Abstract
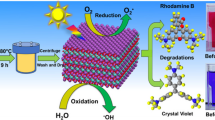
This study investigates the synthesis and photocatalytic properties of lithium niobate. X-ray diffraction (XRD) analysis reveals that a 48-h synthesis yields lithium niobate with a superior crystalline structure compared to 24-h and 12-h samples, evidenced by a smaller full width at half maximum (FWHM). Optimal Nb2O5/LiOH ratios of 3:5.7 result in a pure lithium niobate phase, while deviations lead to Nb2O5 or unidentified phases, corroborated by SEM observations of square lithium niobate structures at the ideal ratio and irregular forms at others. Under UV irradiation in the first hour, lithium niobate exhibits rapid degradation of methylene blue at 82.04%, twice as fast as TiO2, surpassing the efficiency of TiO2. In addition, lithium niobate significantly outperformed TiO2 in degrading methyl blue up to 67.14% under visible LED irradiation during the first hour, while TiO2 showed no degradation effect. This demonstrates its exceptional degradation characteristics and potential for enhanced photocatalytic applications.






Similar content being viewed by others
References
R. Mehrkhah, K. Goharshadi, E.K. Goharshadi, H.-S. Sajjadizadeh, ChemistrySelect (2023). https://doi.org/10.1002/slct.202204386
R. Mehrkhah, M.M. Ghafurian, H. Niazmand, E. Goharshadi, O. Mahian, Adv. Nanofluid Heat. Transf. (2022). https://doi.org/10.1016/B978-0-323-88656-7.00009-X
R. Mehrkhah, E.K. Goharshadi, E. Lichtfouse, H.S. Ahn, S. Wongwises, W. Yu, O. Mahian, Environ. Chem. Lett. 21, 285 (2023). https://doi.org/10.1007/s10311-022-01501-1
L. Wang, Wu. Jinsheng, Inorg. Chem. Commun. 129, 108619 (2021). https://doi.org/10.1016/j.inoche.2021.108619
S. Nahar, M. Zain, A.A.H. Kadhum, H.A. Hasan, M. Hasan, Materials 10(6), 629 (2017). https://doi.org/10.3390/ma10060629
J. Hong, K.-H. Cho, V. Presser, Su. Xiao, Curr. Opin. Green Sustain. Chem. 13, 100644 (2022). https://doi.org/10.1016/j.cogsc.2022.100644
A. Benzaouak, I. Ellouzi, F. Ouanji, N. Touach, M. Kacimi, M. Ziyad, M.E. Mahi, E.M. Lotfi, Colloids Surf A Physicochem Eng Asp 553, 586 (2018). https://doi.org/10.1016/j.colsurfa.2018.06.011
S. Wang, W. Zhang, F. Jia, H. Fu, T. Liu, X. Zhang, B. Liu, A. Núñez-Delgado, N. Han, J. Environ. Manag. 292, 1127 (2021). https://doi.org/10.1016/j.jenvman.2021.112763
S. Tawkaew, Y. Fujishiro, S. Yin, T. Sato, Colloids Surf A Physicochem Eng Asp 179, 139 (2001). https://doi.org/10.1016/S0927-7757(00)00649-X
H.O. Tugaoen, P. Herckes, K. Hristovski, P. Westerhoff, Appl. Catal. B Environ. 220, 597 (2018). https://doi.org/10.1016/j.apcatb.2017.08.078
S.G. Kumar, L.G. Devi, J. Phys. Chem. A 115, 13211 (2011). https://doi.org/10.1021/jp204364a
L. Liu, Y. Li, Aerosol Air Qual. Res. 14, 453 (2014). https://doi.org/10.4209/AAQR.2013.06.0186
B. Zielińska, E. Borowiak-Palen, R.J. Kalenzuk, J. Phys. Chem. Solids 69, 236 (2008). https://doi.org/10.1016/j.jpcs.2007.09.001
R.C. Miller, A. Savage, Appl. Phys. Lett. 9, 169 (1966). https://doi.org/10.1063/1.1754695
R.K. Nath, M.F.M. Zain, A.A.H. Kadhum, Catal. Rev. Sci. Eng. 56, 175 (2014). https://doi.org/10.1080/01614940.2013.872013
C.D. Fierro-Ruiz, O. Sánchez-Dena, E.M. Cabral-Larquier, J.T. Elizalde-Galindo, R. Farías, Crystals 8(3), 108 (2018). https://doi.org/10.3390/cryst8030108
P. Reichenbach, T. Kämpfe, A. Thiessen, A. Haußmann, T. Woike, L.M. Eng, Appl. Phys. Lett. 105, 122906 (2014). https://doi.org/10.1063/1.4896579
