Abstract
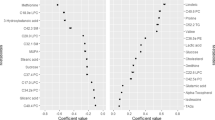
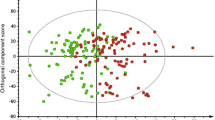
A metabolically healthy status, whether obese or not, is a transient stage with the potential to develop into metabolic disorders during the course of life. We investigated the incidence of metabolic disorders in 1078 metabolically healthy Chinese adults from the Shanghai Changfeng Study and looked for metabolites that discriminated the participants who would develop metabolic disorders in the future. Participants were divided into metabolically healthy overweight/obesity (MHO) and metabolically healthy normal weight (MHNW) groups according to their body mass index (BMI) and metabolic status. Their serum metabolomic profile was measured using a 1H nuclear magnetic resonance spectrometer (1H-NMR). The prevalence of diabetes, hypertriglyceridemia, hypercholesterolemia and metabolic syndrome was similar between the MHNW and MHO participants at baseline. After a median of 4.2 years of follow-up, more MHO participants became metabolically unhealthy than MHNW participants. However, a subgroup of MHO participants who remained metabolically healthy (MHO → MHO) had a similar prevalence of metabolic disorders as the MHNW participants at the follow-up examination, despite a significant reduction in their serum concentrations of high-density lipoprotein (HDL) and an elevation in valine, leucine, alanine and tyrosine. Further correlation analysis indicated that serum intermediate-density lipoprotein (IDL) and very low-density lipoprotein cholesterol (VLDL-CH) might be involved in the transition from metabolically healthy to unhealthy status and could be valuable to identify the MHNW and MHO with increased metabolic risks.



Similar content being viewed by others
Availability of data and materials
The data generated and analyzed during the current study are not publicly available due to the relevant policy of data management from the sponsors in the Chinese national and local government, but the data are available from the corresponding authors upon reasonable request with the permission of the Chinese national and local government.
Code Availability
Not applicable.
Abbreviations
- MHO:
-
Metabolically healthy overweight/obesity
- MUO:
-
Metabolically unhealthy overweight/obesity
- MHNW:
-
Metabolically healthy normal weight
- MUNW:
-
Metabolically unhealthy normal weight
- MHNW → MHNW:
-
Remaining in MHNW
- MHNW → MHO:
-
MHNW transition to MUO
- MHNW → MUNW:
-
MHNW transition to MUNW
- MHNW → MUO:
-
MHNW transition to MUO
- MHO → MHNW:
-
MHO transition to MHNW
- MHO → MHO:
-
Remaining in MHO
- MHO → MUNW:
-
MHO transition to MUNW
- MHO → MUO:
-
MHO transition to MUO
- BMI:
-
Body mass index
- BP:
-
Blood pressure
- SBP:
-
Systolic blood pressure
- DBP:
-
Diastolic blood pressure
- FBG:
-
Fasting blood glucose
- TC:
-
Total cholesterol
- TG:
-
Triglycerides
- HDL-C:
-
High-density lipoprotein cholesterol
- LDL-C:
-
Low-density lipoprotein cholesterol
- 2hPG:
-
2-H Post-challenge plasma glucose
- OGTT:
-
Oral glucose tolerance test
- VLDL-PN:
-
Very low-density lipoprotein particle numbers
- VLDL-CH:
-
Very low-density lipoprotein cholesterol
- VLDL-CE:
-
Very low-density lipoprotein cholesteryl ester
- VLDL-FC:
-
Very low-density lipoprotein free cholesterol
- VLDL-AB:
-
Very low-density lipoprotein apolipoprotein B-100
- VLDL-PL:
-
Very low-density lipoprotein phospholipid
- VLDL-TG:
-
Very low-density lipoprotein triglycerides
- IDL-PN:
-
Intermediate-density lipoprotein particle numbers
- IDL-CH:
-
Intermediate-density lipoprotein cholesterol
