Abstract
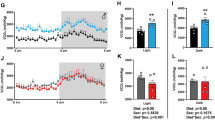
This study aims to determine the effects of Catha edulis (Vahl) Forssk. ex Endl., Celastraceae, and its bioactive alkaloid, cathinone, on liver histology, gene expression of inflammatory biomarkers in white adipose tissue, hypothalamus, and histomorphological bone parameters. Thirty male mice (C57BL/6J) were fed a high-fat diet for 8 weeks, divided into 5 groups, and treated daily for another 8 weeks high-fat diet + vehicle (control), HFD + 15 mg/kg orlistat, high-fat diet + 200 mg/kg plant extract, high-fat diet + 400 mg/kg plant extract, and high-fat diet + 3.2mg/kg cathinone. Treatments were carried out once daily by gastric gavage. Blood and tissue samples were collected for histological, biochemical, and gene expression analysis. The results showed the plant extract (400 mg/kg) and cathinone (3.2 mg/kg) displayed a marked reduction of liver fatty changes. All treated groups showed higher serum adiponectin levels compared to the control group (p <0.05). Plant extract–treated groups showed upregulation of adiponectin gene expression in white adipose tissue, however, at the concentration of 200 mg/kg upregulated interleukin-6 in the hypothalamus; in addition, this group showed less-eroded surfaces and higher bone mass compared to control. Cathinone-treated group exhibited increased osteoblast and osteoclast surfaces compared to the plant extract treatment. The study concluded that the plant extract and cathinone treatment are associated with minimal fat steatosis in the liver of obese mice probably by upregulation of anti-inflammatory adiponectin gene expression in adipose tissue. At the concentration of 200 mg/kg, the plant extract showed a protective effect on bone loss compared to orlistat, while cathinone promotes bone formation and resorption by a centrally mediated mechanism.
Graphical Abstract






Similar content being viewed by others
References
Al-Ghamdi S (2012) The reproductive and thyroid hormonal profile of khat (Catha edulis) chewers. Pharmacol Pharm 3:447–452. https://doi.org/10.4236/pp.2012.34060
Al-Zubairi A, Ismail P, Pei PC (2019) Rahmat A (2008) Genotoxic effect of Catha edulis (khat) crude extract after sub-chronic administration in rats. Environ Toxicol Pharmacol 25:298–303. https://doi.org/10.1016/j.etap.2007.10.032. Accessed 03 Dec 2019
Al-Zubairi AS, Ismail P, Chong PP, Abdul AB, Ali RS, Wahab SIA, Rahmat A (2008b) Short-term repeated dose biochemical effects of Catha edulis (khat) crude extract administration in rats. Int J Trop Med 3:19–25
Alshagga MA, Alshawsh MA, Seyedan A, Alsalahi A, Pan Y, Mohankumar SK, Alkebsi A, Kassim S, Mohamed Z (2016) Khat (Catha edulis) and obesity: a scoping review of animal and human studies. Ann Nutr Metab 69:200–211. https://doi.org/10.1159/000452895
Alshagga MA, Mohamed Z, Seyedan A, Ebling FJP, Alshawsh MA (2020) Khat (Catha edulis) upregulates lipolytic genes in white adipose tissue of male obese mice (C57BL/6J). J Ethnopharmacol 262:113187. https://doi.org/10.1016/j.jep.2020.113187
Balistreri CR, Caruso C, Candore G (2010) The role of adipose tissue and adipokines in obesity-related inflammatory diseases. Mediat Inflamm 2010:802078. https://doi.org/10.1155/2010/802078
Bancroft JD, Layton C, Suvarna SK (2018) Bancroft’s Theory and practice of histological techniques. 8th ed, Elsevier Inc. https://doi.org/10.1016/s0031-3025(16)35811-1
Bobbo VCD, Jara CP, Mendes NF, Morari J, Velloso LA, Araújo EP (2019) Interleukin-6 expression by hypothalamic microglia in multiple inflammatory contexts: a systematic review. Biomed Res Int 2019:1365210. https://doi.org/10.1155/2019/1365210
