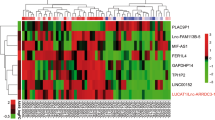
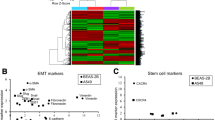
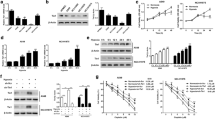
Abstract
Hypoxia is one of the hallmarks of the solid cancer microenvironment that dominates cancer progression and exacerbation. Under the oxygen-deprived condition, cancer resists and circumvents all interventions for permanence. Genomic and genetic instability of hypoxic cancer remains to be insightfully investigated and interpreted, as in lung adenocarcinoma. Herein, non-small cell lung cancer (NSCLC) cell line A549 was exposed to hypoxic shots parallel with running non-hypoxic (normoxic) A549 cells. Based on the isolated total RNA, gene transcriptomic profiling was identified using microarray and analysed via Ingenuity Pathway Analysis (IPA). As well, wound healing and cytotoxicity of doxorubicin were performed to assess hypoxic lung cancer cell response. Gene expression analysis revealed that TP53 (p53) is the most activated signaling, along with suppression of chromosomal signalings (DNA replication and repair). Besides, TP53 and its dependent target CDKN1A/p21 (cell cycle down-regulator) were identified as the topmost significant upstream transcriptional regulators, commanding a 37-gene targetable panel for cancer survival. In addition, hypoxic A549 cells were more chemo-resistant and higher motile. Notably, other emerging alterations were detected regarding efflux transporters (ABC-As and ABC-G1) that have been selectively up-regulated over the rest of under-transcribed (ABC) transporters subfamilies. Together, these involved findings suggest that the p53 signaling could be a potential survival mediator of hypoxic NSCLC cells (A549), which triggered the prominent cell cycle down-regulation to frustrate apoptotic response in hypoxic NSCLC. Coupled with the impairing of DNA replication and repair signalings that promoted molecular alterations for survival. Represented by the remarkable epithelial-mesenchymal transition of hypo-proliferating hypoxic A549 cells by which was described at the transcriptional level alongside phenotypic level. Consequently, hypoxic A549 cells showed higher resistance to doxorubicin targeting DNA replication, regardless of any transcriptionally down-regulated (ABC) transporters.








Similar content being viewed by others
Data availability
The datasets used or analyzed during the present research are available from the corresponding author on reasonable request.
References
AbuHammad, S., & Zihlif, M. (2013). Gene expression alterations in doxorubicin resistant MCF7 breast cancer cell line. Genomics, 101(4), 213–220. https://doi.org/10.1016/j.ygeno.2012.11.009
Alhawarat, F. M., Hammad, H. M., Hijjawi, M. S., Sharab, A. S., Abuarqoub, D. A., Shhab, M. A. A., & Zihlif, M. A. (2019). The effect of cycling hypoxia on MCF-7 cancer stem cells and the impact of their microenvironment on angiogenesis using human umbilical vein endothelial cells (HUVECs) as a model. PeerJ, 7, e5990. https://doi.org/10.7717/peerj.5990
Bayer, C., & Vaupel, P. (2012). Acute versus chronic hypoxia in tumors: Controversial data concerning time frames and biological consequences. Strahlentherapie Und Onkologie, 188(7), 616–627. https://doi.org/10.1007/s00066-012-0085-4
Bhaskara, V. K., Mohanam, I., Rao, J. S., & Mohanam, S. (2012). Intermittent hypoxia regulates stem-like characteristics and differentiation of neuroblastoma Cells. PLoS ONE, 7(2), e30905. https://doi.org/10.1371/journal.pone.0030905
Cairns, R. A., Kalliomaki, T., & Hill, R. P. (2001). Acute (cyclic) hypoxia enhances spontaneous metastasis of KHT murine tumors. Cancer Research, 61(24), 8903–8908.
