Abstract
Objectives
The accurate prediction of osteoarthritis (OA) severity in patients can be helpful to make the proper decision of intervention. This study aims to build up a powerful model to assess predictive risk factors and severity of knee osteoarthritis (KOA) in the clinical scenario.
Methods
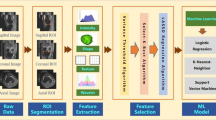
A total of 4796 KOA cases and 1205 features were selected by feature selections from the public OA database, Osteoarthritis Initiative (OAI). Six machine learning-based models were constructed and compared for the accuracy of OA prediction. The gradient-boosting decision tree was used to identify important prediction features in the extreme gradient boosting (XGBoost) model. The performance of models was evaluated by F1-score.
Results
Twenty features were determined as predictors for KOA risk and severity, including the subject characteristics, knee symptoms/risk factors and physical exam. The XGBoost model demonstrated 100% prediction accuracy for 54.7% of examined samples, and the remaining 45.3% of samples showed Kellgren and Lawrence (KL) gradings very close to the actual levels. It showed the highest prediction accuracy with an F1-score of 0.553 among the tested six models.
Conclusions
We demonstrate that the XGBoost is the best model for the prediction of KOA severity in the six examined models. In addition, 20 risk features were determined as the essential predictors of KOA, including the physical exam, knee symptoms/risk factors and subject characteristics, which may be useful for the identification of high-risk KOA cases and for making appropriate treatment decisions as well.





Similar content being viewed by others
Data availability
Data are from the Osteoarthritis Initiative (OAI) database, which is available upon request at https://nda.nih.gov/oai/.
References
Global, regional, and national incidence, prevalence, and years lived with disability for 301 acute and chronic diseases and injuries in 188 countries, 1990–2013: A systematic analysis for the Global Burden of Disease Study 2013. Lancet. 2015;386(9995):743–800. doi: https://doi.org/10.1016/s0140-6736(15)60692-4.
McGuire, D. A., Carter, T. R., & Shelton, W. R. (2002). Complex knee reconstruction: Osteotomies, ligament reconstruction, transplants, and cartilage treatment options. Arthroscopy, 18(9 Suppl 2), 90–103. https://doi.org/10.1053/jars.2002.36511
Peat, G., McCarney, R., & Croft, P. (2001). Knee pain and osteoarthritis in older adults: A review of community burden and current use of primary health care. Annals of the Rheumatic Diseases, 60(2), 91–97. https://doi.org/10.1136/ard.60.2.91
Blagojevic, M., Jinks, C., Jeffery, A., & Jordan, K. P. (2010). Risk factors for onset of osteoarthritis of the knee in older adults: A systematic review and meta-analysis. Osteoarthritis Cartilage, 18(1), 24–33. https://doi.org/10.1016/j.joca.2009.08.010
Chan, L. C., Li, H. H. T., Chan, P. K., & Wen, C. (2021). A machine learning-based approach to decipher multi-etiology of knee osteoarthritis onset and deterioration. Osteoarthritis and Cartilage Open, 3(1), 100135. https://doi.org/10.1016/j.ocarto.2020.100135
Karsdal, M. A., Michaelis, M., Ladel, C., Siebuhr, A. S., Bihlet, A. R., Andersen, J. R., et al. (2016). Disease-modifying treatments for osteoarthritis (DMOADs) of the knee and hip: Lessons learned from failures and opportunities for the future. Osteoarthritis Cartilage, 24(12), 2013–2021. https://doi.org/10.1016/j.joca.2016.07.017
Park, H. J., Kim, S. S., Lee, S. Y., Park, N. H., Park, J. Y., Choi, Y. J., et al. (2013). A practical MRI grading system for osteoarthritis of the knee: Association with Kellgren-Lawrence radiographic scores. European Journal of Radiology, 82(1), 112–117. https://doi.org/10.1016/j.ejrad.2012.02.023
Wellner, B., Grand, J., Canzone, E., Coarr, M., Brady, P. W., Simmons, J., et al. (2017). Predicting unplanned transfers to the intensive care unit: A machine learning approach leveraging diverse clinical elements. JMIR Med Informatics, 5(4), e45. https://doi.org/10.2196/medinform.8680
