for liver diseases, was modified onto the surface of magnetic nanoparticles to fabricate the specific selective separation material (Fe3O4@SiO2@PAA@SOF). Based on protein–SOF interaction, the material was applied to adsorb specific proteins from different serum samples. After MS analysis, specific proteins, most of which were liver related, were successfully profiled from FBS and HCC patient serum, fully demonstrating the superb specificity and selectivity of this protein profiling strategy.








Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article and its supplementary information files.
References
N.L. Anderson, N.G. Anderson, The human plasma proteome: history, character, and diagnostic prospects. Mol. Cell. Proteom. 1, 845–867 (2002)
N. Rifai, M.A. Gillette, S.A. Carr, Protein biomarker discovery and validation: the long and uncertain path to clinical utility. Nat. Biotechnol. 24, 971–983 (2006)
M.M. Akhilesh Pandey, Proteomics to study genes and genomes. Nature 405, 837–846 (2000)
S. Mahrus, J.C. Trinidad, D.T. Barkan, A. Sali, A.L. Burlingame, J.A. Wells, Global sequencing of proteolytic cleavage sites in apoptosis by specific labeling of protein N termini. Cell 134, 866–876 (2008)
S. Gallien, E. Perrodou, C. Carapito, C. Deshayes, J.-M. Reyrat, A. Van Dorsselaer, O. Poch, C. Schaeffer, Lecompte, Ortho-proteogenomics: multiple proteomes investigation through orthology and a new MS-based protocol. Genome Res. 19, 128–135 (2009)
J.-S. Kim, Z. Dai, U.K. Aryal, R.J. Moore, D.G. Camp, S.E. Baker, R.D. Smith, W.-J. Qian, Resin-assisted enrichment of N-terminal peptides for characterizing proteolytic processing. Anal. Chem. 85, 7 (2013)
W. Dormeyer, S. Mohammed, B.V. Breukelen, J. Krijgsveld, A.J.R. Heck, Targeted analysis of protein termini. J. Proteome Res. 6, 4634–4645 (2007)
G. Xu, S.B.Y. Shin, S.R. Jaffrey, Chemoenzymatic labeling of protein C-termini for positive selection of C-terminal peptides. ACS Chem. Biol. 6, 1015–1020 (2011)
H.B.R. Eivazzadeh-Keihan, Z. Amiri et al., Functionalized magnetic nanoparticles for the separation and purification of proteins and peptides. Trends Anal. Chem. 141, 20 (2021)
G.K. Sofer, L. Hagel, G.K. Sofer, et al. Handbook of process chromatography: a guide to optimization, scale up, and validation[J] (1997)
R.H. Davis, Theory for crossflow microfiltration (1992)
S.L.M.L. Mir, V. Goel, R. Kaiser, Crossflow microfiltration: applications, design, and cost (1992)
Z.A.L. Zeman, Microfiltration and ultrafiltration: principles and applications (1996)
M.B.P.F. García, M. Wolf, S. Reinlein, M.F. von Roman, S. Berensmeier, High-gradient magnetic separation for technical scale protein recovery using low cost magnetic nanoparticles. Sep. Purif. 150, 8 (2015)
F.G.F. Jiao, H. Wang, Y. Deng, Y. Zhang, X. Qian, Y. Zhang, Polymeric hydrophilic ionic liquids used to modify magnetic nanoparticles for the highly selective enrichment of N-linked glycopeptides. Sci. Rep. 7, 11 (2017)
T. Wang, D. Babusis, Y. Park et al., Species differences in liver accumulation and metabolism of nucleotide prodrug sofosbuvir. Drug Metab. Pharmacokinet. 35, 7 (2020)