- IDL-CE:
-
Intermediate-density lipoprotein cholesteryl ester
- IDL-FC:
-
Intermediate-density lipoprotein free cholesterol
- IDL-AB:
-
Intermediate-density lipoprotein apolipoprotein B-100
- IDL-PL:
-
Intermediate-density lipoprotein phospholipid
- IDL-TG:
-
Intermediate-density lipoprotein triglyceride
- HDL-CH:
-
High-density lipoprotein cholesterol
- HDL-CE:
-
High-density lipoprotein cholesteryl ester
- HDL-FC:
-
High-density lipoprotein free cholesterol
- HDL-PL:
-
High-density lipoprotein phospholipid
- HDL-TG:
-
High-density lipoprotein triglycerides
- AA:
-
Amino acid
- VLDL1-CH:
-
Very low-density lipoprotein-1 cholesterol
- VLDL1-CE:
-
Very low-density lipoprotein-1 cholesteryl ester
- VLDL1-FC:
-
Very low-density lipoprotein-1 free cholesterol
- VLDL1-PL:
-
Very low-density lipoprotein-1 phospholipid
- VLDL1-TG:
-
Very low-density lipoprotein-1 triglycerides
- VLDL2-CH:
-
Very low-density lipoprotein-2 cholesterol
- VLDL2-CE:
-
Very low-density lipoprotein-2 cholesteryl ester
- VLDL2-FC:
-
Very low-density lipoprotein-2 free cholesterol
- VLDL2-PL:
-
Very low-density lipoprotein-2 phospholipid
- VLDL2-TG:
-
Very low-density lipoprotein-2 triglycerides
- VLDL3-CH:
-
Very low-density lipoprotein-3 cholesterol
- VLDL3-CE:
-
Very low-density lipoprotein-3 cholesteryl ester
- VLDL3-FC:
-
Very low-density lipoprotein-3 free cholesterol
- VLDL3-PL:
-
Very low-density lipoprotein-3 phospholipid
- VLDL3-TG:
-
Very low-density lipoprotein-3 triglycerides
- VLDL4-CH:
-
Very low-density lipoprotein-4 cholesterol
- VLDL4-CE:
-
Very low-density lipoprotein-4 cholesteryl ester
- VLDL4-FC:
-
Very low-density lipoprotein-4 free cholesterol
- VLDL4-PL:
-
Very low-density lipoprotein-4 phospholipid
- VLDL4-TG:
-
Very low-density lipoprotein-4 triglycerides
- VLDL5-CH:
-
Very low-density lipoprotein-5 cholesterol
- VLDL5-CE:
-
Very low-density lipoprotein-5 cholesteryl ester
- VLDL5-FC:
-
Very low-density lipoprotein-5 free cholesterol
- VLDL5-PL:
-
Very low-density lipoprotein-5 phospholipid
- VLDL5-TG:
-
Very low-density lipoprotein-5 triglycerides
References
Alberti KG, Zimmet PZ (1998) Definition, diagnosis and classification of diabetes mellitus and its complications part 1—diagnosis and classification of diabetes mellitus provisional report of a WHO consultation. Diabet Med 15:539–553. https://doi.org/10.1002/(SICI)1096-9136(199807)15:7%3c539::AID-DIA668%3e3.0.CO;2-S
Alberti KGMM, Zimmet P, Shaw J (2006) Metabolic syndrome—a new world-wide definition. A consensus statement from the international diabetes federation. Diabet Med 23:469–480. https://doi.org/10.1111/j.1464-5491.2006.01858.x
Al-Mrabeh A, Zhyzhneuskaya SV, Peters C, Barnes AC, Melhem S, Jesuthasan A, Aribisala B, Hollingsworth KG, Lietz G, Mathers JC et al (2020) Hepatic lipoprotein export and remission of human type 2 diabetes after weight loss. Cell Metab 31:233–249. https://doi.org/10.1016/j.cmet.2019.11.018 (e234)
Badoud F, Perreault M, Zulyniak MA, Mutch DM (2015) Molecular insights into the role of white adipose tissue in metabolically unhealthy normal weight and metabolically healthy obese individuals. FASEB J 29:748–758. https://doi.org/10.1096/fj.14-263913
Bell JA, Hamer M, Sabia S, Singh-Manoux A, Batty GD, Kivimaki M (2015) The natural course of healthy obesity over 20 years. J Am Coll Cardiol 65:101–102. https://doi.org/10.1016/j.jacc.2014.09.077
Cadenas-Sanchez C, Ruiz JR, Labayen I, Huybrechts I, Manios Y, Gonzalez-Gross M, Breidenassel C, Kafatos A, De Henauw S, Vanhelst J et al (2017) Prevalence of metabolically healthy but overweight/obese phenotype and its association with sedentary time, physical activity, and fitness. J Adolesc Health 61:107–114. https://doi.org/10.1016/j.jadohealth.2017.01.018
