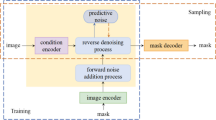
Abstract
Intravascular Ultrasound images (IVUS) is a useful guide for medical practitioners to identify the vascular status of coronary arteries in human beings. IVUS is a unique intracoronary imaging modality that is used as an adjunct to angioplasty to view vessel structures using a catheter with high resolutions. Segmentation of IVUS images has always remained a challenging task due to various impediments, for example, similar tissue components, vessel structures, and artifacts imposed during the acquisition process. Many researchers have applied various techniques to develop standard methods of image interpretation, however, the ultimate goal is still elusive to most researchers. This challenge was presented at the MICCAI- Computing and Visualization for (Intra)Vascular Imaging (CVII) workshop in 2011. This paper presents a major review of recently reported work in the field, with a detailed analysis of various segmentation techniques applied in IVUS, and highlights the directions for future research. The findings recommend a reference database with a larger number of samples acquired at varied transducer frequencies with special consideration towards complex lesions, suitable validation metrics, and ground-truth definition as a standard against which to compare new and current algorithms.








Similar content being viewed by others
References
WHO (2020) Cardiovascular diseases. https://www.who.int/health-topics/cardiovascular-diseases/
Gao Z, Guo W, Liu X, et al (2014) Automated detection framework of the calcified plaque with acoustic shadowing in IVUS images. PLoS ONE 9:. https://doi.org/10.1371/journal.pone.0109997
Subban, Raffel OC, Vasu N, et al (2018) Intravascular ultrasound and optical coherence tomography for the assessment of coronary artery disease and percutaneous coronary intervention optimization: Specific lesion subsets. Indian Heart Journal Interventions 1:95. https://doi.org/10.4103/IHJI.IHJI_33_18
Su S, Hu Z, Lin Q, et al (2017) An artificial neural network method for lumen and media-adventitia border detection in IVUS. Computerized Medical Imaging and Graphics 57:29–39. https://doi.org/10.1016/j.compmedimag.2016.11.003
Bentzon JF, Otsuka F, Virmani R, Falk E (2014) Mechanisms of plaque formation and rupture. Circulation Research 114:1852–1866. https://doi.org/10.1161/CIRCRESAHA.114.302721
Katouzian A, Karamalis A, Sheet D, et al (2012) Iterative self-organizing atherosclerotic tissue labeling in intravascular ultrasound images and comparison with virtual histology. IEEE Transactions on Biomedical Engineering 59:3039–3049. https://doi.org/10.1109/TBME.2012.2213338
Lee J, Hwang YN, Kim GY, et al (2019) Automated classification of dense calcium tissues in gray-scale intravascular ultrasound images using a deep belief network. BMC Medical Imaging 19:1–13. https://doi.org/10.1186/s12880-019-0403-8
Falk E (1992) Why do plaques rupture? Circulation 86:III30-42
VIRMANI R, BURKE AP, KOLODGIE FD, FARB A (2003) Pathology of the Thin-Cap Fibroatheroma: Journal of Interventional Cardiology 16:267–272. https://doi.org/10.1034/j.1600-0854.2003.8042.x
Yock PG, Linker DT, Angelsen BAJ (1989) Two-Dimensional Intravascular Ultrasound: Technical Development and Initial Clinical Experience. Journal of the American Society of Echocardiography 2:296–304. https://doi.org/10.1016/S0894-7317(89)80090-2
Escolar E, Weigold G, Fuisz A, Weissman NJ (2006) New imaging techniques for diagnosing coronary artery disease. Cmaj 174:487–495. https://doi.org/10.1503/cmaj.050925
Waller BF, Pinkerton CA, Slack JD (1992) Intravascular ultrasound: A histological study of vessels during life. The new “gold standard” for vascular imaging. Circulation 85:2305–2310. https://doi.org/10.1161/01.CIR.85.6.2305
Katouzian A, Angelini ED, Carlier SG, et al (2012) A state-of-the-art review on segmentation algorithms in intravascular ultrasound (IVUS) images. IEEE Transactions on Information Technology in Biomedicine 16:823–834. https://doi.org/10.1109/TITB.2012.2189408
Szarski M, Chauhan S (2021) Improved real-time segmentation of Intravascular Ultrasound images using coordinate-aware fully convolutional networks. Computerized Medical Imaging and Graphics 91:101955. https://doi.org/10.1016/j.compmedimag.2021.101955
Faraji M, Cheng I, Naudin I, Basu A (2018) Segmentation of arterial walls in intravascular ultrasound cross-sectional images using extremal region selection. Ultrasonics 84:356–365. https://doi.org/10.1016/j.ultras.2017.11.020
Nishi T, Yamashita R, Imura S, et al (2021) Deep learning-based intravascular ultrasound segmentation for the assessment of coronary artery disease. International Journal of Cardiology 333:55–59. https://doi.org/10.1016/j.ijcard.2021.03.020
