Abstract
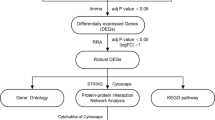
Atherosclerosis is a chronic inflammatory disease that affects arterial walls and is a leading cause of cardiovascular disease. Gene co-expression modules can provide insight into the molecular mechanisms underlying atherosclerosis progression. In this study, gene co-expression network analysis (WGCNA) was done to identify gene co-expression modules associated with atherosclerosis progression. Before conducting WGCNA, preprocessing and soft power selection were performed on the GSE28829, GSE100927, GSE43292, GSE10334, and GSE16134 datasets (https://www.ncbi.nlm.nih.gov/geo/query/acc.cgi). Co-expression modules were identified using dynamic tree cuts, and their correlations and trait associations were visualized. Enrichment analysis was performed on the blue and magenta modules to identify biological processes (BP) and pathways related to atherosclerosis. The CIBERSORT algorithm was used to predict immune cell infiltration in early and advanced atherosclerotic plaques. We identified 12 co-expression modules, in which blue and magenta were most highly correlated with atherosclerosis progression. The blue module was enriched for inflammation- and immune-related BP and pathways, including phagosome, lysosome, osteoclast differentiation, chemokine signaling pathway, platelet activation, NF-kappa B signaling pathway, Fc gamma R-mediated phagocytosis, lipid and atherosclerosis, autophagy, and apoptosis. The magenta module was significantly enriched for vascular permeability regulation, positive and negative regulation of epithelial to mesenchymal transition, and lamellipodium. Additionally, the CIBERSORT algorithm predicted less abundance of T regulatory cells and monocytes in advanced compared to early atherosclerotic plaques. The enrichment analysis of BP, cellular components, molecular functions, and atherosclerosis-related pathways in the blue and magenta modules showed that inflammation and immune response played a key role in the progression of atherosclerosis. Our study provides insights into the molecular mechanisms underlying atherosclerosis progression and identifies potential therapeutic targets for the treatment of atherosclerosis. The identification of immune cell subtypes associated with atherosclerosis could lead to the development of immunomodulatory therapies to prevent or treat atherosclerosis.




Similar content being viewed by others
Data Availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Chen B, Khodadoust MS, Liu CL, Newman AM, Alizadeh AA (2018) Profiling tumor infiltrating immune cells with CIBERSORT. Methods Mol Biol 1711:243–259. https://doi.org/10.1007/978-1-4939-7493-1_12
Chen M, Chen S, Yang D, Zhou J, Liu B, Chen Y et al (2021) Weighted gene co-expression network analysis identifies crucial genes mediating progression of carotid plaque. Front Physiol 12:601952. https://doi.org/10.3389/fphys.2021.601952
Feng S, Xu Y, Dai Z, Yin H, Zhang K, Shen Y (2022) Integrative analysis from multicenter studies identifies a WGCNA-derived cancer-associated fibroblast signature for ovarian cancer. Front Immunol 13:951582. https://doi.org/10.3389/fimmu.2022.951582
Fruchart JC, Santos RD, Aguilar-Salinas C, Aikawa M, Al Rasadi K, Amarenco P et al (2019) The selective peroxisome proliferator-activated receptor alpha modulator (SPPARMalpha) paradigm: conceptual framework and therapeutic potential: a consensus statement from the International Atherosclerosis Society (IAS) and the Residual Risk Reduction Initiative (R3i) Foundation. Cardiovasc Diabetol 18(1):71. https://doi.org/10.1186/s12933-019-0864-7
Gimbrone MA Jr, García-Cardeña G (2016) Endothelial cell dysfunction and the pathobiology of atherosclerosis. Circ Res 118(4):620–636. https://doi.org/10.1161/CIRCRESAHA.115.306301
Herrington W, Lacey B, Sherliker P, Armitage J, Lewington S (2016) Epidemiology of atherosclerosis and the potential to reduce the global burden of atherothrombotic disease. Circ Res 118(4):535–546. https://doi.org/10.1161/CIRCRESAHA.115.307611
Kobiyama K, Ley K (2018) Atherosclerosis. Circ Res 123(10):1118–1120. https://doi.org/10.1161/CIRCRESAHA.118.313816
Langfelder P, Horvath S (2008) WGCNA: an R package for weighted correlation network analysis. BMC Bioinformatics 9:559. https://doi.org/10.1186/1471-2105-9-559
Liang W, Sun F, Zhao Y, Shan L, Lou H (2020) Identification of susceptibility modules and genes for cardiovascular disease in diabetic patients using WGCNA analysis. J Diabetes Res 2020:4178639. https://doi.org/10.1155/2020/4178639
Libby P, Buring JE, Badimon L, Hansson GK, Deanfield J, Bittencourt MS et al (2019) Atherosclerosis. Nat Rev Dis Primers 5(1):56. https://doi.org/10.1038/s41572-019-0106-z
Ridker PM, Everett BM, Thuren T, MacFadyen JG, Chang WH, Ballantyne C et al (2017) Antiinflammatory therapy with canakinumab for atherosclerotic disease. N Engl J Med 377(12):1119–1131. https://doi.org/10.1056/NEJMoa1707914
Tardif JC, Kouz S, Waters DD, Bertrand OF, Diaz R, Maggioni AP et al (2019) Efficacy and safety of low-dose colchicine after myocardial infarction. N Engl J Med 381(26):2497–2505. https://doi.org/10.1056/NEJMoa1912388
Winkels H, Ehinger E, Vassallo M, Buscher K, Dinh HQ, Kobiyama K et al (2018) Atlas of the immune cell repertoire in mouse atherosclerosis defined by single-cell RNA-sequencing and mass cytometry. Circ Res 122(12):1675–1688. https://doi.org/10.1161/CIRCRESAHA.117.312513
Wolf D, Ley K (2019) Immunity and inflammation in atherosclerosis. Circ Res 124(2):315–327. https://doi.org/10.1161/CIRCRESAHA.118.313591
Wu T, Hu E, Xu S, Chen M, Guo P, Dai Z et al (2021) clusterProfiler 4.0: a universal enrichment tool for interpreting omics data. Innovation (Camb) 2(3):100141. https://doi.org/10.1016/j.xinn.2021.100141
Zhang T, Liu N, Wei W, Zhang Z, Li H (2021) Integrated analysis of weighted gene coexpression network analysis identifying six genes as novel biomarkers for Alzheimer’s disease. Oxid Med Cell Longev 2021:9918498. https://doi.org/10.1155/2021/9918498
Zhang X, He D, Xiang Y, Wang C, Liang B, Li B et al (2022) DYSF promotes monocyte activation in atherosclerotic cardiovascular disease as a DNA methylation-driven gene. Transl Res 247:19–38. https://doi.org/10.1016/j.trsl.2022.04.001
Zhao W, Langfelder P, Fuller T, Dong J, Li A, Hovarth S (2010) Weighted gene coexpression network analysis: state of the art. J Biopharm Stat 20(2):281–300. https://doi.org/10.1080/10543400903572753
Zhu F, Zuo L, Hu R, Wang J, Yang Z, Qi X et al (2021) A ten-genes-based diagnostic signature for atherosclerosis. BMC Cardiovasc Disord 21(1):513. https://doi.org/10.1186/s12872-021-02323-9
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by: Ewa Ziętkiewicz
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Gu, J., Yang, W., Lin, S. et al. Identification of co-expressed genes and immune infiltration features related to the progression of atherosclerosis. J Appl Genetics 65, 331–339 (2024). https://doi.org/10.1007/s13353-023-00801-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13353-023-00801-8