Lazzarini, N., Runhaar, J., Bay-Jensen, A. C., Thudium, C. S., Bierma-Zeinstra, S. M. A., Henrotin, Y., et al. (2017). A machine learning approach for the identification of new biomarkers for knee osteoarthritis development in overweight and obese women. Osteoarthritis Cartilage, 25(12), 2014–2021. https://doi.org/10.1016/j.joca.2017.09.001
Jamshidi, A., Pelletier, J. P., & Martel-Pelletier, J. (2019). Machine-learning-based patient-specific prediction models for knee osteoarthritis. Nature Reviews Rheumatology, 15(1), 49–60. https://doi.org/10.1038/s41584-018-0130-5
Faschingbauer, M., Kasparek, M., Waldstein, W., Schadler, P., Reichel, H., & Boettner, F. (2020). Cartilage survival of the knee strongly depends on malalignment: A survival analysis from the Osteoarthritis Initiative (OAI). Knee Surgery, Sports Traumatology, Arthroscopy, 28(5), 1346–1355. https://doi.org/10.1007/s00167-019-05434-1
Huang, Z., Guo, W., & Martin, J. T. (2021). Unsupervised and supervised machine learning for establishing back pain phenotypes: Data from the OAI. Osteoarthritis and Cartilage., 29, S300–S301. https://doi.org/10.1016/j.joca.2021.02.394
Eckstein, F., Hudelmaier, M., Wirth, W., Kiefer, B., Jackson, R., Yu, J., et al. (2006). Double echo steady state magnetic resonance imaging of knee articular cartilage at 3 Tesla: A pilot study for the Osteoarthritis Initiative. Annals of the Rheumatic Diseases, 65(4), 433–441. https://doi.org/10.1136/ard.2005.039370
Eckstein, F., Wirth, W., & Nevitt, M. C. (2012). Recent advances in osteoarthritis imaging–the osteoarthritis initiative. Nature Reviews Rheumatology, 8(10), 622–630. https://doi.org/10.1038/nrrheum.2012.113
Bany Muhammad, M., & Yeasin, M. (2021). Interpretable and parameter optimized ensemble model for knee osteoarthritis assessment using radiographs. Scientific Reports, 11(1), 14348. https://doi.org/10.1038/s41598-021-93851-z
Liu, L., Yu, Y., Fei, Z., Li, M., Wu, F.-X., Li, H.-D., et al. (2018). An interpretable boosting model to predict side effects of analgesics for osteoarthritis. BMC Systems Biology, 12(Suppl 6), 105. https://doi.org/10.1186/s12918-018-0624-4
Wang, Q. Q., Yu, S. C., Qi, X., Hu, Y. H., Zheng, W. J., Shi, J. X., et al. (2019). Overview of logistic regression model analysis and application. Zhonghua Yu Fang Yi Xue Za Zhi, 53(9), 955–960. https://doi.org/10.3760/cma.j.issn.0253-9624.2019.09.018
Huang, S., Cai, N., Pacheco, P. P., Narrandes, S., Wang, Y., & Xu, W. (2018). Applications of support vector machine (SVM) learning in cancer genomics. Cancer Genomics & Proteomics, 15(1), 41–51. https://doi.org/10.21873/cgp.20063
Hand, D. J., & Yu, K. (2001). Idiot’s bayes: Not so stupid after all? International Statistical Review, 69(3), 385–398. https://doi.org/10.2307/1403452
Du, Y., Almajalid, R., Shan, J., & Zhang, M. (2018). A novel method to predict knee osteoarthritis progression on MRI using machine learning methods. IEEE Transactions on Nanobioscience, 17(3), 228–236. https://doi.org/10.1109/tnb.2018.2840082
Chen, T., & Guestrin, C. (Ed.). (2016). Xgboost: A scalable tree boosting system. In Proceedings of the 22nd acm sigkdd international conference on knowledge discovery and data mining.
Gupta, J., Patrick, J., & Poon, S. (2019). Clinical safety incident taxonomy performance on C4.5 decision tree and random forest. Studies in Health Technology and Informatics, 266, 83–88. https://doi.org/10.3233/shti190777
Ntakolia, C., Kokkotis, C., Moustakidis, S., & Tsaopoulos, D. (2021). Prediction of joint space narrowing progression in knee osteoarthritis patients. Diagnostics (Basel). https://doi.org/10.3390/diagnostics11020285
Kokkotis, C., Moustakidis, S., Giakas, G., & Tsaopoulos, D. (2020). Identification of risk factors and machine learning-based prediction models for knee osteoarthritis patients. Applied Sciences, 10(19), 6797.
Weir, C., & Silk, B. (1992). Paramax systems corporation: MUC-4 test results and analysis. In Proceedings of the 4th conference on message understanding (pp. 128–131). McLean, VA: Association for Computational Linguistics.