Choi A, McPherson DD, Kim H (2017) Visualization of plaque distribution in a curved artery: three-dimensional intravascular ultrasound imaging. Computer Assisted Surgery 22:120–126. https://doi.org/10.1080/24699322.2017.1389389
Yang J, Faraji M, Basu A (2019) Robust segmentation of arterial walls in intravascular ultrasound images using Dual Path U-Net. Ultrasonics 96:24–33. https://doi.org/10.1016/j.ultras.2019.03.014
Balocco S, Gatta C, Ciompi F, et al (2014) Standardized evaluation methodology and reference database for evaluating IVUS image segmentation. Computerized Medical Imaging and Graphics 38:70–90. https://doi.org/10.1016/j.compmedimag.2013.07.001
Räber L, Mintz GS, Koskinas KC, et al (2018) Clinical use of intracoronary imaging. Part 1: guidance and optimization of coronary interventions. An expert consensus document of the European Association of Percutaneous Cardiovascular Interventions. European heart journal 39:3281–3300. https://doi.org/10.1093/eurheartj/ehy285
Zhang P, Li H, Wang R, et al (2021) IVUS plus multivariate analysis for evaluating the stability of coronary artery plaque in coronary heart disease. American journal of translational research 13:9168–9174
Kwon O, Lee PH, Lee S-W, et al (2021) Clinical outcomes of post-stent intravascular ultrasound examination for chronic total occlusion intervention with drug-eluting stents. EuroIntervention 17:e639–e646
Buccheri D, Piraino D, Andolina G, Cortese B (2016) Understanding and managing in-stent restenosis: a review of clinical data, from pathogenesis to treatment. Journal of thoracic disease 8:E1150–E1162. https://doi.org/10.21037/jtd.2016.10.93
Si D, Liu G, Tong Y, He Y (2018) Rotational atherectomy ablation for an unexpandable stent under the guide of IVUS A case report. https://doi.org/10.1097/MD.0000000000009978
Chiang M-H, Yi H-T, Tsao C-R, et al (2013) Rotablation in the treatment of high-risk patients with heavily calcified left-main coronary lesions. Journal of geriatric cardiology: JGC 10:217–225. https://doi.org/10.3969/j.issn.1671-5411.2013.03.009
Guddeti RR, Matsuo Y, Matsuzawa Y, et al (2014) Clinical implications of intracoronary imaging in cardiac allograft vasculopathy. Circulation: Cardiovascular Imaging 8:1–8. https://doi.org/10.1161/CIRCIMAGING.114.002636
Vijayvergiya R, Kasinadhuni G, Revaiah PC, et al (2021) Role of Intravascular Imaging for the Diagnosis of Recanalized Coronary Thrombus. Cardiovascular Revascularization Medicine 32:13–17. https://doi.org/10.1016/j.carrev.2020.12.031
Secemsky EA, Parikh SA, Kohi M, et al (2020) Intravascular ultrasound guidance for lower extremity arterial and venous interventions. EuroIntervention 18:598–608
Herrington DM, Johnson T, Santago P, Snyder WE (1992) Semi-automated boundary detection for intravascular ultrasound. Proceedings - Computers in Cardiology, CIC 1992 103–106. https://doi.org/10.1109/CIC.1992.269436
Sonka M, Zhang X, Siebes M, et al (1995) Segmentation of Intravascular Ultrasound Images: A Knowledge-Based Approach. IEEE Transactions on Medical Imaging 14:719–732. https://doi.org/10.1109/42.476113
Papadogiorgaki M, Mezaris V, Chatzizisis YS, et al (2008) Image Analysis Techniques for Automated IVUS Contour Detection. Ultrasound in Medicine and Biology 34:1482–1498. https://doi.org/10.1016/j.ultrasmedbio.2008.01.022
Dos Santos Filho E, Saijo Y, Yambe T, et al (2006) Segmentation of calcification regions in intravascular ultrasound images by adaptive thresholding. Proceedings - IEEE Symposium on Computer-Based Medical Systems 2006:446–450. https://doi.org/10.1109/CBMS.2006.142
Katouzian A, Angelini ED, Sturm B, Laine AF (2012) Brushlet segmentation for automatic detection of lumen borders in IVUS images: A comparison study. Proceedings - International Symposium on Biomedical Imaging 242–245. https://doi.org/10.1109/ISBI.2012.6235529
Mendizabal-Ruiz EG, Rivera M, Kakadiaris IA (2013) Segmentation of the luminal border in intravascular ultrasound B-mode images using a probabilistic approach. Medical Image Analysis 17:649–670. https://doi.org/10.1016/j.media.2013.02.003
(2011) Proceedings of CVII’11. In: 3rd MICCAI-Workshop on Computation and Visualization for (Intra) Vascular Imaging. Toronto, Canada
Lo Vercio L, Del Fresno M, Larrabide I (2017) Detection of morphological structures for vessel wall segmentation in IVUS using random forests. 12th International Symposium on Medical Information Processing and Analysis 10160:1016012. https://doi.org/10.1117/12.2255748
Destrempes F, Roy Cardinal MH, Saijo Y, et al (2017) Assessment of inter-expert variability and of an automated segmentation method of 40 and 60 MHz IVUS images of coronary arteries. PLoS ONE 12:1–22. https://doi.org/10.1371/journal.pone.0168332
Nishi T, Imura S, Kitahara H, et al (2021) Head-to-head comparison of quantitative measurements between intravascular imaging systems: An in vitro phantom study. IJC Heart and Vasculature 36:100867. https://doi.org/10.1016/j.ijcha.2021.100867
