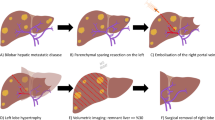
Abstract
The management of patients with locally advanced mid/low rectal cancer with resectable liver metastases is complex because of the need to combine the optimal treatment of both tumors. This study aims to review the available treatment strategies and compare their outcome, focusing on radiotherapy (RT) and liver-first approach (LFA). A systematic review was performed in PubMed, Embase, and web sources including articles published between 2000 and 02/2023 and reporting mid-/long-term outcomes. Overall, twenty studies were included (n = 1837 patients). Three- and 5-year overall survival (OS) rates were 51–88% and 36–59%. Although several strategies were reported, most patients received RT (1448/1837, 79%; > 85% neoadjuvant). RT reduced the pelvic recurrence risk (5.8 vs. 13.5%, P = 0.005) but did not impact OS. Six studies analyzed LFA (n = 307 patients). LFA had a completion rate similar to the rectum-first approach (RFA, 81% vs. 79%) but the interval strategy—an LFA variant with liver surgery in the interval between radiotherapy and rectal surgery—had a better completion rate than standard LFA (liver surgery/radiotherapy/rectal surgery, 92% vs. 75%, P = 0.011) and RFA (79%, P = 0.048). Across all series, LFA achieved the best survival rates, and in one paper it led to a survival advantage in patients with multiple metastases. In conclusion, different strategies can be adopted, but RT should be included to decrease the pelvic recurrence risk. LFA should be considered, especially in patients with high hepatic tumor burden, and RT before liver surgery (interval strategy) could maximize its completion rate.






Similar content being viewed by others
Data availability
The datasets generated during the current study are available from the corresponding author on reasonable request.
Code availability
Not applicable.
References
Xi Y, Xu P (2021) Global colorectal cancer burden in 2020 and projections to 2040. Transl Oncol 14(10):101174. https://doi.org/10.1016/j.tranon.2021.101174
Arnold M, Sierra MS, Laversanne M et al (2017) Global patterns and trends in colorectal cancer incidence and mortality. Gut 66:683–691. https://doi.org/10.1136/gutjnl-2015-310912
Adam R, de Gramont A, Figueras J et al (2015) Managing synchronous liver metastases from colorectal cancer: a multidisciplinary international consensus. Cancer Treat Rev 41(9):729–741. https://doi.org/10.1016/j.ctrv.2015.06.006
Engstrand J, Nilsson H, Strömberg C et al (2018) Colorectal cancer liver metastases—a population-based study on incidence, management and survival. BMC Cancer. https://doi.org/10.1186/s12885-017-3925-x
Kopetz S, Chang GJ, Overman MJ et al (2009) Improved survival in metastatic colorectal cancer is associated with adoption of hepatic resection and improved chemotherapy. J Clin Oncol 27:3677–3683. https://doi.org/10.1200/JCO.2008.20.5278
Creasy JM, Sadot E, Koerkamp BG et al (2018) Actual 10-year survival after hepatic resection of colorectal liver metastases: what factors preclude cure? Surgery (United States) 163:1238–1244. https://doi.org/10.1016/j.surg.2018.01.004
Viganò L, Ferrero A, Lo Tesoriere R, Capussotti L (2008) Liver surgery for colorectal metastases: results after 10 years of follow-up. Long-term survivors, late recurrences, and prognostic role of morbidity. Ann Surg Oncol 15:2458–2464. https://doi.org/10.1245/s10434-008-9935-9
Petrelli F, Trevisan F, Tomasello G et al (2022) Different neoadjuvant therapies for locally advanced rectal cancer: a systematic review and network meta-analysis. Crit Rev Oncol Hematol 180:103853. https://doi.org/10.1016/j.critrevonc.2022.103853
Nordlinger B, Sorbye H, Glimelius B et al (2008) Perioperative chemotherapy with FOLFOX4 and surgery versus surgery alone for resectable liver metastases from colorectal cancer (EORTC Intergroup trial 40983): a randomised controlled trial. Lancet 371(9617):1007–1016