Chang Y, Jung HS, Yun KE, Cho J, Ahn J, Chung EC, Shin H, Ryu S (2016) Metabolically healthy obesity is associated with an increased risk of diabetes independently of nonalcoholic fatty liver disease. Obesity (silver Spring) 24:1996–2003. https://doi.org/10.1002/oby.21580
Charles BA, Doumatey A, Huang H, Zhou J, Chen G, Shriner D, Adeyemo A, Rotimi CN (2011) The roles of IL-6, IL-10, and IL-1RA in obesity and insulin resistance in African-Americans. J Clin Endocrinol Metab 96:E2018-2022. https://doi.org/10.1210/jc.2011-1497
Ctoi AF, Parvu AE, Andreicut AD, Mironiuc A, Crciun A, Ctoi C, Pop ID (2018) Metabolically healthy versus unhealthy morbidly obese: chronic inflammation, nitro-oxidative stress, and insulin resistance. Nutrients. https://doi.org/10.3390/nu10091199
Doumatey AP, Zhou J, Zhou M, Prieto D, Rotimi CN, Adeyemo A (2016) Proinflammatory and lipid biomarkers mediate metabolically healthy obesity: a proteomics study. Obesity (silver Spring) 24:1257–1265. https://doi.org/10.1002/oby.21482
Eckel N, Li Y, Kuxhaus O, Stefan N, Hu FB, Schulze MB (2018) Transition from metabolic healthy to unhealthy phenotypes and association with cardiovascular disease risk across BMI categories in 90,257 women (the Nurses’ Health Study): 30 year follow-up from a prospective cohort study. Lancet Diabetes Endocrinol 6:714–724. https://doi.org/10.1016/s2213-8587(18)30137-2
Eslam M, Newsome PN, Sarin SK, Anstee QM, Targher G, Romero-Gomez M, Zelber-Sagi S, Wai-Sun Wong V, Dufour JF, Schattenberg JM et al (2020) A new definition for metabolic dysfunction-associated fatty liver disease: an international expert consensus statement. J Hepatol 73:202–209. https://doi.org/10.1016/j.jhep.2020.03.039
Espinosa De Ycaza AE, Donegan D, Jensen MD (2018) Long-term metabolic risk for the metabolically healthy overweight/obese phenotype. Int J Obes (lond) 42:302–309. https://doi.org/10.1038/ijo.2017.233
Feng S, Gong X, Liu H, Lu R, Duan T, Wang M, Wang J, Wang H, Chen J, Liu Y et al (2020) The diabetes risk and determinants of transition from metabolically healthy to unhealthy phenotypes in 49,702 older adults: 4-year cohort study. Obesity (silver Spring) 28:1141–1148. https://doi.org/10.1002/oby.22800
Gao X, Hofman A, Hu Y, Lin H, Zhu C, Jeekel J, Jin X, Wang J, Gao J, Yin Y et al (2010) The Shanghai Changfeng Study: a community-based prospective cohort study of chronic diseases among middle-aged and elderly: objectives and design. Eur J Epidemiol 25:885–893. https://doi.org/10.1007/s10654-010-9525-6
Gilardini L, Zambon A, Soranna D, Croci M, Invitti C (2018) Predictors of the transition from metabolically healthy obesity to unhealthy obesity. Eat Weight Disord 23:739–744. https://doi.org/10.1007/s40519-018-0600-4
Gomez-Huelgas R, Ruiz-Nava J, Santamaria-Fernandez S, Vargas-Candela A, Alarcon-Martin AV, Tinahones FJ, Bernal-Lopez MR (2019) Impact of intensive lifestyle modification on levels of adipokines and inflammatory biomarkers in metabolically healthy obese women. Mediators Inflamm 2019:4165260. https://doi.org/10.1155/2019/4165260
Hashimoto Y, Hamaguchi M, Fukuda T, Ohbora A, Kojima T, Fukui M (2017) Fatty liver as a risk factor for progression from metabolically healthy to metabolically abnormal in non-overweight individuals. Endocrine 57:89–97. https://doi.org/10.1007/s12020-017-1313-6