Peng C, Wu H, Kim S, et al (2021) Recent Advances in Transducers for Intravascular Ultrasound (IVUS) Imaging. Sensors (Basel, Switzerland) 21:. https://doi.org/10.3390/s21103540
Finet G, Cachard C, Delachartre P, et al (1998) Artifacts in intravascular ultrasound imaging during coronary artery stent implantation. Ultrasound in Medicine and Biology 24:793–802. https://doi.org/10.1016/S0301-5629(98)00041-6
Hindi A, Peterson C, Barr RG (2013) Artifacts in diagnostic ultrasound. Reports in Medical Imaging 6:29–48. https://doi.org/10.2147/RMI.S33464
Park H-B, Cho Y-H, Cho D-K (2018) IVUS Artifacts and Image Control. In: Coronary Imaging and Physiology. pp 9–17
Bangalore S, Bhatt DL (2013) Coronary intravascular ultrasound. Circulation 127:868–874. https://doi.org/10.1161/CIRCULATIONAHA.113.003534
Ye Y, Yang M, Zhang S, Zeng Y (2017) Percutaneous coronary intervention in left main coronary artery disease with or without intravascular ultrasound: A meta-analysis. PLoS ONE 12:1–13. https://doi.org/10.1371/journal.pone.0179756
LEE C-H (2012) Intravascular Ultrasound Guided Percutaneous Coronary Intervention: A Practical Approach. Journal of Interventional Cardiology 25:86–94. https://doi.org/10.1111/j.1540-8183.2011.00651.x
Lee SY, Choi KH, Song Y Bin, et al (2022) Use of intravascular ultrasound and long-term cardiac death or myocardial infarction in patients receiving current generation drug-eluting stents. Scientific Reports 12:8237. https://doi.org/10.1038/s41598-022-12339-6
Maria GL De, Banning AP (2018) Use of Intravascular Ultrasound Imaging in Percutaneous Coronary Intervention to Treat Left Main Coronary Artery Disease. Radcliffe Cardiology 12:8–12. https://doi.org/10.15420/icr.2017
Gong X, Huang Z, Sun Z, et al (2021) Role of IVUS in the rectification of angiographically judged ramus intermedius and its clinical significance. BMC Cardiovascular Disorders 21:218. https://doi.org/10.1186/s12872-021-02034-1
Vijayvergiya R, Gupta A, Kasinadhuni G, et al (2018) Intravascular ultrasound supported percutaneous coronary intervention of a large diameter right coronary artery. IHJ Cardiovascular Case Reports (CVCR) 2:106–107. https://doi.org/10.1016/j.ihjccr.2018.02.004
Choi KH, Song Y Bin, Lee JM, et al (2019) Impact of Intravascular Ultrasound-Guided Percutaneous Coronary Intervention on Long-Term Clinical Outcomes in Patients Undergoing Complex Procedures. JACC: Cardiovascular Interventions 12:607–620. https://doi.org/10.1016/j.jcin.2019.01.227
Choi IJ, Lim S, Choo EH, et al (2021) Impact of Intravascular Ultrasound on Long-Term Clinical Outcomes in Patients With Acute Myocardial Infarction. JACC Cardiovascular interventions 14:2431–2443. https://doi.org/10.1016/j.jcin.2021.08.021
Kang DY, Ahn JM, Yun SC, et al (2021) Long-Term Clinical Impact of Intravascular Ultrasound Guidance in Stenting for Left Main Coronary Artery Disease. Circulation Cardiovascular interventions 14:e011011. https://doi.org/10.1161/CIRCINTERVENTIONS.121.011011
Vemmou E, Khatri J, Doing AH, et al (2020) Impact of Intravascular Ultrasound Utilization for Stent Optimization on 1-Year Outcomes After Chronic Total Occlusion Percutaneous Coronary Intervention. The Journal of invasive cardiology 32:392–399
Shlofmitz E, Torguson R, Zhang C, et al (2021) Impact of intravascular ultrasound on Outcomes following PErcutaneous coronary interventioN for In-stent Restenosis (iOPEN-ISR study). International Journal of Cardiology 340:17–21. https://doi.org/10.1016/j.ijcard.2021.08.003
Andres V, Mistry N, Singh J (2014) Impact of Intravascular Ultrasound in Clinical Practice. Redcliffe Cardiology 9:156–163
Gao Z, Chung J, Abdelrazek M, et al (2020) Privileged Modality Distillation for Vessel Border Detection in Intracoronary Imaging. IEEE Transactions on Medical Imaging 39:1524–1534. https://doi.org/10.1109/tmi.2019.2952939
Taha AA, Hanbury A (2015) Metrics for evaluating 3D medical image segmentation: analysis, selection, and tool. BMC Medical Imaging 15:29. https://doi.org/10.1186/s12880-015-0068-x
Müller D, Soto-Rey I, Kramer F (2022) Towards a guideline for evaluation metrics in medical image segmentation. BMC Research Notes 15:210. https://doi.org/10.1186/s13104-022-06096-y
Yan J, Lv D, Cui Y (2017) A Novel Segmentation Approach for Intravascular Ultrasound Images. Journal of Medical and Biological Engineering 37:386–394. https://doi.org/10.1007/s40846-017-0233-5
Sethian JA (1996) A fast marching level set method for monotonically advancing fronts. Proceedings of the National Academy of Sciences of the United States of America 93:1591–1595. https://doi.org/10.1073/pnas.93.4.1591
Deschamps T, Cohen LD (2001) Fast extraction of minimal paths in 3D images and applications to virtual endoscopy. Medical Image Analysis 5:281–299. https://doi.org/10.1016/S1361-8415(01)00046-9
