Abstract
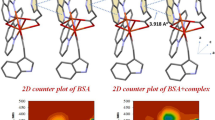
One-dimensional (1D) copper(II) glycinate coordination polymer {[Cu(gly)2(H2O)]n; (gly: glycine)} has been synthesized and characterized by CHN analysis, ESI–MS, FTIR and single-crystal X-ray diffraction techniques. Interaction of the complex with biomacromolecules {calf thymus DNA (CT-DNA) and bovine serum albumin (BSA)} has been investigated by electronic absorption and fluorescence spectroscopy methods. The experimental outcomes indicate that the complex binds to CT-DNA by means of a moderate intercalation mode. Furthermore, the fluorescence quenching mechanism between the complex and BSA is a static quenching process. The Stern–Volmer constants, binding constants, binding sites and the corresponding thermodynamic parameters (ΔG, ΔH, ΔS) of BSA + the complex systems were determined at different temperatures. The binding distance between the complex and BSA was calculated according to Förster non-radiation energy transfer theory (FRET). The effect of the complex on the conformation of BSA was also examined using synchronous, two-dimensional (2D) and three-dimensional (3D) fluorescence spectroscopy. Radical scavenging activity of the complex was determined in terms of IC50, using the DPPH and H2O2 method. In biomacromolecules interactions and radical activity studies, the complex was found to give good results.
Graphic abstract
A potent drug candidature of one-dimensional (1D) copper(II) glycinate coordination polymer—[Cu(gly)2(H2O)]n—(gly: glycine) has been synthesized and characterized by CHN analysis, FTIR, ESI–MS and single-crystal X-ray diffraction techniques. The complex has been tested for their in vitro biomacromolecular interactions by the spectroscopic methods. Furthermore, radical scavenging activities of the complex were also investigated.








Similar content being viewed by others
References
B. Joarder, A.K. Chaudhari, S.S. Nagarkar, B. Manna, S.K. Ghosh, Chem. Eur. J. 19, 11178 (2013)
C. Li, K. Deng, Z.Y. Tang, L. Jiang, J. Am. Chem. Soc. 132, 8202 (2010)
J.A. Gould, J.T.A. Jones, J. Bacsa, Y.Z. Khimyak, M.J. Rosseinsky, Chem. Commun. 46, 2793 (2010)
J.J. Zhang, T.L. Sheng, S.M. Hu, S.Q. Xia, G. Leibeling, F. Meyer, Z.Y. Fu, L. Chen, R.B. Fu, X.T. Wu, Chem. Eur. J. 10, 3963 (2004)
J.R. Soares, T.C.P. Dinis, A.P. Cunha, L.M. Almeida, Free Radic. Res. 26, 469 (1997)
A. Cetin, I.H. Geçibesler, J. Appl. Pharm. Sci. 5(6), 120 (2015)
B. Halliwell, J.M. Gutteridge, Methods Enzymol. 186, 1 (1990)
K. Jomova, D. Vondrakova, M. Lawson, M. Valko, Mol. Cell Biochem. 345, 91 (2010)
T.S. Kamatchi, N. Chitrapriya, S.K. Kim, F.R. Fronczek, K. Natarajan, Eur. J. Med. Chem. 59, 253 (2013)
Y.W. Li, B. Li, J. Theor. Biol. 318, 29 (2013)
L. Tabrizi, D.Q. Dao, T.A. Vu, RSC Adv. 9, 3320 (2019)
D. İnci, R. Aydin, Y. Zorlu, J. Coord. Chem. 69, 2677 (2016)
D. İnci, R. Aydın, H. Huriyet, Ö. Vatan, Y. Zorlu, B. Çoşut, N. Çinkılıç, Appl. Organomet. Chem. 33(1), e4652 (2019)
