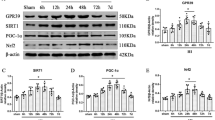
Abstract
The protective anti-inflammatory effects of glycyrrhizic acid have been demonstrated in many diseases. This study aimed to explore the neuroprotective role of glycyrrhizic acid in ras-related C3 botulinum toxin substrate 1/signal transducer and activator of transcription factor 3 signalling-mediated neuroinflammation after hypoxia-ischemia insult in neonatal mice. A mouse model of neonatal hypoxic-ischemic encephalopathy was constructed. The animals were randomly divided into three groups: (1) Sham, (2) hypoxia-ischemia, and (3) hypoxia-ischemia + glycyrrhizic acid. Behavioural tests were used to assess the motor and cognitive functions of the different groups. The expression of proinflammatory cytokines and ras-related C3 botulinum toxin substrate 1/signal transducer and activator of transcription factor 3 signalling were detected by quantitative real-time polymerase chain reaction and western blotting. The morphological features of brain tissues were observed by haematoxylin-eosin staining. Glycyrrhizic acid attenuated the reduction in weight gain and brain oedema and prevented neurological behaviour deficits after hypoxia-ischemia insult. The secretion of proinflammatory cytokines was reduced by glycyrrhizic acid treatment. The related proteins of ras-related C3 botulinum toxin substrate 1/signal transducer and activator of transcription factor 3 signalling were inhibited by glycyrrhizic acid treatment after hypoxia-ischemia insult. Our study indicates that glycyrrhizic acid promotes neurofunctional recovery and relieves neuroinflammation-induced brain injury by suppressing the ras-related C3 botulinum toxin substrate 1/signal transducer and activator of transcription factor 3 signalling pathway, which may provide a novel therapeutic strategy for ameliorating neonatal hypoxic-ischemic encephalopathy.
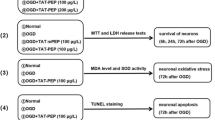
Graphical Abstract






Similar content being viewed by others
Data Availability
The data sets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Ahmed HA, Ishrat T, Pillai B, Fouda AY, Sayed MA, Eldahshan W, Waller JL, Ergul A, Fagan SC (2018) RAS modulation prevents progressive cognitive impairment after experimental stroke: a randomized, blinded preclinical trial. J Neuroinflammation 15:229. https://doi.org/10.1186/s12974-018-1262-x
Akutagawa K, Fujita T, Ouhara K, Takemura T, Tari M, Kajiya M, Matsuda S, Kuramitsu S, Mizuno N, Shiba H, Kurihara H (2019) Glycyrrhizic acid suppresses inflammation and reduces the increased glucose levels induced by the combination of Porphyromonas gulae and ligature placement in diabetic model mice. Int Immunopharmacol 68:30–38. https://doi.org/10.1016/j.intimp.2018.12.045
Bartha AI, Foster-Barber A, Miller SP, Vigneron DB, Glidden DV, Barkovich AJ, Ferriero DM (2004) Neonatal encephalopathy: association of cytokines with MR spectroscopy and outcome. Pediatr Res 56:960–966. https://doi.org/10.1203/01.PDR.0000144819.45689.BB
Bassi R, Giussani P, Anelli V, Colleoni T, Pedrazzi M, Patrone M, Viani P, Sparatore B, Melloni E, Riboni L (2008) HMGB1 as an autocrine stimulus in human T98G glioblastoma cells: role in cell growth and migration. J Neuro-Oncol 87:23–33. https://doi.org/10.1007/s11060-007-9488-y
Chen H et al (2020) Glycyrrhizin prevents hemorrhagic transformation and improves neurological outcome in ischemic stroke with delayed thrombolysis through targeting peroxynitrite-mediated HMGB1 signaling. Transl Stroke Res 11:967–982. https://doi.org/10.1007/s12975-019-00772-1