Heianza Y, Kato K, Kodama S, Suzuki A, Tanaka S, Hanyu O, Sato K, Sone H (2014) Stability and changes in metabolically healthy overweight or obesity and risk of future diabetes: Niigata wellness study. Obesity (silver Spring) 22:2420–2425. https://doi.org/10.1002/oby.20855
Hirano T (2018) Pathophysiology of diabetic dyslipidemia. J Atheroscler Thromb 25:771–782. https://doi.org/10.5551/jat.RV17023
Hwang YC, Hayashi T, Fujimoto WY, Kahn SE, Leonetti DL, McNeely MJ, Boyko EJ (2015) Visceral abdominal fat accumulation predicts the conversion of metabolically healthy obese subjects to an unhealthy phenotype. Int J Obes (lond) 39:1365–1370. https://doi.org/10.1038/ijo.2015.75
Hwang YC, Ahn HY, Park CY (2019) Association between nonalcoholic fatty liver disease and future deterioration of metabolic health: a cohort study. Obesity (silver Spring) 27:1360–1366. https://doi.org/10.1002/oby.22536
Jiang L, Huang J, Wang Y, Tang H (2012) Eliminating the dication-induced intersample chemical-shift variations for NMR-based biofluid metabonomic analysis. Analyst 137:4209–4219. https://doi.org/10.1039/c2an35392j
Jimenez B, Holmes E, Heude C, Tolson RF, Harvey N, Lodge SL, Chetwynd AJ, Cannet C, Fang F, Pearce JTM et al (2018) Quantitative lipoprotein subclass and low molecular weight metabolite analysis in human serum and plasma by (1)H NMR spectroscopy in a multilaboratory trial. Anal Chem 90:11962–11971. https://doi.org/10.1021/acs.analchem.8b02412
Jung CH, Kang YM, Jang JE, Hwang JY, Kim EH, Park JY, Kim HK, Lee WJ (2016) Fatty liver index is a risk determinant of incident type 2 diabetes in a metabolically healthy population with obesity. Obesity (silver Spring) 24:1373–1379. https://doi.org/10.1002/oby.21483
Kanagasabai T, Thakkar NA, Kuk JL, Churilla JR, Ardern CI (2015) Differences in physical activity domains, guideline adherence, and weight history between metabolically healthy and metabolically abnormal obese adults: a cross-sectional study. Int J Behav Nutr Phys Act 12:64. https://doi.org/10.1186/s12966-015-0227-z
Kang EY, Yim JE (2019) Differences in dietary intakes, body compositions, and biochemical indices between metabolically healthy and metabolically abnormal obese Korean women. Nutr Res Pract 13:488–497. https://doi.org/10.4162/nrp.2019.13.6.488
Karpe F, Pinnick KE (2015) Biology of upper-body and lower-body adipose tissue–link to whole-body phenotypes. Nat Rev Endocrinol 11:90–100. https://doi.org/10.1038/nrendo.2014.185
Kramer CK, Zinman B, Retnakaran R (2013) Review 21.317 Ann Intern Med. Are metabolically healthy overweight and obesity benign conditions?—A systematic review and meta-analysis. Ann Intern Med 159:758–769. https://doi.org/10.7326/0003-4819-159-11-201312030-00008
Mongraw-Chaffin M, Foster MC, Kalyani RR, Vaidya D, Burke GL, Woodward M, Anderson CA (2016) Obesity severity and duration are associated with incident metabolic syndrome: evidence against metabolically healthy obesity from the multi-ethnic study of atherosclerosis. J Clin Endocrinol Metab 101:4117–4124. https://doi.org/10.1210/jc.2016-2460
Mongraw-Chaffin M, Foster MC, Anderson CAM, Burke GL, Haq N, Kalyani RR, Ouyang P, Sibley CT, Tracy R, Woodward M et al (2018) Metabolically healthy obesity, transition to metabolic syndrome, and cardiovascular risk. J Am Coll Cardiol 71:1857–1865. https://doi.org/10.1016/j.jacc.2018.02.055
Navarro-Gonzalez D, Sanchez-Inigo L, Fernandez-Montero A, Pastrana-Delgado J, Alfredo Martinez J (2016) Are all metabolically healthy individuals with obesity at the same risk of diabetes onset? Obesity (silver Spring) 24:2615–2623. https://doi.org/10.1002/oby.21667