D.S. Raja, N.S. Bhuvanesh, K. Natarajan, Dalton Trans. 41, 4365 (2012)
A. Jahanban-Esfahlan, V. Panahi-Azar, S. Sajedi, Biopolymers 103, 638 (2015)
T.A. Wani, A.H. Bakheit, A.R.A. Al-Majed, M.A. Bhat, S. Zargar, Molecules 22, 1258 (2017)
G.F. Shen, T.T. Liu, Q. Wang, M. Jiang, J.H. Shi, J. Photochem. Photobiol. B Biol. 153, 380 (2015)
Z. Chi, R. Liu, Y. Teng, X. Fang, C. Gao, J. Agric. Food Chem. 58, 10262 (2010)
APEX2, version 2014.11-0, Bruker (2014) Bruker AXS Inc., Madison
SAINT, version 8.34A, Bruker (2013) Bruker AXS Inc., Madison
SADABS, version2014/5, Bruker (2014) Bruker AXS Inc., Madison
G.M. Sheldrick, Acta Crystallogr. A A71, 3 (2015)
G.M. Sheldrick, Acta Crystallogr. C C71, 3 (2015)
A.L. Spek, Acta Cryst. D D65, 148 (2009)
C.F. Macrae, I. Sovago, S.J. Cottrell, P.T.A. Galek, P. McCabe, E. Pidcock, M. Platings, G.P. Shields, J.S. Stevens, M. Towler, P.A. Wood, J. Appl. Crystallogr. 53, 226 (2020)
M.E. Reichmann, S.A. Rice, C.A. Thomas, P. Doty, J. Am. Chem. Soc. 76, 3047 (1954)
A.M. Pyle, J.P. Rehmann, R. Meshoyrer, C.V. Kumar, N.J. Turro, J.K. Barton, J. Am. Chem. Soc. 111, 3051 (1989)
K.D. Karlin, B.I. Cohen, J.C. Hayes, A. Farooq, J. Zubieta, Inorg. Chem. 26, 147 (1987)
M. Lee, A.L. Rhodes, M.D. Wyatt, S. Forrow, J.A. Hartley, Biochemistry 32, 4237 (1993)
J.R. Lakowicz, G. Weber, Biochemistry 12, 4161 (1973)
F. Mohammadi, H. Mansouri-Torshizi, J. Biomol. Struct. Dyn. 38(10), 3059 (2020)
N. Shahabadi, M. Hakimi, T. Morovati, S. Hadidi, K. Moeini, Luminescence 32, 43 (2017)
M.S. Ali, H.A. Al-Lohedan, Spectrochim. Acta Part A 203, 434 (2018)
J.R. Lakowicz, Principles of Fluorescence Spectroscopy (Plenum Press, New York, 1999).
F. Wang, W. Huang, Z.X. Dai, J. Mol. Struct. 875, 509 (2008)
S.M.T. Shaikh, J. Seetharamappa, P.B. Kandagal, S. Ashoka, J. Mol. Struct. 786, 46 (2006)
I. Gulcin, Toxicology 217(2–3), 213 (2006)
R.J. Ruch, S.J. Cheng, J.E. Klaunig, Carcinogenesis 10(6), 1003 (1989)
S.R. Liao, X.Y. Le, X.L. Feng, J. Coord. Chem. 61(6), 847 (2008)
S. Konar, K. Gagnon, A. Clearfıeld, C. Thompson, J. Hartle, C. Erıcson, C. Nelson, J. Coord. Chem. 63(19), 3335 (2010)
G.J. Chen, P.Q. Qiao, G.J. Xu, J.Y. Xu, J.L. Tian, W. Gu, X. Liu, S.P. Yan, J. Inorg. Biochem. 105, 119 (2011)
G. Psomas, J. Inorg. Biochem. 102, 1798 (2008)
A.K. Patra, T. Bhowmick, S. Roy, S. Ramakumar, A.R. Chakravarty, Inorg. Chem. 48, 2932 (2009)
D. İnci, R. Aydın, O. Vatan, T. Sevgi, D. Yılmaz, Y. Zorlu, Y. Yerli, B. Çoşut, E. Demirkan, N. Çinkılıç, J. Biol. Inorg. Chem. 22, 61 (2017)
D. İnci, R. Aydın, H. Huriyet, Y. Zorlu, N. Çinkılıç, Appl. Organomet. Chem. 32, e4309 (2018)
D. İnci, R. Aydın, Ö. Vatan, O. Şahin, N. Çinkılıç, New J. Chem. 43(12), 4681 (2019)
M.J. Waring, J. Mol. Biol. 13, 269 (1965)
S. Jain, T.A. Khan, Y.P. Patil, D. Pagariya, N. Kishore, S. Tapryal, A.D. Naik, S.G. Naik, J. Photochem. Photobiol. B 174, 35 (2017)