Cheng G, Diebold BA, Hughes Y, Lambeth JD (2006) Nox1-dependent reactive oxygen generation is regulated by Rac1. J Biol Chem 281:17718–17726. https://doi.org/10.1074/jbc.M512751200
Dell’Albani P, Santangelo R, Torrisi L, Nicoletti VG, de Vellis J, Giuffrida Stella AM (2001) JAK/STAT signaling pathway mediates cytokine-induced iNOS expression in primary astroglial cell cultures. J Neurosci Res 65:417–424. https://doi.org/10.1002/jnr.1169
Douglas-Escobar M, Weiss MD (2015) Hypoxic-Ischemic Encephalopathy: a Review for the Clinician. JAMA Pediatr 169:397–403. https://doi.org/10.1001/jamapediatrics.2014.3269
Downen M, Amaral TD, Hua LL, Zhao ML, Lee SC (1999) Neuronal death in cytokine-activated primary human brain cell culture: role of tumor necrosis factor-alpha. Glia 28:114–127. https://doi.org/10.1002/(SICI)1098-1136(199911)28:2%3c114::AID-GLIA3%3e3.0.CO;2-O
Ekanayaka SA, McClellan SA, Barrett RP, Kharotia S, Hazlett LD (2016) Glycyrrhizin reduces HMGB1 and bacterial load in Pseudomonas aeruginosa keratitis. Invest Ophthalmol Vis Sci 57:5799–5809. https://doi.org/10.1167/iovs.16-20103
Erlandsson Harris H, Andersson U (2004) Mini-review: the nuclear protein HMGB1 as a proinflammatory mediator. Eur J Immunol 34:1503–1512. https://doi.org/10.1002/eji.200424916
Faruqi TR, Gomez D, Bustelo XR, Bar-Sagi D, Reich NC (2001) Rac1 mediates STAT3 activation by autocrine IL-6. Proc Natl Acad Sci USA 98:9014–9019. https://doi.org/10.1073/pnas.161281298
Finder M, Boylan GB, Twomey D, Ahearne C, Murray DM, Hallberg B (2020) Two-year neurodevelopmental outcomes after mild hypoxic ischemic encephalopathy in the era of therapeutic hypothermia. JAMA Pediatr 174:48–55. https://doi.org/10.1001/jamapediatrics.2019.4011
Hagberg H, Mallard C, Ferriero DM, Vannucci SJ, Levison SW, Vexler ZS, Gressens P (2015) The role of inflammation in perinatal brain injury. Nat Rev Neurol 11:192–208. https://doi.org/10.1038/nrneurol.2015.13
Harraz MM, Marden JJ, Zhou W, Zhang Y, Williams A, Sharov VS, Nelson K, Luo M, Paulson H, Schöneich C, Engelhardt JF (2008) SOD1 mutations disrupt redox-sensitive Rac regulation of NADPH oxidase in a familial ALS model. J Clin Invest 118:659–670. https://doi.org/10.1172/JCI34060
Hua Y, Schallert T, Keep RF, Wu J, Hoff JT, Xi G (2002) Behavioral tests after intracerebral hemorrhage in the rat. Stroke 33:2478–2484. https://doi.org/10.1161/01.str.0000032302.91894.0f
Hua S, Ma M, Fei X, Zhang Y, Gong F, Fang M (2019) Glycyrrhizin attenuates hepatic ischemia-reperfusion injury by suppressing HMGB1-dependent GSDMD-mediated kupffer cells pyroptosis. Int Immunopharmacol 68:145–155. https://doi.org/10.1016/j.intimp.2019.01.002
Jiang J-X, Zhang S-J, Shen H-J, Guan Y, Liu Q, Zhao W, Jia Y-L, Shen J, Yan X-F, Xie Q-M (2017) Rac1 signaling regulates cigarette smoke-induced inflammation in the lung via the Erk1/2 MAPK and STAT3 pathways. BBA|-Mol Basis Dis 1863:1778–1788. https://doi.org/10.1016/j.bbadis.2017.04.013
Karabiyik C, Fernades R, Figueiredo FR, Socadato R, Brakebusch C, Lambertsen KL, Relvas JB, Santos SD (2018) Neuronal Rho GTPase Rac1 elimination confers neuroprotection in a mouse model of permanent ischemic stroke. Brain Pathol 28:569–580. https://doi.org/10.1111/bpa.12562
Khan R, Rehman MU, Khan AQ, Tahir M, Sultana S (2018) Glycyrrhizic acid suppresses 1,2-dimethylhydrazine-induced colon tumorigenesis in Wistar rats: alleviation of inflammatory, proliferation, angiogenic, and apoptotic markers. Environ Toxicol 33:1272–1283. https://doi.org/10.1002/tox.22635
