Abstract
Purpose
Obesity is a risk factor and poor prognostic factor for pancreatic ductal adenocarcinoma (PDAC), but the underlying mechanisms remain unclear.
Methods
PDAC cells and obese visceral adipocytes (O-Ad) derived from mice and humans were used to analyze interactions between the two cell types, and human microvascular endothelial cells were used for angiogenesis assay. A xenograft mouse model with subcutaneously injected PDAC cells was used for animal studies. The relationship between visceral fat and prognosis was analyzed using resected tissues from PDAC patients with and without obesity.
Results
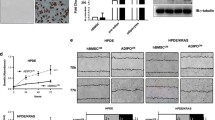
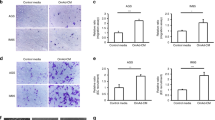
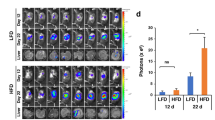
Conditioned media (CM) from O-Ad significantly increased PDAC cell growth and migration and angiogenic capacity in both human and mice cells, and blocking osteopontin (OPN) in O-Ad canceled O-Ad-induced effects in both mouse and human cells. In addition, O-Ad directly increased the migratory and tube-forming capacities of endothelial cells, while blocking OPN canceled these effects. O-Ad increased AKT phosphorylation and VEGFA expression in both PDAC and endothelial cells, and OPN inhibition in O-Ad canceled those O-Ad-induced effects. In the xenograft model, PDAC tumor volume was significantly increased in obese mice compared with lean mice, whereas blocking OPN significantly inhibited obesity-accelerated tumor growth. OPN expression in adipose tissues adjacent to human PDAC tumor was significantly higher in obese patients than in non-obese patients. In PDAC patients with obesity, high OPN expression in adipose tissues was significantly associated with poor prognosis.
Conclusion
Obese adipocytes trigger aggressive transformation in PDAC cells to induce PDAC progression and accelerate angiogenesis via OPN secretion.






Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
R.L. Siegel, K.D. Miller, H.E. Fuchs, A. Jemal, CA Cancer J Clin 71, 7–33 (2021). https://doi.org/10.3322/caac.21654
G.A. Manji, K.P. Olive, Y.M. Saenger, P. Oberstein, Clin Cancer Res 23, 1670–1678 (2017). https://doi.org/10.1158/1078-0432.CCR-16-2319
D.P. Ryan, T.S. Hong, N. Bardeesy, N Engl J Med 371, 1039–1049 (2014). https://doi.org/10.1056/NEJMra1404198
A.G. Renehan, M. Tyson, M. Egger, R.F. Heller, M. Zwahlen, Lancet 371, 569–578 (2008). https://doi.org/10.1016/S0140-6736(08)60269-X
D. Li, J.S. Morris, J. Liu, M.M. Hassan, R.S. Day, M.L. Bondy, J.L. Abbruzzese, JAMA 301, 2553–2562 (2009). https://doi.org/10.1001/jama.2009.886
E.E. Calle, C. Rodriguez, K. Walker-Thurmond, M.J. Thun, N Engl J Med 348, 1625–1638 (2003). https://doi.org/10.1056/NEJMoa021423
C. Yuan, Y. Bao, C. Wu, P. Kraft, S. Ogino, K. Ng, Z.R. Qian, D.A. Rubinson, M.J. Stampfer, E.L. Giovannucci, B.M. Wolpin, J Clin Oncol 31, 4229–4234 (2013). https://doi.org/10.1200/JCO.2013.51.7532
Y. Luo, Y. Yang, M. Liu, D. Wang, F. Wang, Y. Bi, J. Ji, S. Li, Y. Liu, R. Chen, H. Huang, X. Wang, A.K. Swidnicka-Siergiejko, T. Janowitz, S. Beyaz, G. Wang, S. Xu, A.B. Bialkowska, C.K. Luo, C.L. Pin, G. Liang, X. Lu, M. Wu, K.R. Shroyer, R.A. Wolff, W. Plunkett, B. Ji, Z. Li, E. Li, X. Li, V.W. Yang, C.D. Logsdon, J.L. Abbruzzese, W. Lu, Gastroenterology (2019). https://doi.org/10.1053/j.gastro.2019.07.030