Udupa JK, Samarasekera S (1996) Fuzzy Connectedness and Object Definition: Theory, Algorithms, and Applications in Image Segmentation. Graphical Models and Image processing 58:246–261. https://doi.org/10.1109/TPAMI.2004.1265723
Unal G, Bucher S, Carlier S, et al (2008) Shape-driven segmentation of the arterial wall in intravascular ultrasound images. IEEE Transactions on Information Technology in Biomedicine 12:335–347. https://doi.org/10.1109/TITB.2008.920620
Cardinal MHR, Soulez G, Tardif JC, et al (2010) Fast-marching segmentation of three-dimensional intravascular ultrasound images: A pre- and post-intervention study. Medical Physics 37:3633–3647. https://doi.org/10.1118/1.3438476
Mikolajczyk K, Tuytelaars T, Schmid C, et al (2005) A comparison of affine region detectors. International Journal of Computer Vision 65:43–72. https://doi.org/10.1007/s11263-005-3848-x
Faraji M, Shanbehzadeh J, Nasrollahi K, Moeslund TB (2015) Extremal Regions Detection Guided by Maxima of Gradient Magnitude. IEEE Transactions on Image Processing 24:5401–5415. https://doi.org/10.1109/TIP.2015.2477215
Ridler TW, Calvard S (1978) Picture Thresholding Using Iterative Selection Method. IEEE Transactions on Systems, Man and Cybernetics SMC-8:630–632
Xia M, Yan W, Huang Y, et al (2019) IVUS images segmentation using spatial fuzzy clustering and hierarchical level set evolution. Computers in Biology and Medicine 109:207–217. https://doi.org/10.1016/j.compbiomed.2019.04.029
Osher S, Sethian JA (1988) Fronts Propagating with Curvature Dependent Speed: Algorithms Based on Hamilton-Jacobi Formulations. Journal of Computational Physics 79:12–49
Lo Vercio L, Orlando JI, del Fresno M, Larrabide I (2016) Assessment of image features for vessel wall segmentation in intravascular ultrasound images. International Journal of Computer Assisted Radiology and Surgery 11:1397–1407. https://doi.org/10.1007/s11548-015-1345-4
Li C, Xu C, Gui C, Fox MD (2010) Distance regularized level set evolution and its application to image segmentation. IEEE Transactions on Image Processing 19:3243–3254. https://doi.org/10.1109/TIP.2010.2069690
Hammouche A, Cloutier G, Tardif JC, et al (2019) Automatic IVUS lumen segmentation using a 3D adaptive helix model. Computers in Biology and Medicine 107:58–72. https://doi.org/10.1016/j.compbiomed.2019.01.023
Kermani A, Ayatollahi A (2019) A new nonparametric statistical approach to detect lumen and Media-Adventitia borders in intravascular ultrasound frames. Computers in Biology and Medicine 104:10–28. https://doi.org/10.1016/j.compbiomed.2018.10.024
Yang J, Tong L, B MF, Basu A (2018) IVUS-Net: An Intravascular Ultrasound Segmentation Network. In: Basu A. BS (ed) Smart Multimedia ICSM 2018 Lecture Notes in Computer Science. Springer International Publishing, Springer, Cham, pp 367–377
Jodas DS, Pereira AS, Tavares JMRS (2017) Automatic segmentation of the lumen region in intravascular images of the coronary artery. Medical Image Analysis 40:60–79. https://doi.org/10.1016/j.media.2017.06.006
Lo Vercio L, del Fresno M, Larrabide I (2019) Lumen-intima and media-adventitia segmentation in IVUS images using supervised classifications of arterial layers and morphological structures. Computer Methods and Programs in Biomedicine 177:113–121. https://doi.org/10.1016/j.cmpb.2019.05.021
Xia M, Yan W, Huang Y, et al (2019) IVUS image segmentation using superpixel-wise fuzzy clustering and level set evolution. Applied Sciences (Switzerland) 9:1–18. https://doi.org/10.3390/APP9224967
Wang YY, Peng WX, Qiu CH, et al (2019) Fractional-order Darwinian PSO-based feature selection for media-adventitia border detection in intravascular ultrasound images. Ultrasonics 92:1–7. https://doi.org/10.1016/j.ultras.2018.06.012
Essa E, Xie X (2017) Automatic segmentation of cross-sectional coronary arterial images. Computer Vision and Image Understanding 165:97–110. https://doi.org/10.1016/j.cviu.2017.11.004
Balocco S, Gatta C, Ciompi F, et al (2011) Combining growcut and temporal correlation for IVUS lumen segmentation. Lecture Notes in Computer Science (including subseries Lecture Notes in Artificial Intelligence and Lecture Notes in Bioinformatics) 6669 LNCS:556–563. https://doi.org/10.1007/978-3-642-21257-4_69
Michael K, Andrew W, Terzopoulos D (1988) Snakes: Active Contour Models. International Journal of Computer Vision 1:321–331. https://doi.org/10.1016/B978-0-12-386454-3.00786-7
Williams DJ, Shah M (1992) A Fast Algorithm for Active Contours and Curvature Estimation. CVGIP: Image Understanding 55:14–26
Klingensmith JD, Shekhar R, Vince D (2000) Evaluation of three- dimensional segmentation algorithms for the identification of luminal and medial-adventitial borders in intravascular ultrasound images. IEEE Transactions on Medical Imaging 19:996–1011. https://doi.org/10.1109/42.887615