Kim HS, Ye S-K, Cho IH, Jung JE, Kim D-H, Choi S, Kim Y-S, Park C-G, Kim T-Y, Lee JW, Chung MH (2006) 8-Hydroxydeoxyguanosine suppresses NO production and COX-2 activity via Rac1/STATs signaling in LPS-induced brain microglia. Free Radic Bio Med 41:1392–1403. https://doi.org/10.1016/j.freeradbiomed.2006.07.018
Kim SW, Jin Y, Shin J-H, Kim I-D, Lee H-K, Park S, Han P-L, Lee J-K (2012) Glycyrrhizic acid affords robust neuroprotection in the postischemic brain via anti-inflammatory effect by inhibiting HMGB1 phosphorylation and secretion. Neurobiol Dis 46:147–156. https://doi.org/10.1016/j.nbd.2011.12.056
Kuniyasu H, Yano S, Sasaki T, Sasahira T, Sone S, Ohmori H (2005) Colon cancer cell-derived high mobility group 1/amphoterin induces growth inhibition and apoptosis in macrophages. Am J Pathol 166:751–760. https://doi.org/10.1016/S0002-9440(10)62296-1
Kurinczuk JJ, White-Koning M, Badawi N (2010) Epidemiology of neonatal encephalopathy and hypoxic-ischaemic encephalopathy. Early Hum Dev 86:329–338. https://doi.org/10.1016/j.earlhumdev.2010.05.010
Lamprecht R, LeDoux J (2004) Structural plasticity and memory. Nat Rev Neurosci 5:45–54. https://doi.org/10.1038/nrn1301
Le K, Chibaatar Daliv E, Wu S, Qian F, Ali AI, Yu D, Guo Y (2019) SIRT1-regulated HMGB1 release is partially involved in TLR4 signal transduction: a possible anti-neuroinflammatory mechanism of resveratrol in neonatal hypoxic-ischemic brain injury. Int Immunopharmacol 75:105779. https://doi.org/10.1016/j.intimp.2019.105779
Le K, Wu S, Chibaatar E, Ali AI, Guo Y (2020) Alarmin HMGB1 plays a detrimental role in hippocampal dysfunction caused by hypoxia-ischemia insult in neonatal mice: evidence from the application of the HMGB1 inhibitor glycyrrhizin. ACS Chem Neurosci 11:979–993. https://doi.org/10.1021/acschemneuro.0c00084
Leerach N, Harashima A, Munesue S, Kimura K, Oshima Y, Goto H, Yamamoto H, Higashida H, Yamamoto Y (2021) Glycation reaction and the role of the receptor for advanced glycation end-products in immunity and social behavior. Glycoconjugate J 38:303–310. https://doi.org/10.1007/s10719-020-09956-6
Liu F, McCullough LD (2013) Inflammatory responses in hypoxic ischemic encephalopathy. Acta Pharmacol Sin 34:1121–1130. https://doi.org/10.1038/aps.2013.89
Liu W, Huang J, Doycheva D, Gamdzyk M, Tang J, Zhang JH (2019) RvD1binding with FPR2 attenuates inflammation via Rac1/NOX2 pathway after neonatal hypoxic-ischemic injury in rats. Exp Neurol 320:112982. https://doi.org/10.1016/j.expneurol.2019.112982
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods 25:402–408. https://doi.org/10.1006/meth.2001.1262
Marei H, Malliri A (2017) Rac1 in human diseases: the therapeutic potential of targeting Rac1 signaling regulatory mechanisms. Small GTPases 8:139–163. https://doi.org/10.1080/21541248.2016.1211398
Pang H, Huang T, Song J, Li D, Zhao Y, Ma X (2016) Inhibiting HMGB1 with glycyrrhizic acid protects brain injury after DAI via its anti-inflammatory effect. Mediat Inflamm 2016:4569521. https://doi.org/10.1155/2016/4569521
Park EJ, Ji KA, Jeon S-B, Choi W-H, Han I-O, You H-J, Kim J-H, Jou I, Joe E-H (2004) Rac1 contributes to maximal activation of STAT1 and STAT3 in IFN-gamma-stimulated rat astrocytes. J Immunol 173:5697–5703. https://doi.org/10.4049/jimmunol.173.9.5697