K.M. Chung, J. Singh, L. Lawres, K.J. Dorans, C. Garcia, D.B. Burkhardt, R. Robbins, A. Bhutkar, R. Cardone, X. Zhao, A. Babic, S.A. Vayrynen, A. Dias Costa, J.A. Nowak, D.T. Chang, R.F. Dunne, A.F. Hezel, A.C. Koong, J.J. Wilhelm, M.D. Bellin, V. Nylander, A.L. Gloyn, M.I. McCarthy, R.G. Kibbey, S. Krishnaswamy, B.M. Wolpin, T. Jacks, C.S. Fuchs, M.D. Muzumdar, Cell 181, 832–847 e818 (2020). https://doi.org/10.1016/j.cell.2020.03.062
J. Incio, H. Liu, P. Suboj, S.M. Chin, I.X. Chen, M. Pinter, M.R. Ng, H.T. Nia, J. Grahovac, S. Kao, S. Babykutty, Y. Huang, K. Jung, N.N. Rahbari, X. Han, V.P. Chauhan, J.D. Martin, J. Kahn, P. Huang, V. Desphande, J. Michaelson, T.P. Michelakos, C.R. Ferrone, R. Soares, Y. Boucher, D. Fukumura, R.K. Jain, Cancer Discov 6, 852–869 (2016). https://doi.org/10.1158/2159-8290.CD-15-1177
E.D. Rosen, B.M. Spiegelman, Cell. 156, 20–44 (2014). https://doi.org/10.1016/j.cell.2013.12.012
C.L. Donohoe, S.L. Doyle, J.V. Reynolds, Diabetol Metab Syndr 3, 12 (2011). https://doi.org/10.1186/1758-5996-3-12
J. Folkman, N Engl J Med 285, 1182–1186 (1971). https://doi.org/10.1056/NEJM197111182852108
M.A. Rupnick, D. Panigrahy, C.Y. Zhang, S.M. Dallabrida, B.B. Lowell, R. Langer, M.J. Folkman, Proc Natl Acad Sci U S A 99, 10730–10735 (2002). https://doi.org/10.1073/pnas.162349799
M. Natsume, T. Shimura, H. Iwasaki, Y. Okuda, K. Hayashi, S. Takahashi, H. Kataoka, Br J Cancer 123, 459–470 (2020). https://doi.org/10.1038/s41416-020-0898-3
R. Roy, J. Yang, T. Shimura, L. Merritt, J. Alluin, E. Man, C. Daisy, R. Aldakhlallah, D. Dillon, S. Pories, L.A. Chodosh, M.A. Moses, Proc Natl Acad Sci U S A 119, e2204758119 (2022). https://doi.org/10.1073/pnas.2204758119
Fukusada, S., Shimura, T., Iwasaki, H., Okuda, Y., Katano, T. Nishigaki, R., Ozeki, T., Kitagawa, M., Nishie, H., Tanaka, M., Ozeki, K., Kubota, E., Tanida, S., Kataoka H.: Digestion, 1–8 (2021). https://doi.org/10.1159/000514812
J.P. Vandenbroucke, E. von Elm, D.G. Altman, P.C. Gotzsche, C.D. Mulrow, S.J. Pocock, C. Poole, J.J. Schlesselman, M. Egger, S. Initiative, Epidemiology 18, 805–835 (2007). https://doi.org/10.1097/EDE.0b013e3181577511
E.A. Eisenhauer, P. Therasse, J. Bogaerts, L.H. Schwartz, D. Sargent, R. Ford, J. Dancey, S. Arbuck, S. Gwyther, M. Mooney, L. Rubinstein, L. Shankar, L. Dodd, R. Kaplan, D. Lacombe, J. Verweij, Eur J Cancer 45, 228–247 (2009). https://doi.org/10.1016/j.ejca.2008.10.026
Y. Kanda, Bone Marrow Transplant 48, 452–458 (2013). https://doi.org/10.1038/bmt.2012.244
Z. Sun, D. Sun, Y. Feng, B. Zhang, P. Sun, B. Zhou, L. Du, Y. Wang, Z. Fan, J. Yang, Y. Li, S. Hu, H. Zhan, Mol. Ther. Nucleic Acids 26, 253–268 (2021). https://doi.org/10.1016/j.omtn.2021.06.001
M. Takehara, Y. Sato, T. Kimura, K. Noda, H. Miyamoto, Y. Fujino, J. Miyoshi, F. Nakamura, H. Wada, Y. Bando, T. Ikemoto, M. Shimada, N. Muguruma, T. Takayama, Cancer Sci 111, 2883–2894 (2020). https://doi.org/10.1111/cas.14527
G. Chakraborty, S. Jain, G.C. Kundu, Cancer Res 68, 152–161 (2008). https://doi.org/10.1158/0008-5472.CAN-07-2126
R. Wei, J.P.C. Wong, P. Lyu, X. Xi, O. Tong, S.D. Zhang, H.F. Yuen, S. Shirasawa, H.F. Kwok, J. Cell. Mol. Med. 22, 4097–4105 (2018). https://doi.org/10.1111/jcmm.13686
R. Urtasun, A. Lopategi, J. George, T.M. Leung, Y. Lu, X. Wang, X. Ge, M.I. Fiel, N. Nieto, Hepatology 55, 594–608 (2012). https://doi.org/10.1002/hep.24701