K. Lipa, M.J. Jabłońska, P. Kaczor et al., Sofosbuvir as a drug in recurrent HCV therapy occurring after liver transplantation. J. Educ. Health Sport 8, 15 (2018)
N. Gupta, R. Bodin et al., Autoimmune hepatitis in association with sofosbuvir. Am. J. Ther. 10, 1–3 (2015)
W.S. Qin E, Cui Y, et al., A case of liver injury caused by DAA based therapy, Hepatol. Int. 11, S1043–S1044 (2017)
D. Bojkova, S. Westhaus, R. Costa et al., Sofosbuvir activates EGFR-dependent pathways in hepatoma cells with implications for liver-related pathological processes. Cells 9, 18 (2020)
F.M. Varshosaz, Nanoparticles for targeted delivery of therapeutics and small interfering RNAs in hepatocellular carcinoma. World J. Gastroenterol. 21, 12022–12041 (2015)
Y.A. Li, X.D. Zhao, H.P. Yin, G.J. Chen, S. Yang, Y.B. Dong, A drug-loaded nanoscale metal-organic framework with a tumor targeting agent for highly effective hepatoma therapy. Chem. Commun. (Camb) 52, 14113–14116 (2016)
Z. Li, W. Tao, D. Zhang, C. Wu, B. Song, S. Wang, T. Wang, M. Hu, X. Liu, Y. Wang, Y. Sun, J. Sun, The studies of PLGA nanoparticles loading atorvastatin calcium for oral administration in vitro and in vivo, Asian. J. Pharm. Sci. 12, 285–291 (2017)
I.U.K. Yasir Mehmooda, Y. Shahzad et al., Amino-decorated mesoporous silica nanoparticles for controlled sofosbuvir delivery. Eur. J. Pharmaceut. Sci. 143, 10 (2020)
S.A. Loutfy, H.G. Abdelhady, M.H. Elberry, et al., In vitro evaluation of cytotoxic and anti-HCV-4 properties of sofosbuvir encapsulated chitosan nanoparticles (2020). https://doi.org/10.48550/arXiv.2009.06041[P]
M.J.B. Sofia, D., Chang, W., Du, J., Nagarathnam, D. et al., Discovery of a β-d-2′-deoxy-2′-α-fluoro-2′-β-C-methyluridine nucleotide prodrug (PSI-7977) for the treatment of hepatitis C virus, J. Med. Chem. 53, 7202–7218 (2010)
R. Ovadia, A. Khalil, Hea Li, Synthesis and anti-HCV activity of β-D -20-deoxy-20-α-chloro-20-β-fluoro and β-d-20-deoxy-20-α-bromo-20-β-fluoro nucleosides and their phosphoramidate prodrugs. Bioorg. Med. Chem. 27, 13 (2019)
B.Y. Yanping Li, Y. Quan, Z. Li, Advancement of prodrug approaches for nucleotide antiviral agents. Curr. Topics Med. Chem. 21, 19 (2021)
Y. Pan, C. Zhang, R. Xiao, L. Zhang, W. Zhang, Dual-functionalized magnetic bimetallic metal-organic framework composite for highly specific enrichments of phosphopeptides and glycopeptides. Anal. Chim. Acta 1158, 338412 (2021)
C. Szabó, G. Veres, T. Radovits, et al., Infusion of sodium sulfide improves myocardial and endothelial function in a canine model of cardiopulmonary bypass. Crit. Care 11 (Suppl 2), P1 (2007)
O.A. Gressner, C. Gao, M. Siluschek, P. Kim, A.M. Gressner, Inverse association between serum concentrations of actin-free vitamin D-binding protein and the histopathological extent of fibrogenic liver disease or hepatocellular carcinoma. Eur. J. Gastroenterol. Hepatol. 21, 990–995 (2009)
Z.H. Jiang, Z.Y. Zhang, M. He et al., The screening and identification of Apolipoprotein A-II from serum differential proteins in hepatocellular carcinoma patients. Chin. J. Hepatol. 18(6), 445–449 (2010)