Planas AM, Gorina R, Chamorro A (2006) Signalling pathways mediating inflammatory responses in brain ischaemia. Biochem Soc T 34:1267–1270. https://doi.org/10.1042/BST0341267
Qu L, Chen D, He W, Chen Y, Li Y, Wen Y, Zhou S, Jiang Y, Yang X, Zhang R, Shen L (2019) Glycyrrhizic acid ameliorates LPS-induced acute lung injury by regulating autophagy through the PI3K/AKT/mTOR pathway. Am J Transl Res 11:2042–2055
Ravichandran A, Low BC (2013) SmgGDS antagonizes BPGAP1-induced Ras/ERK activation and neuritogenesis in PC12 cell differentiation. Mol Biol Cell 24:145–156. https://doi.org/10.1091/mbc.E12-04-0300
Shao M, Tang ST, Liu B, Zhu HQ (2016) Rac1 mediates HMGB1induced hyperpermeability in pulmonary microvascular endothelial cells via MAPK signal transduction. Mol Med Rep 13:529–535. https://doi.org/10.3892/mmr.2015.4521
Shen L, Cui Z, Lin Y, Wang S, Zheng D, Tan Q (2015) Anti-inflammative effect of glycyrrhizin on rat thermal injury via inhibition of high-mobility group box 1 protein. Burns : J Int Soc Burn Injuries 41:372–378. https://doi.org/10.1016/j.burns.2014.05.008
Simon AR, Vikis HG, Stewart S, Fanburg BL, Cochran BH, Guan KL (2000) Regulation of STAT3 by direct binding to the Rac1 GTPase. Science 290:144–147. https://doi.org/10.1126/science.290.5489.144
Singh V, Roth S, Veltkamp R, Liesz A (2016) HMGB1 as a key mediator of immune mechanisms in ischemic stroke antioxid redox. Signal 24:635–651. https://doi.org/10.1089/ars.2015.6397
Stankiewicz TR, Linseman DA (2014) Rho family GTPases: key players in neuronal development, neuronal survival, and neurodegeneration. Front Cell Neurosci 8:314. https://doi.org/10.3389/fncel.2014.00314
Su X, Wu L, Hu M, Dong W, Xu M, Zhang P (2017) Glycyrrhizic acid: a promising carrier material for anticancer therapy. Biomed Pharmacother 95:670–678. https://doi.org/10.1016/j.biopha.2017.08.123
Sugimoto K, Ohkawara H, Nakamura Y, Takuwa Y, Ishibashi T, Takeishi Y (2014) Receptor for advanced glycation end products - membrane type1 matrix metalloproteinase axis regulates tissue factor expression via RhoA and Rac1 activation in high-mobility group box-1 stimulated endothelial cells. PloS One 9:e114429. https://doi.org/10.1371/journal.pone.0114429
Sun CX, Downey GP, Zhu F, Koh AL, Thang H, Glogauer M (2004) Rac1 is the small GTPase responsible for regulating the neutrophil chemotaxis compass. Blood 104:3758–3765. https://doi.org/10.1182/blood-2004-03-0781
Sun ZG, Zhao TT, Lu N, Yang YA, Zhu HL (2019) Research progress of glycyrrhizic acid on antiviral activity. Mini-Rev Med Chem 19:826–832. https://doi.org/10.2174/1389557519666190119111125
Teng TS, Lin B, Manser E, Ng DC, Cao X (2009) Stat3 promotes directional cell migration by regulating Rac1 activity via its activator betaPIX. J Cell Sci 122:4150–4159. https://doi.org/10.1242/jcs.057109
Tourette C, Li B, Bell R, O’Hare S, Kaltenbach LS, Mooney SD, Hughes RE (2014) A large scale Huntingtin protein interaction network implicates Rho GTPase signaling pathways in Huntington disease. J Biol Chem 289:6709–6726. https://doi.org/10.1074/jbc.M113.523696
Tuttolomondo A, Di Raimondo D, di Sciacca R, Pinto A, Licata G (2008) Inflammatory cytokines in acute ischemic stroke. Curr Pharm Des 14:3574–3589. https://doi.org/10.2174/138161208786848739
Vannucci RC, Vannucci SJ (2005) Perinatal hypoxic-ischemic brain damage: evolution of an animal model. Dev Neurosci-Basel 27:81–86. https://doi.org/10.1159/000085978