Kovalski G, Beyar R, Shofti R, Azhari H (2000) Three-dimensional automatic quantitative analysis of intravascular ultrasound images. Ultrasound in Medicine and Biology 26:527–537. https://doi.org/10.1016/S0301-5629(99)00167-2
Xu C, Prince JL (1998) Snakes, shapes, and gradient vector flow. IEEE Transactions on Image Processing 7:359–369. https://doi.org/10.1109/83.661186
China D, Nag MK, Mandana KM, et al (2016) Automated in vivo delineation of lumen wall using intravascular ultrasound imaging. In: 2016 38th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC),. pp 4125–4127
Plissiti ME, Fotiadis DI, Michalis LK, Bozios GE (2004) An automated method for lumen and media-adventitia border detection in a sequence of IVUS frames. IEEE Transactions on Information Technology in Biomedicine 8:131–141. https://doi.org/10.1109/TITB.2004.828889
Giannoglou GD, Chatzizisis YS, Koutkias V, et al (2007) A novel active contour model for fully automated segmentation of intravascular ultrasound images: In vivo validation in human coronary arteries. Computers in Biology and Medicine 37:1292–1302. https://doi.org/10.1016/j.compbiomed.2006.12.003
Tayel MB, Massoud MA, Shehata YF (2014) An Automatic Segmentation for Determination of IV Vessel Boundaries. International Journal of Bioscience, Biochemistry and Bioinformatics 4:218–223. https://doi.org/10.7763/ijbbb.2014.v4.343
Tayel MB, Massoud MA, Farouk Y (2017) A modified segmentation method for determination of IV vessel boundaries. Alexandria Engineering Journal 56:449–457. https://doi.org/10.1016/j.aej.2017.04.002
Gao Z, Hau WK, Lu M, et al (2015) Automated framework for detecting lumen and media-adventitia borders in intravascular ultrasound images. Ultrasound in Medicine and Biology 41:2001–2021. https://doi.org/10.1016/j.ultrasmedbio.2015.03.022
Chan TF, Vese LA (2001) Active Contours Without Edges. IEEE Transactions on Image Processing 10:266–277
Sofian H, Than J, Ming C, et al (2015) Detection of the Lumen Boundary in the Coronary Artery Disease. In: IEEE International WIE Conference on Electrical and Computer Engineering (WIECON-ECE). Dhaka, Bangladesh, pp 143–146
Destrempes F, Roy Cardinal M-H, Allard L, et al (2014) Segmentation method of intravascular ultrasound images of human coronary arteries. Computerized Medical Imaging and Graphics 38:91–103. https://doi.org/10.1016/j.compmedimag.2013.09.004
Taki A, Najafi Z, Roodaki A, et al (2008) Automatic segmentation of calcified plaques and vessel borders in IVUS images. International Journal of Computer Assisted Radiology and Surgery 3:347–354. https://doi.org/10.1007/s11548-008-0235-4
Zakeri FS, Setarehdan SK, Norouzi S (2017) Automatic media-adventitia IVUS image segmentation based on sparse representation framework and dynamic directional active contour model. Computers in Biology and Medicine 89:561–572. https://doi.org/10.1016/j.compbiomed.2017.03.022
Elad M (2010) Sparse and Redundant Representations: from Theory to Applications in Signal and Image Processing. Springer New York
Wang G, Liang J, Wang Y (2011) Dynamic Directional Convolution Vector Field. In: International Conference on Internet Computing and Information Services. IEEE, Hong Kong, pp 107–110
Garcia D (2010) Robust smoothing of gridded data in one and higher dimensions with missing values. Computational statistics & data analysis 54:1167–1178. https://doi.org/10.1016/j.csda.2009.09.020.Robust
Cheng J, Foo SW (2006) Dynamic Directional Gradient Vector Flow for Snakes. IEEE Transactions on Image Processing 15:1563–1571
Santos Filho E, Saijo Y, Tanaka A, Yoshizawa M (2008) Detection and Quantification of Calcifications in Intravascular Ultrasound Images by Automatic Thresholding. Ultrasound in Medicine and Biology 34:160–165. https://doi.org/10.1016/j.ultrasmedbio.2007.06.025
Basij M, Taki A, Yazdchi M (2014) Automatic Shadow Enhancement in Intra Vascular Ultrasound (IVUS) Images. In: Middle East Conference on Biomedical Engineering (MECBME). pp 309–312
Basij M, Yazdchi M, Taki A, Moallem P (2017) An automatic approach for artifacts detection and shadow enhancement in intravascular ultrasound images. Signal, Image and Video Processing 11:1009–1016. https://doi.org/10.1007/s11760-016-1051-x
Lee JH, Hwang YN, Kim GY, Sung Min K (2018) Segmentation of the lumen and media-adventitial borders in intravascular ultrasound images using a geometric deformable model. IET Image Processing 12:1881–1891. https://doi.org/10.1049/iet-ipr.2017.1143
Werlberger M, Trobin W, Pock T, et al (2009) Anisotropic huber-L1 optical flow. British Machine Vision Conference, BMVC 2009 - Proceedings 1–11. https://doi.org/10.5244/C.23.108
Preparata FP, Shamos MI (1985) Computational geometry: an introduction. Springer-Verlag, Berlin, Heidelberg.