Okuno M, Hatano E, Nishino H et al (2017) Does response rate of chemotherapy with molecular target agents correlate with the conversion rate and survival in patients with unresectable colorectal liver metastases?: a systematic review. Eur J Surg Oncol 43(6):1003–1012. https://doi.org/10.1016/j.ejso.2016.08.019
Vigano L, Terrone A, Costa G et al (2022) Effect of chemotherapy on tumour-vessel relationship in colorectal liver metastases. Br J Surg 109(5):401–404. https://doi.org/10.1093/bjs/znac010
Viganò L, Jayakody Arachchige VS, Fiz F et al (2022) Is precision medicine for colorectal liver metastases still a utopia? New perspectives by modern biomarkers, radiomics, and artificial intelligence. World J Gastroenterol 28(6):608–623. https://doi.org/10.3748/wjg.v28.i6.608
Rocca A, Cipriani F, Belli G et al (2021) The Italian Consensus on minimally invasive simultaneous resections for synchronous liver metastasis and primary colorectal cancer: a Delphi methodology. Updates Surg 73:1247–1265. https://doi.org/10.1007/s13304-021-01100-9
Mentha G, Majno PE, Andres A et al (2006) Neoadjuvant chemotherapy and resection of advanced synchronous liver metastases before treatment of the colorectal primary. Br J Surg 93(7):872–878. https://doi.org/10.1002/bjs.5346
Valdimarsson VT, Syk I, Lindell G et al (2018) Outcomes of liver-first strategy and classical strategy for synchronous colorectal liver metastases in Sweden. HPB 20:441–447. https://doi.org/10.1016/j.hpb.2017.11.004
Vallance AE, van der Meulen J, Kuryba A et al (2018) The timing of liver resection in patients with colorectal cancer and synchronous liver metastases: a population-based study of current practice and survival. Colorectal Dis 20:486–495. https://doi.org/10.1111/codi.14019
Carbone F, Chee Y, Rasheed S et al (2022) Which surgical strategy for colorectal cancer with synchronous hepatic metastases provides the best outcome? A comparison between primary first, liver first and simultaneous approach. Updates Surg 74(2):451–465. https://doi.org/10.1007/s13304-021-01234-w
Erlandsson J, Holm T, Pettersson D et al (2017) Optimal fractionation of preoperative radiotherapy and timing to surgery for rectal cancer (Stockholm III): a multicentre, randomised, non-blinded, phase 3, non-inferiority trial. Lancet Oncol 18:336–346. https://doi.org/10.1016/S1470-2045(17)30086-4
Agas RAF, Co LBA, Jacinto JCKM et al (2018) Neoadjuvant radiotherapy versus no radiotherapy for stage IV rectal cancer: a systematic review and meta-analysis. J Gastrointest Cancer 49:389–401. https://doi.org/10.1007/s12029-018-0141-0
Bregni G, Akin Telli T, Camera S et al (2020) Grey areas and evidence gaps in the management of rectal cancer as revealed by comparing recommendations from clinical guidelines. Cancer Treat Rev 82:101930. https://doi.org/10.1016/j.ctrv.2019.101930
Page MJ, McKenzie JE, Bossuyt PM et al (2021) The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. The BMJ. https://doi.org/10.1136/bmj.n71
Viganò L, Karoui M, Ferrero A et al (2011) Locally advanced mid/low rectal cancer with synchronous liver metastases. World J Surg 35:2788–2795. https://doi.org/10.1007/s00268-011-1272-7
Kim JW, Kim YB, Kim NK et al (2010) The role of adjuvant pelvic radiotherapy in rectal cancer with synchronous liver metastasis: a retrospective study. Radiation Oncol 5:75
Shin SJ, Yoon HI, Kim NK et al (2011) Upfront systemic chemotherapy and preoperative short-course radiotherapy with delayed surgery for locally advanced rectal cancer with distant metastases. Radiation Oncol. https://doi.org/10.1186/1748-717X-6-99
An HJ, Yu CS, Yun SC et al (2012) Adjuvant chemotherapy with or without pelvic radiotherapy after simultaneous surgical resection of rectal cancer with liver metastases: analysis of prognosis and patterns of recurrence. Int J Radiat Oncol Biol Phys 84:73–80. https://doi.org/10.1016/j.ijrobp.2011.10.070