M.M. İbrahim, S.Y. Shaban, A.E.-M.M. Ramadan, M.A. Alruqi, G.A.M. Mersal, S.A. El-Shazly, S. Al-Juaid, J. Mol. Struct. 1198, 126911 (2019)
S. Bi, H. Zhang, C. Qiao, Y. Sun, C. Liu, Spectrochim. Acta A A69, 123 (2008)
M.A. Khajeh, G. Dehghan, S. Dastmalchi, M. Shaghaghi, M. Iranshahi, Spectrochim. Acta A 192, 384 (2018)
Md.F. Alam, S. Varshney, M.A. Khan, A.A. Laskar, H. Younus, Int. J. Biol. Macromol. 113, 300 (2018)
L. Ma, J. Wang, Y. Zhang, Toxins 9, 209 (2017)
P.S. Dorraji, F. Jalali, J. Braz. Chem. Soc. 24(6), 939 (2013)
E.S. Baker, M.T. Bowers, J. Am. Soc. Mass Spectrom. 18(7), 1188 (2007)
W. Harada, T. Nojima, A. Shibayama, H. Ueda, H. Shindo, M. Chikira, J. Inorg. Biochem. 64(4), 273 (1996)
Q. Gan, C.-L. Zhang, B.-F. Wang, Y.-H. Xiong, Y.-L. Fu, Z.-W. Mao, X.-Y. Le, RSC Adv. 6, 35952 (2016)
J. Zhang, Q. Yan, J. Liu, X. Lu, Y. Zhu, J. Wang, S. Wang, J. Lumin. 134, 747 (2013)
J. Kang, Y. Liu, M.X. Xie, S. Li, M. Jiang, Y.D. Wang, Biochim. Biophys. Acta 1674(2), 205 (2004)
T.A. Wani, H. AlRabiah, A.H. Bakheit, M.A. Kalam, S. Zargar, Chem. Cent. J. 11, 134 (2017)
W.R. Ware, J. Phys. Chem. 66(3), 455 (1962)
A. Garzón, I. Bravo, M.R. Carrión-Jiménez, Á. Rubio-Moraga, J. Albaladejo, Spectrochim. Acta Part A 150, 26 (2015)
R. Ferreira de Freitas, M. Schapira, Med. Chem. Commun. 8, 1970 (2017)
L.A. Sklar, B.S. Hudson, R.D. Simoni, Biochemistry 16, 5100 (1977)
X.X. Cheng, Y. Lui, B. Zhou, X.H. Xiao, Y. Liu, Spectrochim. Acta A 72, 922 (2009)
S. Prasanth, D.R. Rithesh Raj, T.V. Vineeshkumar, R.K. Thomas, C. Sudarsanakumar, RSC Adv. 6, 58288 (2016)
Z. Asadi, M. Golchin, V. Eigner, M. Dusek, Z. Amirghofran, Inorg. Chim. Acta 465, 50 (2017)
W. Widowati, A.P. Rani, R.A. Hamzah, S. Arumwardana, E. Afifah, H.S.W. Kusuma, D.D. Rihibiha, H. Nufus, A. Amalia, Nat. Prod. Sci. 23(3), 192 (2017)
M.M. Rahman, M.B. Islam, M. Biswas, A.H.M.K. Alam, BMC Res. Notes 8, 621 (2015)
A.E. Fazary, Y.-H. Ju, A.Q. Rajhi, A.S. Alshihri, M.Y. Alfaifi, M.A. Alshehri, K.A. Saleh, S.E.I. Elbehairi, K.F. Fawy, H.S.M. Abd-Rabboh, Open Chem. 14, 287 (2016)
M. Fiorani, R. de Sanctis, R. de Bellis, M. Dachá, Free Radic. Biol. Med. 32, 64 (2002)
D.A. Köse, E. Toprak, A. Kaşarcı, E. Avcı, G.A. Avcı, O. Şahin, O. Büyükgüngör, Synth. React. Inorg. Met. Org. Nano Met. Chem. 46(7), 1109 (2016)
N. Xu, G. Chen, H. Liu, Molecules 22(12), 2066 (2017)
Acknowledgements
This research was financed by Bursa Uludağ University Scientist Training and Development Project (Project Number BİYGP-2018/1).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
There are no conflicts to declare. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
İnci, D., Aydın, R. & Zorlu, Y. Biomacromolecular interactions and radical scavenging activities of one-dimensional (1D) copper(II) glycinate coordination polymer. J IRAN CHEM SOC 18, 3017–3030 (2021). https://doi.org/10.1007/s13738-021-02249-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13738-021-02249-1