Coenen KR, Hasty AH (2007) Obesity potentiates development of fatty liver and insulin resistance, but not atherosclerosis, in high-fat diet-fed agouti LDLR-deficient mice. Am J Physiol-Endoc M 293:492–499. https://doi.org/10.1152/ajpendo.00171.2007
Dasgupta A (2019) Chapter 33 Abuse of magic mushroom, peyote cactus, LSD, khat, and volatiles, in: Critical Issues in Alcohol and Drugs of Abuse Testing. 2nd ed. Elsevier, pp. 477–494. https://doi.org/10.1016/B978-0-12-815607-0.00033-2
de Souza GO, Wasinski F, Donato JJ (2022) Characterization of the metabolic differences between male and female C57BL/6 mice. Life Sci 301:120636. https://doi.org/10.1016/j.lfs.2022.120636
Drew BS, Dixon AF, Dixon JB (2007) Obesity management: update on orlistat. Vasc Health Risk Manag 3:817–821
El-Sayed MIK, Amin HAK (2012) Effect of Catha edulis on insulin, resistin and cortisol levels in type-2 diabetics and nondiabetics. Am J Biochem Biotechnol 8:157–163. https://doi.org/10.3844/ajbbsp.2012.157.163
Engidawork E (2017) Pharmacological and toxicological effects of Catha edulis F. (Khat). Phytother Res 31:1019–1028. https://doi.org/10.1002/ptr.5832
Ferrucci M, Limanaqi F, Ryskalin L, Biagioni F, Busceti CL, Fornai F (2019) The effects of amphetamine and methamphetamine on the release of norepinephrine, dopamine and acetylcholine from the brainstem reticular formation. Front Neuroanat 13:48. https://doi.org/10.3389/fnana.2019.00048
Gómez-Ambrosi J, Rodríguez A, Catalán V, Frühbeck G (2008) The bone-adipose axis in obesity and weight loss. Obes Surg 18:1134–1143. https://doi.org/10.1007/s11695-008-9548-1
Griffin C, Hutch CR, Abrishami S, Stelmak D, Eter L, Li Z, Chang E, Agarwal D, Zamarron B, Varghese M, Subbaiah P, MacDougald OA, Sandoval DA, Singer K (2019) Inflammatory responses to dietary and surgical weight loss in male and female mice. Biol Sex Differ 10:16. https://doi.org/10.1186/s13293-019-0229-7
Guo X, Cheng L, Yang S, Che H (2019) Pro-inflammatory immunological effects of adipose tissue and risk of food allergy in obesity: focus on immunological mechanisms. Allergol Immunopathol 48:306–312. https://doi.org/10.1016/j.aller.2019.06.004
Halade GV, El Jamali A, Williams PJ, Fajardo RJ, Fernandes G (2011) Obesity-mediated inflammatory microenvironment stimulates osteoclastogenesis and bone loss in mice. Exp Gerontol 46:43–52. https://doi.org/10.1016/j.exger.2010.09.014
Idelevich A, Sato K, Avihai B, Nagano K, Galien A, Rowe G, Gori F, Baron R (2020) Both NPY-expressing and CART-expressing neurons increase energy expenditure and trabecular bone mass in response to AP1 antagonism, but have opposite effects on bone resorption. J Bone Miner Res 35:1107–1118. https://doi.org/10.1002/jbmr.3967
Kalyanasundar B, Perez CI, Arroyo B, Moreno MG, Gutierrez R (2020) The appetite suppressant D-norpseudoephedrine (cathine) acts via D1/D2-like dopamine receptors in the nucleus accumbens shell. Front Neurosci 14:572328. https://doi.org/10.3389/fnins.2020.572328
Khan RA, Kapur P, Jain A, Farah F, Bhandari U (2017) Effect of orlistat on periostin, adiponectin, inflammatory markers and ultrasound grades of fatty liver in obese NAFLD patients. Ther Clin Risk Manag 13:139–149. https://doi.org/10.2147/2FTCRM.S124621
Kotzbeck P, Giordano A, Mondini E, Murano I, Severi I, Venema W, Cecchini MP, Kershaw EE, Barbatelli G, Haemmerle G, Zechner R, Cinti S (2018) Brown adipose tissue whitening leads to brown adipocyte death and adipose tissue inflammation. J Lipid Res 59:784–794. https://doi.org/10.1194/jlr.M079665