Chan, C.Y.-K., Yuen, V.W.-H., & Wong, C.C.-L. (2019). Hypoxia and the metastatic niche. Advances in Experimental Medicine and Biology, 1136, 97–112. https://doi.org/10.1007/978-3-030-12734-3_7
Chen, C., Liang, Q.-Y., Chen, H.-K., Wu, P.-F., Feng, Z.-Y., Ma, X.-M., Wu, H.-R., & Zhou, G.-Q. (2018). DRAM1 regulates the migration and invasion of hepatoblastoma cells via autophagy-EMT pathway. Oncology Letters, 16(2), 2427–2433. https://doi.org/10.3892/ol.2018.8937
Chen, A., Sceneay, J., Gödde, N., Kinwel, T., Ham, S., Thompson, E. W., Humbert, P. O., & Möller, A. (2018). Intermittent hypoxia induces a metastatic phenotype in breast cancer. Oncogene, 37(31), 4214–4225. https://doi.org/10.1038/s41388-018-0259-3
Chenevier-Gobeaux, C., Simonneau, C., Lemarechal, H., Bonnefont-Rousselot, D., Poiraudeau, S., Rannou, F., Ekindjian, O. G., Anract, P., & Borderie, D. (2013). Effect of hypoxia/reoxygenation on the cytokine-induced production of nitric oxide and superoxide anion in cultured osteoarthritic synoviocytes. Osteoarthritis and Cartilage, 21(6), 874–881. https://doi.org/10.1016/j.joca.2013.03.010
Coppé, J.-P., Desprez, P.-Y., Krtolica, A., & Campisi, J. (2010). The senescence-associated secretory phenotype: The dark side of tumor suppression. Annual Review of Pathology: Mechanisms of Disease, 5(1), 99–118. https://doi.org/10.1146/annurev-pathol-121808-102144
Corn, P. G., & El-Deiry, W. S. (2007). Microarray analysis of p53-dependent gene expression in response to hypoxia and DNA damage. Cancer Biology & Therapy, 6(12), 1858–1866. https://doi.org/10.4161/cbt.6.12.5330
Di, G., Liu, Y., Lu, Y., Liu, J., Wu, C., & Duan, H.-F. (2014). IL-6 secreted from senescent mesenchymal stem cells promotes proliferation and migration of breast cancer cells. PLoS ONE, 9(11), e113572. https://doi.org/10.1371/journal.pone.0113572
Drosos, Y., Escobar, D., Chiang, M.-Y., Roys, K., Valentine, V., Valentine, M. B., Rehg, J. E., Sahai, V., Begley, L. A., Ye, J., Paul, L., McKinnon, P. J., & Sosa-Pineda, B. (2017). ATM-deficiency increases genomic instability and metastatic potential in a mouse model of pancreatic cancer. Scientific Reports, 7(1), 11144. https://doi.org/10.1038/s41598-017-11661-8
Du, Y., Zhou, Q., Yin, W., Zhou, L., Di, G., Shen, Z., Shao, Z., & Lu, J. (2011). The role of topoisomerase IIα in predicting sensitivity to anthracyclines in breast cancer patients: A meta-analysis of published literatures. Breast Cancer Research and Treatment, 129(3), 839–848. https://doi.org/10.1007/s10549-011-1694-9
Ferlay, J., Colombet, M., Soerjomataram, I., Parkin, D. M., Piñeros, M., Znaor, A., & Bray, F. (2021). Cancer statistics for the year 2020: An overview. International Journal of Cancer, 149(4), 778–789. https://doi.org/10.1002/ijc.33588
Greaves, W., Xiao, L., Sanchez-Espiridion, B., Kunkalla, K., Dave, K. S., Liang, C. S., Singh, R. R., Younes, A., Medeiros, L., & Vega, F. (2012). Detection of ABCC1 expression in classical Hodgkin lymphoma is associated with increased risk of treatment failure using standard chemotherapy protocols. Journal of Hematology & Oncology, 5(1), 47. https://doi.org/10.1186/1756-8722-5-47
Haronikova, L., Olivares-Illana, V., Wang, L., Karakostis, K., Chen, S., & Fåhraeus, R. (2019). The p53 mRNA: An integral part of the cellular stress response. Nucleic Acids Research, 47(7), 3257–3271. https://doi.org/10.1093/nar/gkz124