Saltiel AR, Olefsky JM (2017) Inflammatory mechanisms linking obesity and metabolic disease. J Clin Invest 127:1–4. https://doi.org/10.1172/JCI92035
Slagter SN, Corpeleijn E, van der Klauw MM, Sijtsma A, Swart-Busscher LG, Perenboom CWM, de Vries JHM, Feskens EJM, Wolffenbuttel BHR, Kromhout D et al (2018) Dietary patterns and physical activity in the metabolically (un)healthy obese: the Dutch Lifelines cohort study. Nutr J 17:18. https://doi.org/10.1186/s12937-018-0319-0
Stefan N, Schick F, Haring HU (2017) Causes, characteristics, and consequences of metabolically unhealthy normal weight in humans. Cell Metab 26:292–300. https://doi.org/10.1016/j.cmet.2017.07.008
Wang B, Zhang M, Wang S, Wang C, Wang J, Li L, Zhang L, Ren Y, Han C, Zhao Y et al (2018) Dynamic status of metabolically healthy overweight/obesity and metabolically unhealthy and normal weight and the risk of type 2 diabetes mellitus: a cohort study of a rural adult Chinese population. Obes Res Clin Pract 12:61–71. https://doi.org/10.1016/j.orcp.2017.10.005
Wen CP, David Cheng TY, Tsai SP, Chan HT, Hsu HL, Hsu CC, Eriksen MP (2009) Are Asians at greater mortality risks for being overweight than Caucasians? Redefining obesity for Asians. Public Health Nutr 12:497–506. https://doi.org/10.1017/S1368980008002802
Whelton PK, Carey RM, Aronow WS, Casey Jr DE, Collins KJ, Himmelfarb CD, DePalma SM, Gidding S, Jamerson KA, Jones DW et al (2018) 2017 ACC/AHA/AAPA/ABC/ACPM/AGS/APhA/ASH/ASPC/NMA/PCNA guideline for the prevention, detection, evaluation, and management of high blood pressure in adults. A report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Hypertension 71:e13–e115. https://doi.org/10.1161/HYP.0000000000000065/
Wing RR, Lang W, Wadden TA, Safford M, Knowler WC, Bertoni AG, Hill JO, Brancati FL, Peters A, Wagenknecht L et al (2011) Benefits of modest weight loss in improving cardiovascular risk factors in overweight and obese individuals with type 2 diabetes. Diabetes Care 34:1481–1486. https://doi.org/10.2337/dc10-2415
Zhu J, Gao R, Zhao S, Lu G, Zhao D, Li J (2017) Guidelines for the prevention and treatment of dyslipidemia in adults in china (2016 revised edition). Chin J Health Manag 11:7–28
Acknowledgements
We acknowledge the financial support of the Shanghai Municipal Science and Technology Major Project (2017SHZDZX01), and the Science and Technology Commission of Shanghai Municipality (16JC1400500).
Funding
This work was supported by the Shanghai Municipal Science and Technology Major Project (2017SHZDZX01) and the Science and Technology Commission of Shanghai Municipality (16JC1400500).
Author information
Authors and Affiliations
Contributions
Study conception and design: XG, HT. Data collection: HL. Sample examination: QW, SM, HZ. Data management: QH. Data analysis: MX. Manuscript preparation: QW. Manuscript revision: GX and MX.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no conflicts of interest. HT is the associate editor of Phenomics, and he was not involved in reviewing this paper.
Ethics approval
This prospective cohort study from Shanghai Changfeng Community was approved by the Research Ethics Committee of the Shanghai Health Bureau, China.
Consent to participate
Each participant provided written informed consent.
Consent for publication
Not applicable.
Rights and permissions
About this article
Cite this article
Wu, Q., Huang, Qx., Zeng, Hl. et al. Prediction of Metabolic Disorders Using NMR-Based Metabolomics: The Shanghai Changfeng Study. Phenomics 1, 186–198 (2021). https://doi.org/10.1007/s43657-021-00021-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43657-021-00021-2