Manceau G, Brouquet A, Bachet JB et al (2013) Response of liver metastases to preoperative radiochemotherapy in patients with locally advanced rectal cancer and resectable synchronous liver metastases. Surgery (United States) 154:528–535. https://doi.org/10.1016/j.surg.2013.02.010
Jung M, Holmqvist A, Sun XF, Albertsson M (2014) A clinical study of metastasized rectal cancer treatment: assessing a multimodal approach. Med Oncol. https://doi.org/10.1007/s12032-014-0839-1
Sabbagh C, Cosse C, Ravololoniaina T et al (2015) Oncological strategies for middle and low rectal cancer with synchronous liver metastases. Int J Surg 23:186–193. https://doi.org/10.1016/j.ijsu.2015.08.034
Cho H, Kim JE, Kim KP et al (2016) Phase II study of preoperative capecitabine and oxaliplatin-based intensified chemoradiotherapy with or without induction chemotherapy in patients with locally advanced rectal cancer and synchronous liver-limited resectable metastases. Am JClin Oncol 39:623–629. https://doi.org/10.1097/COC.0000000000000315
Labori KJ, Guren MG, Brudvik KW et al (2017) Resection of synchronous liver metastases between radiotherapy and definitive surgery for locally advanced rectal cancer: short-term surgical outcomes, overall survival and recurrence-free survival. Colorectal Dis 19:731–738. https://doi.org/10.1111/codi.13622
D’Hondt M, Lucidi V, Vermeiren K et al (2017) The interval approach: an adaptation of the liver-first approach to treat synchronous liver metastases from rectal cancer. World J Surg Oncol. https://doi.org/10.1186/s12957-017-1123-6
Fossum CC, Alabbad JY, Romak LB et al (2017) The role of neoadjuvant radiotherapy for locally-advanced rectal cancer with resectable synchronous metastasis. J Gastrointest Oncol 8:650–658
Bird T, Michael M, Bressel M et al (2017) FOLFOX and intensified split-course chemoradiation as initial treatment for rectal cancer with synchronous metastases. Acta Oncol (Madr) 56:646–652. https://doi.org/10.1080/0284186X.2017.1296584
Salvador-Rosés H, López-Ben S, Casellas-Robert M et al (2018) Oncological strategies for locally advanced rectal cancer with synchronous liver metastases, interval strategy versus rectum first strategy: a comparison of short-term outcomes. Clin Transl Oncol 20:1018–1025. https://doi.org/10.1007/s12094-017-1818-8
Kelly ME, Aalbers AGJ, Abdul Aziz N et al (2020) Simultaneous pelvic exenteration and liver resection for primary rectal cancer with synchronous liver metastases: results from the PelvEx Collaborative. Colorectal Dis 22:1258–1262. https://doi.org/10.1111/codi.15064
Conci S, Ruzzenente A, Pedrazzani C et al (2021) Simultaneous approach for patients with synchronous colon and rectal liver metastases: Impact of site of primary on postoperative and oncological outcomes. Eur J Surg Oncol 47:842–849. https://doi.org/10.1016/j.ejso.2020.09.015
Bae HW, Kim HS, Yang SY et al (2021) Upfront chemotherapy and short-course radiotherapy with delayed surgery for locally advanced rectal cancer with synchronous liver metastases. Eur J Surg Oncol 47:2814–2820. https://doi.org/10.1016/j.ejso.2021.05.018
Russolillo N, Gentile V, Ratti F et al (2022) Incidence and predictors of textbook outcome after simultaneous liver and rectal surgeries for Stage IV rectal cancer. Colorectal Dis 24:50–58. https://doi.org/10.1111/codi.15912
Giuliante F, Viganò L, De Rose AM et al (2021) Liver-first approach for synchronous colorectal metastases: analysis of 7360 patients from the livermetsurvey registry. Ann Surg Oncol. https://doi.org/10.1245/s10434
Chen J, Chen H, Zhao F et al (2022) Survival outcomes in unresectable metastatic rectal cancer patients after both primary site resection and chemoradiotherapy: a SEER-based observational study. Transl Cancer Res 11:171–180. https://doi.org/10.21037/tcr-21-1399
Van Rees JM, Krul MF, Kok NFM et al (2023) Treatment of locally advanced rectal cancer and synchronous liver metastases: multicentre comparison of two treatment strategies. Br J Surg 110:1049–1052. https://doi.org/10.1093/bjs/znad013