Harris, A. L. (2002). Hypoxia—A key regulatory factor in tumour growth. Nature Reviews Cancer, 2(1), 38–47. https://doi.org/10.1038/nrc704
Herbst, R. S., Heymach, J. V., & Lippman, S. M. (2008). Lung cancer. The New England Journal of Medicine, 359(13), 1367–1380. https://doi.org/10.1056/NEJMra0802714
Jänicke, R. U., Sohn, D., & Schulze-Osthoff, K. (2008). The dark side of a tumor suppressor: Anti-apoptotic p53. Cell Death and Differentiation, 15(6), 959–976. https://doi.org/10.1038/cdd.2008.33
Jarrar, Y., Zihlif, M., Al Bawab, A. Q., & Sharab, A. (2020). Effects of intermittent hypoxia on expression of glucose metabolism genes in MCF7 breast cancer cell line. Current Cancer Drug Targets, 20(3), 216–222. https://doi.org/10.2174/1568009619666191116095847
Jat, P., Perkins, N., & Mowla, S. (2013). Friend or foe: Emerging role of nuclear factor kappa-light-chain-enhancer of activated B cells in cell senescence. OncoTargets and Therapy. https://doi.org/10.2147/OTT.S36160
Jiang, H., Reinhardt, H. C., Bartkova, J., Tommiska, J., Blomqvist, C., Nevanlinna, H., Bartek, J., Yaffe, M. B., & Hemann, M. T. (2009). The combined status of ATM and p53 link tumor development with therapeutic response. Genes & Development, 23(16), 1895–1909. https://doi.org/10.1101/gad.1815309
Jin, M. H., & Oh, D.-Y. (2019). ATM in DNA repair in cancer. Pharmacology & Therapeutics, 203, 107391. https://doi.org/10.1016/j.pharmthera.2019.07.002
Liu, Y., Song, X., Wang, X., Wei, L., Liu, X., Yuan, S., & Lv, L. (2010). Effect of chronic intermittent hypoxia on biological behavior and hypoxia-associated gene expression in lung cancer cells. Journal of Cellular Biochemistry, 111(3), 554–563. https://doi.org/10.1002/jcb.22739
Mavrogonatou, E., Pratsinis, H., & Kletsas, D. (2020). The role of senescence in cancer development. Seminars in Cancer Biology, 62, 182–191. https://doi.org/10.1016/j.semcancer.2019.06.018
Mittal, V., El Rayes, T., Narula, N., McGraw, T. E., Altorki, N. K., & Barcellos-Hoff, M. H. (2016). The microenvironment of lung cancer and therapeutic implications. Advances in Experimental Medicine and Biology, 890, 75–110. https://doi.org/10.1007/978-3-319-24932-2_5
Muz, B., de la Puente, P., Azab, F., & Azab, A. K. (2015). The role of hypoxia in cancer progression, angiogenesis, metastasis, and resistance to therapy. Hypoxia (auckland, NZ), 3, 83–92. https://doi.org/10.2147/HP.S93413
Olbryt, M., Habryka, A., Student, S., Jarząb, M., Tyszkiewicz, T., & Lisowska, K. M. (2014). Global gene expression profiling in three tumor cell lines subjected to experimental cycling and chronic hypoxia. PLoS ONE, 9(8), e105104. https://doi.org/10.1371/journal.pone.0105104
Peinado, H., Olmeda, D., & Cano, A. (2007). Snail, Zeb and bHLH factors in tumour progression: An alliance against the epithelial phenotype? Nature Reviews. Cancer, 7(6), 415–428. https://doi.org/10.1038/nrc2131
Popper, H. H. (2016). Progression and metastasis of lung cancer. Cancer Metastasis Reviews, 35(1), 75–91. https://doi.org/10.1007/s10555-016-9618-0
Rodriguez-Canales, J., Parra-Cuentas, E., & Wistuba, I. I. (2016). Diagnosis and molecular classification of lung cancer. Cancer Treatment and Research, 170, 25–46. https://doi.org/10.1007/978-3-319-40389-2_2
Roh, Y.-G., Mun, M.-H., Jeong, M.-S., Kim, W.-T., Lee, S.-R., Chung, J.-W., Kim, S. I., Kim, T. N., Nam, J. K., & Leem, S.-H. (2018). Drug resistance of bladder cancer cells through activation of ABCG2 by FOXM1. BMB Reports, 51(2), 98–103. https://doi.org/10.5483/bmbrep.2018.51.2.222