Downe RW, Wahle A, Kovarnik T, et al (2008) Segmentation of intravascular ultrasound images using graph search and a novel cost function. In: 2nd MICCAI Workshop on Computer Vision for Intravascular and Intracardiac Imaging. New York, pp 71–79
Wang Y, Gao X, Wang Y, Sun J (2021) Adventitia segmentation in intravascular ultrasound images based on improved Snake algorithm. Optik 241:167175. https://doi.org/10.1016/j.ijleo.2021.167175
Moshfegh A, Javadzadegan A, Mohammadi M, et al (2019) Development of an innovative technology to segment luminal borders of intravascular ultrasound image sequences in a fully automated manner. Computers in Biology and Medicine 108:111–121. https://doi.org/10.1016/j.compbiomed.2019.03.008
Gil D, Radeva P, J. Saludes, Mauri J (2000) Automatic segmentation of artery wall in coronary IVUS images: A probabilistic approach. Computers in Cardiology 27:687–690. https://doi.org/10.1109/CIC.2000.898617
Yan J, Liu H, Cui Y (2014) A random walk-based method for segmentation of intravascular ultrasound images. In: Molthen RC, Weaver JB (eds) Proc. SPIE 9038, Medical Imaging 2014: Biomedical Applications in Molecular, Structural, and Functional Imaging, 903825. SPIE, San Diego, California, United States, pp 529–537
Sun S, Sonka M, Beichel RR (2013) Graph-based IVUS segmentation with efficient computer-aided refinement. IEEE Transactions on Medical Imaging 32:1536–1549. https://doi.org/10.1109/TMI.2013.2260763
Y Y, X Z, R W, et al (2010) LOGISMOS–layered optimal graph image segmentation of multiple objects and surfaces: cartilage segmentation in the knee joint. IEEE Transactions on Medical Imaging 29:2023–2037. https://doi.org/10.1109/TMI.2010.2058861
Oguz I, Sonka M (2014) LOGISMOS-B: Layered Optimal graph image segmentation of multiple objects and surfaces for the brain. IEEE Transactions on Medical Imaging 33:1220–1235. https://doi.org/10.1109/TMI.2014.2304499
Sonka M, Abramoff MD (2016) Quantitative Analysis of Retinal OCT. Medical Image Analysis 33:165–169. https://doi.org/10.1016/j.media.2016.06.001
Quo Z, Zhang L, Lu L, et al (2018) Deep LOGISMOS: Deep learning graph-based 3D segmentation of pancreatic tumors on CT scans. Proceedings - International Symposium on Biomedical Imaging 2018-April:1230–1233. https://doi.org/10.1109/ISBI.2018.8363793
Zhang H, Lee K, Chen Z, et al (2019) LOGISMOS-JEI: Segmentation using optimal graph search and just-enough interaction. Elsevier Inc.