X.Y. Liu, K. Kitamura, K. Terabe, H. Hatano, N. Ohashi, Appl. Phys. Lett. (2007). https://doi.org/10.1063/1.2759472
X. Wang, W. Yan, Y. Zhang, L. Zhang, L. Shi, Y. Huang, M. Wu, X. Wang, H. Chen, J. Am. Ceram. Soc. 100, 739 (2017). https://doi.org/10.1111/jace.14571
X. Li, S. Wang, H. An, G. Dong, J. Feng, T. Wei, Y. Ren, J. Ma, Appl. Surf. Sci. 539, 148257 (2021). https://doi.org/10.1016/j.apsusc.2020.148257
B. Zielinska, Mater. Sci. 37, 911 (2014). https://doi.org/10.1007/s12034-014-0025-2
H.W. Fu, Y. Song, Y.Q. Wu, H.T. Huang, G.Z. Fan, J. Xu, Z.S. Li, Z.G. Zou, Appl. Phys. Lett. 112, 073901 (2018). https://doi.org/10.1063/1.5021377
X. Li, C. He, D. Dai, S. Zuo, X. Yan, C. Yao, C. Ni, Appl. Nanosci. 10, 3477 (2020). https://doi.org/10.1007/s13204-020-01443-6
K.P. Petrov, L. Goldberg, W.K. Burns, R.F. Curl, F.K. Tittle, Opt. Lett. 21, 86 (1996). https://doi.org/10.1364/OL.21.000086
D. Richter, Appl. Phys. B 67(3), 347 (1998). https://doi.org/10.1007/s003400050514
R.K. Nath, M.F.M. Zain, A.A.H. Kadhum, A.B.M.A. Kaish, Constr. Build. Mater. 54, 384 (2014). https://doi.org/10.1016/j.conbuildmat.2013.12.072
N. Touach, V.M. Ortiz-Martínez, M.J. Salar-García, A. Benzaouak, F. Hernández-Fernández, A.P. de Ríos, M.E. Mahi, E.M. Lotfi, Particuology 34, 147 (2017). https://doi.org/10.1016/j.partic.2017.02.006
R.K. Nath, M.F.M. Zain, A. A. H. Kadhum (2013). https://doi.org/10.1155/2013/686497
H. Xu, Y. Li, M. Ding, W. Chen, K. Wang, C. Lu, A.C.S. Sustain, Chem. Eng. 6, 7042 (2018). https://doi.org/10.1021/ACSSUSCHEMENG.8B00917
J. Zhao, N. Li, R. Yu, Z. Zhao, J. Nan, Chem. Eng. J. 349, 530 (2018). https://doi.org/10.1016/j.cej.2018.05.124
A. Salabat, F. Mirhoseini, F.H. Nouri, J. Iran. Chem. Soc. 20, 599 (2023). https://doi.org/10.1007/s13738-022-02693-7
Y. Rilda, D. Pernando, S. Arief, S. Syukri, R. Refinel, A. Agustien, H. Pardi, J. Iran. Chem. Soc. 19, 2023 (2022). https://doi.org/10.1007/s13738-021-02439-x
Z. Munawar, S. Ghazanfar, H.M. Asif, M.A. Khan, M. Sirajuddin, M. Tariq, J.H. Shirazi, A. Haider, J. Iran. Chem. Soc. 20, 2245 (2023). https://doi.org/10.1007/s13738-023-02824-8
G. Liu, S. You, M. Ma, H. Huang, N. Ren, Environ. Sci. Technol. 50, 11218 (2016). https://doi.org/10.1021/acs.est.6b03455
W. Xu, R. Wang, X. Zhen, W. Zhang, X. Chen, Z. Wang, Ferroelectrics 253, 153 (2001). https://doi.org/10.1080/00150190108008453
E. Cantelar, J.A. Sanz-GarcmHa, F. CussoH, J. Cryst, J. Cryst. Growth 205, 196 (1999). https://doi.org/10.1016/S0022-0248(99)00239-0
Yu. Ji, X. Liu, Mater. Lett. 61, 355 (2006). https://doi.org/10.1016/j.matlet.2006.04.087
A.R. Kamali, D.J. Fray, Ceram. Int. 40, 1835 (2014). https://doi.org/10.1016/j.ceramint.2013.07.085
M. Stock, S. Dunn, J. Phys. Chem. C 116(39), 20854 (2012). https://doi.org/10.1021/jp305217z
Y. Al-Douri, C.H. Voon, A. Bouhemadou, M. Ameri, Optik 172, 519 (2018). https://doi.org/10.1016/j.ijleo.2018.07.007
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yeh, M.Y., Chen, YJ., Chang, S.H. et al. Hydrothermal preparation of crystalline lithium niobate photocatalysts for effective degradation of dye-containing contaminated wastewater by ultraviolet to visible light irradiation. J IRAN CHEM SOC 21, 943–949 (2024). https://doi.org/10.1007/s13738-024-02966-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13738-024-02966-3