F.W. Kiefer, M. Zeyda, J. Todoric, J. Huber, R. Geyeregger, T. Weichhart, O. Aszmann, B. Ludvik, G.R. Silberhumer, G. Prager, T.M. Stulnig, Endocrinology 149, 1350–1357 (2008). https://doi.org/10.1210/en.2007-1312
J. Gomez-Ambrosi, V. Catalan, B. Ramirez, A. Rodriguez, I. Colina, C. Silva, F. Rotellar, C. Mugueta, M.J. Gil, J.A. Cienfuegos, J. Salvador, G. Fruhbeck, J Clin Endocrinol Metab 92, 3719–3727 (2007). https://doi.org/10.1210/jc.2007-0349
J. Huang, A. Duran, M. Reina-Campos, T. Valencia, E.A. Castilla, T.D. Muller, M.H. Tschop, J. Moscat, M.T. Diaz-Meco, Cancer Cell 33, 770–784 e776 (2018). https://doi.org/10.1016/j.ccell.2018.03.001
R. Raja, S. Kale, D. Thorat, G. Soundararajan, K. Lohite, A. Mane, S. Karnik, G.C. Kundu, Oncogene. 33, 2053–2064 (2014). https://doi.org/10.1038/onc.2013.171
A. Gupta, C.Q. Zhou, M.A. Chellaiah, Cancers (Basel) 5, 617–638 (2013). https://doi.org/10.3390/cancers5020617
J. Dai, L. Peng, K. Fan, H. Wang, R. Wei, G. Ji, J. Cai, B. Lu, B. Li, D. Zhang, Y. Kang, M. Tan, W. Qian, Y. Guo, Oncogene 28, 3412–3422 (2009). https://doi.org/10.1038/onc.2009.189
A. Kolb, J. Kleeff, A. Guweidhi, I. Esposito, N.A. Giese, H. Adwan, T. Giese, M.W. Buchler, M.R. Berger, H. Friess, Cancer Biol Ther 4, 740–746 (2005). https://doi.org/10.4161/cbt.4.7.1821
J. Cao, J. Li, L. Sun, T. Qin, Y. Xiao, K. Chen, W. Qian, W. Duan, J. Lei, J. Ma, Q. Ma, L. Han, Mol Oncol 13, 228–245 (2019). https://doi.org/10.1002/1878-0261.12399
Acknowledgements
We thank Mao Morimoto (Department of Gastroenterology and Metabolism, Nagoya City University Graduate School of Medical Sciences) for technical assistance.
Funding
This study was supported by Grant-in-Aid for Young Scientists in KAKENHI of Japan Society for the Promotion of Science (#. 19K17407) (to RN).
Author information
Authors and Affiliations
Contributions
Takaya Shimura designed the study conceptualization and protocol. Shigeki Fukusada, Makoto Natsume, Yusuke Okuda, Hiroyasu Iwasaki and Naomi Sugimura conducted experiments. Shigeki Fukusada, Ruriko Nishigaki, Mika Kitagawa, Takahito Katano, Hirotada Nishie, Mamoru Tanaka, Keiji Ozeki and Eiji Kubota analyzed the data, and Shigeki Fukusada and Takaya Shimura interpreted the data. Shigeki Fukusada drafted the manuscript and Takaya Shimura revised that. Makoto Natsume and Kazuki Hayashi contributed to extracting and analyzing the human data. Hiromi Kataoka supervised the conduct of this study. All authors read the manuscript, agree with the content, and approved the final version for submission.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
The current study complied with the STROBE statement. The study protocol was approved by the institutional review board of Nagoya City University Hospital and conducted in accordance with the ethical guidelines in the 1975 Declaration of Helsinki (7th revision, 2013) (permit number: 60-20-006). Animal experiments were performed with the approval of the Nagoya City University Center for Experimental Animal Science. (permit number: PE-230329002)
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Fukusada, S., Shimura, T., Natsume, M. et al. Osteopontin secreted from obese adipocytes enhances angiogenesis and promotes progression of pancreatic ductal adenocarcinoma in obesity. Cell Oncol. 47, 229–244 (2024). https://doi.org/10.1007/s13402-023-00865-y
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13402-023-00865-y