Oguz I, Bogunović H, Kashyap S, et al (2016) LOGISMOS: A FAMILY OF GRAPH-BASED OPTIMAL IMAGE SEGMENTATION METHODS. In: Medical Image Recognition, Segmentation and Parsing. pp 179–208
Will S, Hermes L, Buhmann JM, Puzicha J (2000) On learning texture edge detectors. Proceedings 2000 International Conference on Image Processing (Cat No00CH37101) 3:877–880. https://doi.org/10.1109/ICIP.2000.899596
Dempster AP, Laird NM, Rubin DB (1977) Maximum Likelihood from Incomplete Data via the EM Algorithm. Journal of the Royal Statistical Society Series B (Methodological) 39:1–38
Mendizabal-Ruiz E, Rivera M, Kakadiaris I (2008) A probabilistic segmentation method for the identification of luminal borders in intravascular ultrasound images. In: IEEE Conference on Computer Vision and Pattern Recognition. Anchorage, AK, pp 1–8
Laws KI (1980) Rapid texture identification. In: Wiener TF (ed) SPIE 0238 Conference on Image Processing for Missile Guidance. SPIE, San Diego, United States
Cortes C, Vapnik V (1995) Support-vector networks. Machine Learning 20:273–297. https://doi.org/10.1007/BF00994018
Nocedal J, Stephen JW (1999) Numerical Optimization. Springer, New York, NY
Otsu N (1979) A Thresholding Selection Method from Gray-Level Histograms. IEEE Transactions on Systems, Man and Cybernetics SMC-9:62–66
Ciompi F, Pujol O, Gatta C, et al (2012) Holimab: A holistic approach for media–adventitia border detection in intravascular ultrasound. Medical Image Analysis 16:1085–1100
Starck JL, Murtagh F, Bijaou A (1998) Image Processing and Data Analysis: The Multiscale Approach. Cambridge Univ. Press, U.K.
Valois R De, Valois K De (1988) Spatial Vision. NY: Oxford Univ. Press, New York
Beck J, A. Sutter, Ivry R (1987) Spatial frequency channels and perceptual grouping in texture segregation. Computer Vision, Graphics and Image Processing 37:299–325
Klingensmith E, Nair A, Kuban B, Vince D (2004) Segmentation of three- dimensional intravascular ultrasound images using spectral analysis and a dual active surface model. In: IEEE International Ultrasonics Symposium. IEEE, Montreal, Quebec, Canada, pp 1765–1768
G.Meyer F, R.Coifman R (1997) Brushlets: A Tool for Directional Image Analysis and Image Compression. Applied and Computational Harmonic Analysis 4:147–187. https://doi.org/10.1006/acha.1997.0208
Freeman WT, Adelson EH (1991) The Design and Use of Steerable Filters. IEEE Transactions on Pattern Analysis and Machine Intelligence 13:891–906
Besag J (1986) On the Statistical Analysis of Dirty Pictures. Journal of the Royal Statistical Society Series B (Methodological) 48:259–302
Essa E, Xie X, Sazonov I, et al (2013) Shape Prior Model for Media-Adventitia Border Segmentation in IVUS Using Graph Cut. In: Lecture Notes in Computer Science (including subseries Lecture Notes in Artificial Intelligence and Lecture Notes in Bioinformatics). pp 114–123
Breiman L (2001) Random forests. Machine Learning 45:5–32. https://doi.org/10.1023/A:1010933404324
Chen F, Ma R, Liu J, et al (2018) Lumen and media-adventitia border detection in IVUS images using texture enhanced deformable model. Computerized Medical Imaging and Graphics 66:1–13. https://doi.org/10.1016/j.compmedimag.2018.02.003
Awad J, Krasinski A, Parraga G, Fenster A (2010) Texture analysis of carotid artery atherosclerosis from three-dimensional ultrasound images. Medical Physics 37:1382–1391. https://doi.org/10.1118/1.3301592
Wu CM, Chen YC (1992) Statistical feature matrix for texture analysis. CVGIP: Graphical Models and Image Processing 54:407–419. https://doi.org/10.1016/1049-9652(92)90025-S
Haralick RM, Shanmugam K, Dinstein I (1973) Texture features for image classification. IEEE Transactions on Systems, Man and Cybernetics SMC-3:610–621
Christodoulou CI, Pattichis CS, Pantziaris M, Nicolaides A (2003) Texture-based classification of atherosclerotic carotid plaques. IEEE Transactions on Medical Imaging 22:902–912. https://doi.org/10.1109/TMI.2003.815066
Loizou CP, Theofanous C, Pantziaris M, Kasparis T (2014) Despeckle filtering software toolbox for ultrasound imaging of the common carotid artery. Computer Methods and Programs in Biomedicine 114:109–124. https://doi.org/10.1016/j.cmpb.2014.01.018
Rodríguez J, Kuncheva L, Alonso C (2006) Rotation Forest: A New Classifier Ensemble Method. IEEE transactions on pattern analysis and machine intelligence 28:1619–1630. https://doi.org/10.1109/TPAMI.2006.211
Tong J, Li K, Lin W, et al (2021) Automatic lumen border detection in IVUS images using dictionary learning and kernel sparse representation. Biomedical Signal Processing and Control 66:102489. https://doi.org/10.1016/j.bspc.2021.102489
Marone J, Balocco S, Bolanos M, et al (2016) Learning the Lumen Border using a Convolutional Neural Networks classifier. In: CVII-Stent Workshop - MICCAI. Athens, pp 1–8