Salem, A., Asselin, M.-C., Reymen, B., Jackson, A., Lambin, P., West, C. M. L., O’Connor, J. P. B., & Faivre-Finn, C. (2018). Targeting hypoxia to improve non-small cell lung cancer outcome. Journal of the National Cancer Institute. https://doi.org/10.1093/jnci/djx160
Sarin, N., Engel, F., Kalayda, G. V., Mannewitz, M., Cinatl, J., Rothweiler, F., Michaelis, M., Saafan, H., Ritter, C. A., Jaehde, U., & Frötschl, R. (2017). Cisplatin resistance in non-small cell lung cancer cells is associated with an abrogation of cisplatin-induced G2/M cell cycle arrest. PLoS ONE, 12(7), e0181081. https://doi.org/10.1371/journal.pone.0181081
Sermeus, A., & Michiels, C. (2011). Reciprocal influence of the p53 and the hypoxic pathways. Cell Death & Disease, 2(5), e164–e164. https://doi.org/10.1038/cddis.2011.48
Verduzco, D., Lloyd, M., Xu, L., Ibrahim-Hashim, A., Balagurunathan, Y., Gatenby, R. A., & Gillies, R. J. (2015). Intermittent hypoxia selects for genotypes and phenotypes that increase survival, invasion, and therapy resistance. PLoS ONE, 10(3), e0120958. https://doi.org/10.1371/journal.pone.0120958
Vogelstein, B., Lane, D., & Levine, A. J. (2000). Surfing the p53 network. Nature, 408(6810), 307–310. https://doi.org/10.1038/35042675
Wang, C., Chen, F., Liu, Y., Xu, Q., Guo, L., Zhang, X., Ruan, Y., Shi, Y., Shen, L., Li, M., Du, H., Sun, X., Ma, J., He, L., & Qin, S. (2018). Genetic association of drug response to erlotinib in chinese advanced non-small cell lung cancer patients. Frontiers in Pharmacology, 9, 360. https://doi.org/10.3389/fphar.2018.00360
Wei, Z., Guo, H., Qin, J., Lu, S., Liu, Q., Zhang, X., Zou, Y., Gong, Y., & Shao, C. (2019). Pan-senescence transcriptome analysis identified RRAD as a marker and negative regulator of cellular senescence. Free Radical Biology & Medicine, 130, 267–277. https://doi.org/10.1016/j.freeradbiomed.2018.10.457
Wu, F., Li, J., Jang, C., Wang, J., & Xiong, J. (2014). The role of Axl in drug resistance and epithelial-to-mesenchymal transition of non-small cell lung carcinoma. International Journal of Clinical and Experimental Pathology, 7(10), 6653–6661.
Yousaf, M., & Ali, M. (2020). Modulation of ABCG2 surface expression by Rab5 and Rab21 to overcome multidrug resistance in cancer cells. Xenobiotica, 50(8), 988–996. https://doi.org/10.1080/00498254.2020.1716107
Zhang, X., Qin, Z., & Wang, J. (2010). The role of p53 in cell metabolism. Acta Pharmacologica Sinica, 31(9), 1208–1212. https://doi.org/10.1038/aps.2010.151
Acknowledgements
This research was supported by a grant from the Deanship of Academic Research at The University of Jordan. All of this research was accomplished appreciatively in the Department of Pharmacology, School of Medicine at The University of Jordan.
Funding
This research was funded by the Deanship of Academic Research at The University of Jordan.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare there is no conflict of interest.
Ethical approval
Not applicable.
Consent for publication
All the authors have read the final version of the manuscript and consent to the publication.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Al-Najjar, L.M., Zihlif, M. & Jarrar, Y. Differential molecular alterations promoting non-small cell lung cancer under hypoxia. GENOME INSTAB. DIS. 3, 108–121 (2022). https://doi.org/10.1007/s42764-022-00062-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42764-022-00062-5