Colletti G, Ciniselli CM, Sorrentino L et al (2023) Multimodal treatment of rectal cancer with resectable synchronous liver metastases: a systematic review. Dig Liver Dis. https://doi.org/10.1016/j.dld.2023.05.023
Johnson GGRJ, Park J, Helewa RM et al (2023) Total neoadjuvant therapy for rectal cancer: a guide for surgeons. Can J Surg 66(2):E196–E220. https://doi.org/10.1503/cjs.005822
Yeo HL, Paty PB (2014) Management of recurrent rectal cancer: Practical insights in planning and surgical intervention. J Surg Oncol 109:47–52. https://doi.org/10.1002/jso.23457
Kazi M, Sukumar V, Desouza A, Saklani A (2021) State-of-the-art surgery for recurrent and locally advanced rectal cancers. Langenbecks Arch Surg 406:1763–1774. https://doi.org/10.1007/s00423-021-02285-8
Andres A, Toso C, Adam R et al (2012) A survival analysis of the liver-first reversed management of advanced simultaneous colorectal liver metastases. Ann Surg 256:772–779. https://doi.org/10.1097/SLA.0b013e3182734423
Sturesson C, Valdimarsson VT, Blomstrand E et al (2017) Liver-first strategy for synchronous colorectal liver metastases—an intention-to-treat analysis. HPB (Oxford) 19(1):52–58. https://doi.org/10.1016/j.hpb.2016.10.005
Welsh FKS, Chandrakumaran K, John TG et al (2016) Propensity score-matched outcomes analysis of the liver-first approach for synchronous colorectal liver metastases. Br J Surg 103:600–606. https://doi.org/10.1002/bjs.10099
Vallance AE, vanderMeulen J, Kuryba A et al (2017) Impact of hepatobiliary service centralization on treatment and outcomes in patients with colorectal cancer and liver metastases. Br J Surg 104:918–925. https://doi.org/10.1002/bjs.10501
Viganò L, Langella S, Ferrero A et al (2013) Colorectal cancer with synchronous resectable liver metastases: monocentric management in a hepatobiliary referral center improves survival outcomes. Ann Surg Oncol 20:938–945. https://doi.org/10.1245/s10434-012-2628-4
Acknowledgements
The authors acknowledge Prof. F. Calise and Prof. P. Miccoli, coordinators of the Medical Writing course “Light into a Scientific Paper” of the SIC (Italian Society of Surgery) which was held in Rome in October 2022. They had the merit to create this cooperative group of researchers and to address and mentor their activity.
Standards of reporting
The systematic review was carried out according to the PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) guidelines.
Funding
The present study was funded by the AIRC (Italian Association for Cancer Research) Grant #2019− 23822 (PI: Prof. Luca Viganò).The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
Author information
Authors and Affiliations
Contributions
Conceptualization: R Tutino, A Bonomi, CC Zingaretti, M Paterno, I Pezzoli; Methodology: Vigano’ L; Data collection: R Tutino, A Bonomi, CC Zingaretti, L Risi, E M Ragaini, M Paterno, I Pezzoli; Formal analysis and investigation: All authors; Writing—original draft: R Tutino, A Bonomi, CC Zingaretti, L Risi, M Paterno, I Pezzoli; Writing: review and editing: E M Ragaini, L Viganò; Final approval: all authors.
Corresponding author
Ethics declarations
Conflict of interest
Viganò L: received speaker’s honoraria from Johnson & Johnson. The remaining authors: reported no biomedical financial interests or potential conflicts of interest.
Ethics approval
The present study complies with the guidelines for human studies. The research was conducted ethically in accordance with the World Medical Association Declaration of Helsinki. No Institutional Review Board approval was required (Review article).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Tutino, R., Bonomi, A., Zingaretti, C.C. et al. Locally advanced mid/low rectal cancer with synchronous resectable liver metastases: systematic review of the available strategies and outcome. Updates Surg 76, 345–361 (2024). https://doi.org/10.1007/s13304-023-01735-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13304-023-01735-w