Z.T. Hui Jiang, Pujun Gao, Xu. Yonghong, Xinjuan Kong, Han Zhang et al., Interaction of Beta2-glycoproteinI with lipopolysaccharide leads to NF-kappa B activation in hepatocellular carcinoma. Chin. J. Clin (Electronic Edition) 7, 5 (2013)
V.G. Jonnalagadda, K. Choudhary, V. Kranti Matety, Afamin, a novel biomarker: one for all concerns? J. Clin. Mol. Med. 1(2), 1–2 (2018)
J.-S. Kim, H.-J. Lee, Y.-J. Kim, Y.-B. Kim, The mesh density effect on stress intensity factor calculation using ABAQUS XFEM. J. Mech. Sci. Technol. 33, 4909–4916 (2019)
T.T. Chang, C.H. Ho, Plasma proteome atlas for differentiating tumor stage and post-surgical prognosis of hepatocellular carcinoma and cholangiocarcinoma. PLoS ONE 15, e0238251 (2020)
T. Chen, X. Dai, J. Dai, C. Ding, Z. Zhang, Z. Lin, J. Hu, M. Lu, Z. Wang, Y. Qi, L. Zhang, R. Pan, Z. Zhao, L. Lu, W. Liao, X. Lu, AFP promotes HCC progression by suppressing the HuR-mediated Fas/FADD apoptotic pathway. Cell Death Dis. 11, 822 (2020)
N. Fu, H. Du, D. Li, Y. Lu, W. Li, Y. Wang, L. Kong, J. Du, S. Zhao, W. Ren, F. Han, R. Wang, Y. Zhang, Y. Nan, Clusterin contributes to hepatitis C virus-related hepatocellular carcinoma by regulating autophagy. Life Sci. 256, 117911 (2020)
W.L. Jianwen Zhang, B. Fu, K. Hu, S. Liu, H. Li, Q. Zhang, M. Li, G. Chen, Proteomic screening of serum hepatocellular carcinoma-associated proteins by an acetonitrile-pretreatment method. J. Sun Yan-Sen Univ. (Med. Sci.) 31, 5 (2020)
F. Ozdemir, A. Baskiran, The importance of AFP in liver transplantation for HCC. J. Gastrointest. Cancer 51, 1127–1132 (2020)
F. Xue, L. Yang, B. Dai, H. Xue, L. Zhang, R. Ge, Y. Sun, Bioinformatics profiling identifies seven immune-related risk signatures for hepatocellular carcinoma. PeerJ 8, e8301 (2020)
T.M. Qing-hua Chang, Y. Tao, T. Dong, Xx. Tang, G.-h Ge, Zj. Xu, Pan-cancer analysis identifies ITIH1 as a novel prognostic indicator for hepatocellular carcinoma. Aging 13, 24 (2021)
L.E. Santiesteban-Lores, M.C. Carneiro, L. Isaac, L. Bavia, Complement system in alcohol-associated liver disease. Immunol. Lett. 236, 37–50 (2021)
Y.L. Liya Mu, C. Wang, X. Zhang, Diagnostic value of combined α1-antitrypsin, α1-acid glycoprotein and prealbumin assay for hepatocellular carcinoma. Shanxi Med. J. 17, 3 (1988)
A.T.-C.C. Terence Chuen-Wai Poon, B. Zee, S. King-Wah Ho, T. Shu-Kam Mok, T. Wai-Tong Leung, P. James Johnson, Application of classification tree and neural network algorithms to the identification of serological liver marker profiles for the diagnosis of hepatocellular carcinoma. Oncology 61, 9 (2001)
T. Utsunomiya, K. Ogawa, K. Yoshinaga, M. Ohta, K. Yamashita, K. Mimori, H. Inoue, T. Ezaki, Y. Yoshikawa, M. Mori, Clinicopathologic and prognostic values of apolipoprotein D alterations in hepatocellular carcinoma. Int. J. Cancer 116, 105–109 (2005)
E.Y. Lee, J.H. Kang, K.A. Kim, T.W. Chung, H.J. Kim, D.Y. Yoon, H.G. Lee, D.H. Kwon, J.W. Kim, C.H. Kim, E.Y. Song, Development of a rapid, immunochromatographic strip test for serum asialo alpha1-acid glycoprotein in patients with hepatic disease. J. Immunol. Methods 308, 116–123 (2006)
X.X. Hongguang Han, J. Zhang, Expression and significance of angiotensin II and angiotensin II type I receptor in human hepatocellular carcinoma. Chin. J. General Surg. 18, 4 (2009)