Tseng, P. Y., Chen, Y. T., Wang, C. H., Chiu, K. M., Peng, Y. S., Hsu, S. P., et al. (2020). Prediction of the development of acute kidney injury following cardiac surgery by machine learning. Critical Care, 24(1), 478. https://doi.org/10.1186/s13054-020-03179-9
Kohn, M. D., Sassoon, A. A., & Fernando, N. D. (2016). Classifications in brief: Kellgren–Lawrence classification of osteoarthritis. Clinical Orthopaedics and Related Research, 474(8), 1886–1893. https://doi.org/10.1007/s11999-016-4732-4
Lane, N. E., Brandt, K., Hawker, G., Peeva, E., Schreyer, E., Tsuji, W., et al. (2011). OARSI-FDA initiative: Defining the disease state of osteoarthritis. Osteoarthritis Cartilage, 19(5), 478–482. https://doi.org/10.1016/j.joca.2010.09.013
Sundhedsstyrelsen. NKR og faglige visitationsretningslinjer: Knæartrose - ikke gældende 2012 [cited 2012 08 NOV]. https://www.sst.dk/da/Udgivelser/2012/NKR-Knaeartrose
Altman, R. D., & Gold, G. E. (2007). Atlas of individual radiographic features in osteoarthritis, revised. Osteoarthritis Cartilage, 15(Suppl A), A1-56. https://doi.org/10.1016/j.joca.2006.11.009
Chaisson, C. E., Gale, D. R., Gale, E., Kazis, L., Skinner, K., & Felson, D. T. (2000). Detecting radiographic knee osteoarthritis: What combination of views is optimal? Rheumatology (Oxford), 39(11), 1218–1221. https://doi.org/10.1093/rheumatology/39.11.1218
Pongsakonpruttikul, N., Angthong, C., Kittichai, V., Chuwongin, S., Puengpipattrakul, P., Thongpat, P., et al. (2022). Artificial intelligence assistance in radiographic detection and classification of knee osteoarthritis and its severity: A cross-sectional diagnostic study. European Review for Medical and Pharmacological Sciences, 26(5), 1549–1558. https://doi.org/10.26355/eurrev_202203_28220
Bonakdari, H., Jamshidi, A., Pelletier, J. P., Abram, F., Tardif, G., & Martel-Pelletier, J. (2021). A warning machine learning algorithm for early knee osteoarthritis structural progressor patient screening. Therapeutic Advances in Musculoskeletal Disease. https://doi.org/10.1177/1759720x21993254
Christodoulou, E., Moustakidis, S. P., Papandrianos, N. I., Tsaopoulos, D., & Papageorgiou, E. I. (2019). Exploring deep learning capabilities in knee osteoarthritis case study for classification. In 2019 10th international conference on information, intelligence, systems and applications (IISA) (pp. 1–6).
Kwon, S. B., Han, H. S., Lee, M. C., Kim, H. C., Ku, Y., & Ro, D. H. (2020). Machine learning-based automatic classification of knee osteoarthritis severity using gait data and radiographic images. IEEE Access, 8, 120597–120603. https://doi.org/10.1109/ACCESS.2020.3006335
Kokkotis, C., Ntakolia, C., Moustakidis, S., Giakas, G., & Tsaopoulos, D. (2022). Explainable machine learning for knee osteoarthritis diagnosis based on a novel fuzzy feature selection methodology. Physical and Engineering Sciences in Medicine, 45(1), 219–229. https://doi.org/10.1007/s13246-022-01106-6
Almhdie-Imjabbar, A., Nguyen, K. L., Toumi, H., Jennane, R., & Lespessailles, E. (2022). Prediction of knee osteoarthritis progression using radiological descriptors obtained from bone texture analysis and Siamese neural networks: Data from OAI and MOST cohorts. Arthritis Research & Therapy, 24(1), 66. https://doi.org/10.1186/s13075-022-02743-8
Khalid, A., Senan, E. M., Al-Wagih, K., Ali Al-Azzam, M. M., & Alkhraisha, Z. M. (2023). Hybrid techniques of X-ray analysis to predict knee osteoarthritis grades based on fusion features of CNN and handcrafted. Diagnostics (Basel)., 13(9), 1609. https://doi.org/10.3390/diagnostics13091609
Kim, B. Y., Kim, H. A., Jung, J. Y., Choi, S. T., Kim, J. M., Kim, S. H., et al. (2019). Clinical impact of the fracture risk assessment tool on the treatment decision for osteoporosis in patients with knee osteoarthritis: A multicenter comparative study of the fracture risk assessment tool and world health organization criteria. Journal of Clinical Medicine, 8(7), 918. https://doi.org/10.3390/jcm8070918
Acknowledgements
Data used in the preparation of this manuscript were obtained and analyzed from the controlled access data sets distributed from the Osteoarthritis Initiative (OAI), a data repository housed within the NIMH Data Archive (NDA). OAI is a collaborative informatics system created by the National Institute of Mental Health and the National Institute of Arthritis, Musculoskeletal and Skin Diseases (NIAMS) to provide a worldwide resource to quicken the pace of biomarker identification, scientific investigation and OA drug development. Data set identifier: NIMH Data Archive Digital Object Identifier (DOI): https://doi.org/10.15154/1528318.
Funding
This work has been supported by the Guangdong Provincial Talented Scholar Foundation (Grant Number 220418137), the National Natural Science Foundation of China (Grant Number 82173850) and University Innovative Team Support for Major Chronic Diseases and Drug Development (Grant Number 26330320901).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors have no relevant competing interests to disclose.
Ethical standard statement
A permission to access data in the Osteoarthritis Initiative permission group in the NIMH Data Archive (NDA) was applied for and has been approved, which is valid for a period of 1 year until 12/18/2022. No identifiable information of the participants is included in either the data or the manuscript. NDA data have been permanently deleted from all machines after the research was completed.
Informed consent
For this type of study informed consent is not required.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Su, K., Yuan, X., Huang, Y. et al. Improved Prediction of Knee Osteoarthritis by the Machine Learning Model XGBoost. JOIO 57, 1667–1677 (2023). https://doi.org/10.1007/s43465-023-00936-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43465-023-00936-0