Lecka-Czernik B, Stechschulte LA, Czernik PJ, Dowling AR (2015) High bone mass in adult mice with diet-induced obesity results from a combination of initial increase in bone mass followed by attenuation in bone formation; implications for high bone mass and decreased bone quality in obesity. Mol Cell Endocrinol 410:35–41. https://doi.org/10.1016/j.mce.2015.01.001
Lemieux AM, Li B, Al-Absi M (2015) Khat use and appetite: an overview and comparison of amphetamine, khat and cathinone. J Ethnopharmacol 160:78–85. https://doi.org/10.1016/j.jep.2014.11.002
Lim SYM, Alshagga MA, Alshawsh MA, Ong CE, Pan Y (2021) In vitro effects of 95% khat ethanol extract (KEE) on human recombinant cytochrome P450 (CYP)1A2, CYP2A6, CYP2B6, CYP2C8, CYP2C19, CYP2E1, CYP2J2, and CYP3A5. Drug Metab Pers Ther 37:55–67. https://doi.org/10.1515/dmpt-2021-1000196
Liu X, Tong W, Zhao X, Zhang H, Tang Y, Deng X (2017) Chinese herb extract improves liver steatosis by promoting the expression of high molecular weight adiponectin in NAFLD rats. Mol Med Rep 16:5580–5586. https://doi.org/10.3892/mmr.2017.7284
Lu XM, Zhao H, Wang EH (2013) A high-fat diet induces obesity and impairs bone acquisition in young male mice. Mol Med Rep 7:1203–1208. https://doi.org/10.3892/mmr.2013.1297
Maïmoun L, Mura T, Leprieur E, Avignon A, Mariano-Goulart D, Sultan A (2016) Impact of obesity on bone mass throughout adult life: influence of gender and severity of obesity. Bone 90:23–30. https://doi.org/10.1016/j.bone.2015.11.020
Milan G, Granzotto M, Scarda A, Calcagno A, Pagano C, Federspil G, Vettor R (2002) Resistin and adiponectin expression in visceral fat of obese rats: effect of weight loss. Obes Res 10:1095–1103. https://doi.org/10.1038/oby.2002.149
Mohammed A, Engidawork E (2011) Reproductive parameters are differentially altered following subchronic administration of Catha edulis F. (Khat) extract and cathinone in male rats. J Ethnopharmacol 134:977–983. https://doi.org/10.1016/j.jep.2011.02.006
Mwenda JM, Owuor RA, Kyama CM, Wango EO, M’Arimi M, Langat DK (2006) Khat (Catha edulis) up-regulates testosterone and decreases prolactin and cortisol levels in the baboon. J Ethnopharmacol 103:379–384. https://doi.org/10.1016/j.jep.2005.08.016
National Research Council (2011) Guide for the Care and Use of Laboratory Animals. 8th ed., National Academy of Sciences. https://grants.nih.gov/grants/olaw/guide-for-the-care-and-use-of-laboratory-animals.pdf
Orlien SMS, Sandven I, Berhe NB, Ismael NY, Ahmed TA, Stene-Johansen K, Gundersen SG, Morgan MY, Johannessen A (2018) Khat chewing increases the risk for developing chronic liver disease: a hospital-based case–control study. Hepatology 68:248–257. https://doi.org/10.1002/hep.29809
Picchi MG, de Mattos AM, Barbosa MR, Duarte CP, de Gandini M, A, Portari GV, Jordão AA (2011) A high-fat diet as a model of fatty liver disease in rats. Acta Cir Bras 26:25–30. https://doi.org/10.1590/s0102-86502011000800006
Ramos EJB, Xu Y, Romanova I, Middleton F, Chen C, Quinn R, Inui A, Das U, Meguid MM (2003) Is obesity an inflammatory disease? Surgery 134:329–335. https://doi.org/10.1067/msy.2003.267
Saif A, Norazlina M, Ima-Nirwana S (2016) Quantification of bone histomorphometric parameters using the weibel technique in animals. Med Health 11:278–288. https://doi.org/10.17576/MH.2016.1102.16
Tencerova M, Figeac F, Ditzel N, Taipaleenmäki H, Nielsen TK, Kassem M (2018) High-Fat diet–induced obesity promotes expansion of bone marrow adipose tissue and impairs skeletal stem cell functions in mice. J Bone Miner Res 33:1154–1165. https://doi.org/10.1002/jbmr.3408