P. Darebna, P. Novak, R. Kucera, O. Topolcan, M. Sanda, R. Goldman, P. Pompach, Changes in the expression of N- and O-glycopeptides in patients with colorectal cancer and hepatocellular carcinoma quantified by full-MS scan FT-ICR and multiple reaction monitoring. J. Proteom. 153, 44–52 (2017)
W.Y. Xianlin Zhan, C. Gao, Research progress on alpha-2-macroglobulin and its glycosylation in tumor. Lab. Med. 32, 6 (2017)
G. Aran, L. Sanjurjo, C. Barcena, M. Simon-Coma, E. Tellez, M. Vazquez-Vitali, M. Garrido, L. Guerra, E. Diaz, I. Ojanguren, F. Elortza, R. Planas, M. Sala, C. Armengol, M.R. Sarrias, CD5L is upregulated in hepatocellular carcinoma and promotes liver cancer cell proliferation and antiapoptotic responses by binding to HSPA5 (GRP78). FASEB J. 32, 3878–3891 (2018)
C. Barcena, G. Aran, L. Perea, L. Sanjurjo, E. Tellez, A. Oncins, H. Masnou, I. Serra, M. Garcia-Gallo, L. Kremer, M. Sala, C. Armengol, P. Sancho-Bru, M.R. Sarrias, CD5L is a pleiotropic player in liver fibrosis controlling damage, fibrosis and immune cell content. EBioMedicine 43, 513–524 (2019)
V. Peta, J. Zhu, D.M. Lubman, S. Huguet, F. Imbert-Bismutd, G. Bolbach, G. Clodic, L. Matheron, Y. Ngo, P. Raluca, C. Housset, K. Rezai, T. Poynard, G. FibroFrance-Groupe Hospitalier Pitie Salpetriere, Input of serum haptoglobin fucosylation profile in the diagnosis of hepatocellular carcinoma in patients with non-cirrhotic liver disease. Clin. Res. Hepatol. Gastroenterol. 44, 681–691 (2020)
Y. Shang, M. Luo, F. Yao, S. Wang, Z. Yuan, Y. Yang, Ceruloplasmin suppresses ferroptosis by regulating iron homeostasis in hepatocellular carcinoma cells. Cell Signal 72, 109633 (2020)
H.F. Zhang, X. Gao, X. Wang, X. Chen, Y. Huang, L. Wang, Z.W. Xu, The mechanisms of renin-angiotensin system in hepatocellular carcinoma: from the perspective of liver fibrosis HCC cell proliferation, metastasis and angiogenesis, and corresponding protection measures. Biomed. Pharmacother. 141, 111868 (2021)
D. Li, P. Xie, S. Zhao et al., Hepatocytes derived increased SAA1 promotes intrahepatic platelet aggregation and aggravates liver inflammation in NAFLD. Biochem. Biophys. Res. Commun. 555, 7 (2021)
Funding
Financial support was from National Natural Science Foundation of China (21974045, 82102503), the National Key R&D Program of China (2021YFF0701900), the Science and Technology Commission of Shanghai Municipality (No. 19142201100, 6142200500), the Fundamental Research Funds for the Central Universities (No. JKJ01221718), and Zhongshan Hospital Fudan University Youth Foundation (2020ZSQN30).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
There is no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. The study was approved by the Ethics Committee of Zhongshan Hospital Fudan University.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Pan, Y., Wang, Z., Xu, S. et al. Selective profiling of liver-related specific proteins based on sofosbuvir-modified magnetic separation material. ANAL. SCI. 39, 313–323 (2023). https://doi.org/10.1007/s44211-022-00238-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s44211-022-00238-9