Wang XJ, Kong KM, Qi WL, Ye WL, Song PS (2005) Interleukin-1 beta induction of neuron apoptosis depends on p38 mitogen-activated protein kinase activity after spinal cord injury. Acta Pharmacol Sin 26:934–942. https://doi.org/10.1111/j.1745-7254.2005.00152.x
Wassink G, Davidson JO, Dhillon SK, Zhou K, Bennet L, Thoresen M, Gunn AJ (2019) Therapeutic hypothermia in neonatal hypoxic-ischemic encephalopathy. Curr Neurol Neurosci 19:2. https://doi.org/10.1007/s11910-019-0916-0
Williams LM, Lali F, Willetts K, Balague C, Godessart N, Brennan F, Feldmann M, Foxwell BMJ (2008) Rac mediates TNF-induced cytokine production via modulation of NF-kappaB. Mol Immunol 45:2446–2454. https://doi.org/10.1016/j.molimm.2007.12.011
Wood T, Osredkar D, Puchades M, Maes E, Falck M, Flatebø T, Walløe L, Sabir H, Thoresen M (2016) Treatment temperature and insult severity influence the neuroprotective effects of therapeutic hypothermia. Sci Rep-UK 6:23430. https://doi.org/10.1038/srep23430
Xu C, Liang C, Sun W, Chen J, Chen X (2018) Glycyrrhizic acid ameliorates myocardial ischemic injury by the regulation of inflammation and oxidative state. Drug Des Dev Ther 12:1311–1319. https://doi.org/10.2147/DDDT.S165225
Yan S, Fang C, Cao L, Wang L, Du J, Sun Y, Tong X, Lu Y, Wu X (2019) Protective effect of glycyrrhizic acid on cerebral ischemia/reperfusion injury via inhibiting HMGB1-mediated TLR4/NF-kappaB pathway. Biotechnol Appl Bioc 66:1024–1030. https://doi.org/10.1002/bab.1825
Yildiz EP, Ekici B, Tatli B (2017) Neonatal hypoxic ischemic encephalopathy: an update on disease pathogenesis and treatment. Expert Rev Neurother 17:449–459. https://doi.org/10.1080/14737175.2017.1259567
Zhou K, Zhou Z-H, Yao X-H, Wu F, Yang J, Yang L, Zhang X, Cui Y-H, Bian X-W, Shi Y, Ping Y-F (2018) RAC1-GTP promotes epithelial-mesenchymal transition and invasion of colorectal cancer by activation of STAT3. Lab Invest 98:989–998. https://doi.org/10.1038/s41374-018-0071-2
Zhu F, Wang KB, Rui L (2019) STAT3 Activation and ncogenesis in lymphoma. Cancers 12:19. https://doi.org/10.3390/cancers12010019
Ziemka-Nalecz M, Jaworska J, Zalewska T (2017) Insights into the neuroinflammatory responses after neonatal hypoxia-ischemia. J Neuropathol Exp Neurol 76:644–654. https://doi.org/10.1093/jnen/nlx046
Acknowledgements
We acknowledged the administrative and facility support by Institute of Neurology, Comprehensive Laboratory of Basic Medicine, Southeast University, China.
Funding
This study was supported by the National Natural Science Foundation of China (grant no. 82060290 and no.81471187) and the Natural Science Foundation of Jiangxi Province (grant no. 20212BAB216040).
Author information
Authors and Affiliations
Contributions
GY designed the study. WS and LK performed the experiments. WS drafted the initial manuscript. GY and LK edited and revised the whole manuscript. All authors read and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Ethics Declarations
All experiments were approved by the Animal Ethical and Welfare Committee of Southeast University (Animal Ethics No.20190215016).
Additional information
Yijing Guo, Shanshan Wu, and Kai Le contributed equally to this work as co-first authors
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Guo, Y., Wu, S. & Le, K. Glycyrrhizic Acid Reverses Neurological Deficits by Attenuating Rac1-STAT3 Signalling-Mediated Neuroinflammation in a Mouse Model of Neonatal Hypoxic-Ischemic Encephalopathy. Rev. Bras. Farmacogn. 33, 954–964 (2023). https://doi.org/10.1007/s43450-023-00402-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43450-023-00402-w