Ullah R, Rauf N, Nabi G, Yi S, Yu-Dong Z, Fu J (2021) Mechanistic insight into high-fat diet-induced metabolic inflammation in the arcuate nucleus of the hypothalamus. Biomed Pharmacother 142:112012. https://doi.org/10.1016/j.biopha.2021.112012
Vinokur Y, Levi A, Feygenberg O, Rodov V (2008) Hydrophilic and lipophilic antioxidant capacity and content of phenolic compounds in fresh khat leaves (Catha edulis Forsk.). Ethnobot Leafl 12:557–564. https://opensiuc.lib.siu.edu/cgi/viewcontent.cgi?article=1108&context=ebl&httpsredir=1&referer= Accessed 03 Dec 2019
Wellen KE, Hotamisligil GS (2003) Obesity-induced inflammatory changes in adipose tissue. J Clin Invest 112:1785–1788. https://doi.org/10.1172/JCI20514
West KS, Lawson V, Swanson AM, Dunigan AI, Roseberry AG (2019) Amphetamine dose-dependently decreases and increases binge intake of fat and sucrose independent of sex. Obesity 27:1874–1882. https://doi.org/10.1002/2Foby.22636
Xia B, Shi XC, Xie BC, Zhu MQ, Chen Y, Chu XY, Cai GH, Liu M, Yang SZ, Mitchell GA, Pang WJ, Wu JW 2020. Urolithin A exerts antiobesity effects through enhancing adipose tissue thermogenesis in mice. PLoS Biol 18:e3000688. https://doi.org/10.1371/journal.pbio.3000688
Yibrah M, Negesso AE, Gebregziabher A, Challa F, Mudi K, Tesfay F, Gebretsadkan M, Kinde S, Asmelash D (2019) Gonadal and cortisol hormone profile among male chronic khat, marijuana, and heroin abuses. Int J Endocrinol 2019:4178241. https://doi.org/10.1155/2019/4178241
Yu Y, Miller R, Groth SW (2022) A literature review of dopamine in binge eating. J Eat Disord 10:11. https://doi.org/10.1186/s40337-022-00531-y
Zelber-Sagi S, Kessler A, Brazowsky E, Webb M, Lurie Y, Santo M, Leshno M, Blendis L, Halpern Z, Oren R (2006) A double-blind randomized placebo-controlled trial of orlistat for the treatment of nonalcoholic fatty liver disease. Clin Gastroenterol Hepatol 4:639–644. https://doi.org/10.1016/j.cgh.2006.02.004
Acknowledgements
The authors would like to thank University Kebangsaan Malaysia (UKM) for the continuous support.
Funding
This research was supported by research grants from the University of Malaya, projects number ST070-2021 and PG006-2015A.
Author information
Authors and Affiliations
Contributions
MAA, MA Alshawsh, NM, YP, SYML, and ZM designed the study. MAA and AS performed the experiment on obese mice. MAA and NM performed bone analysis. MAA, MA Alshawsh, and NM analyzed the data. MAA drafted the manuscript. MA Alshawsh, NM, YP, SYML, and ZM revised the paper. All authors read and accept the final version of the manuscript.
Corresponding author
Ethics declarations
Ethical Approval
The Faculty of Medicine approved all protocols in this section, the University of Malaya’s Animal Ethics Committee approved the protocol with ethical approval number (2014-08-05/PHAR/R/MAMA). All animals were handled as instructed in the “Guide of Care and Use of Laboratory Animals” criteria of the National Academy of Sciences (Academy and of Sciences , 2011).
Conflict of Interest
The authors declare no competing interests.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Alshagga, M.A., Mohamed, Z., Mohamed, N. et al. Catha edulis and Cathinone Effect on Bone Parameters and Adipokines Gene Expression in Obese Male Mice. Rev. Bras. Farmacogn. 33, 1012–1021 (2023). https://doi.org/10.1007/s43450-023-00428-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43450-023-00428-0