Su S, Gao Z, Zhang H, et al (2017) A Detection of Lumen and Media-Adventitia Borders in IVUS images using Sparse Auto-Encoder Neural Network. In: 2017 IEEE 14th International Symposium on Biomedical Imaging (ISBI 2017). pp 1120–1124
Litjens G, Kooi T, Bejnordi BE, et al (2017) A survey on deep learning in medical image analysis. Medical Image Analysis 42:60–88. https://doi.org/10.1016/j.media.2017.07.005
Badrinarayanan V, Kendall A, Cipolla R (2017) SegNet: A Deep Convolutional Encoder-Decoder Architecture for Image Segmentation. IEEE Transactions on Pattern Analysis and Machine Intelligence 39:2481–2495. https://doi.org/10.1109/TPAMI.2016.2644615
Ronneberger O, Fischer P, Brox T (2015) U-net: Convolutional networks for biomedical image segmentation. Lecture Notes in Computer Science (including subseries Lecture Notes in Artificial Intelligence and Lecture Notes in Bioinformatics) 9351:234–241. https://doi.org/10.1007/978-3-319-24574-4_28
He K, Zhang X, Ren S, Sun J (2015) Delving Deep into Rectifiers: Surpassing Human-Level Performance on ImageNet Classification. In: 2015 IEEE International Conference on Computer Vision (ICCV). Santiago, pp 1026–1034
Molony D, Hosseini H, Samady H (2018) TCT-2 Deep IVUS: A machine learning framework for fully automatic IVUS segmentation. Journal of the American College of Cardiology. https://doi.org/10.1016/j.jacc.2018.08.1077
Kim S, Yeonggul J, Byunghwan J, et al (2018) Fully Automatic Segmentation of Coronary Arteries Based on Deep Neural Network in Intravascular Ultrasound Images. In: Springer Nature Switzerland AG. Springer International Publishing, pp 161–168
Molony D, Samady H (2019) TCT-342 DeepIVUS: A Machine Learning Platform for Fully Automatic IVUS Segmentation and Phenotyping. Journal of the American College of Cardiology 74:B339. https://doi.org/10.1016/j.jacc.2019.08.424
Mehta R, Sivaswamy J (2017) M-NET: A Convolutional Neural Network for Deep Brain Structure Segmentation. In: IEEE International Symposium on Biomedical Imaging 2017. Melbourne, Australia, pp 437–440
Maturana D, Scherer S (2015) VoxNet: A 3D Convolutional Neural Network for real-time object recognition. In: 2015 IEEE/RSJ International Conference on Intelligent Robots and Systems (IROS). Hamburg, pp 922–928
Jeelani H, Martin J, Vasquez F, et al (2018) Image quality affects deep learning reconstruction of MRI. In: 2018 IEEE 15th International Symposium on Biomedical Imaging (ISBI 2018). Washington, DC, pp 357–360
Abadi M, Agarwal A, Barham P, et al (2015) TensorFlow: Large-Scale Machine Learning on Heterogeneous Systems
Kingma DP, Ba J (2014) Adam: A Method for Stochastic Optimization. In: International Conference on Learning Representations (ICLR)
Vapnik V, Vashist A (2009) A new learning paradigm: Learning using privileged information. Neural Networks 22:544–557. https://doi.org/10.1016/j.neunet.2009.06.042
Vapnik V, Izmailov R (2015) Learning using privileged information: Similarity control and knowledge transfer. Journal of Machine Learning Research 16:2023–2049
Pociask E, Malinowski KP, Ślęzak M, et al (2018) Fully Automated Lumen Segmentation Method for Intracoronary Optical Coherence Tomography. Journal of Healthcare Engineering 2018:1–13. https://doi.org/10.1155/2018/1414076
Cao Y, Jin Q, Chen Y, et al Automatic Side Branch Ostium Detection and Main Vascular Segmentation in Intravascular Optical Coherence Tomography Images
Amrute JM, Athanasiou LS, Rikhtegar F, et al (2018) Polymeric endovascular strut and lumen detection algorithm for intracoronary optical coherence tomography images. Journal of Bioemdical Optics 23:1–14
Ziemer PGP, Bulant CA, Orlando JI, et al (2020) Automated lumen segmentation using multi-frame convolutional neural networks in intravascular ultrasound datasets. European Heart Journal - Digital Health 1:75–82. https://doi.org/10.1093/ehjdh/ztaa014
Li K, Tong J, Zhu X, Xia S (2021) Automatic Lumen Border Detection in IVUS Images Using Deep Learning Model and Handcrafted Features. Ultrasonic Imaging 43:59–73. https://doi.org/10.1177/0161734620987288
Jung-Eun P, Jihoon K, Do-Yoon K, et al (2021) TCTAP A-044 Deep Learning Segmentation of Lumen and Vessel on IVUS Images. Journal of the American College of Cardiology 77:S27–S27. https://doi.org/10.1016/j.jacc.2021.03.075
Shinohara H, Kodera S, Ninomiya K, et al (2021) Automatic detection of vessel structure by deep learning using intravascular ultrasound images of the coronary arteries. Plos One 16:1–14. https://doi.org/10.1371/journal.pone.0255577
Zhu F, Gao Z, Zhao C, et al (2022) A Deep Learning-based Method to Extract Lumen and Media-Adventitia in Intravascular Ultrasound Images. Ultrasonic Imaging. https://doi.org/10.1177/01617346221114137
Cui H, Xia Y, Zhang Y (2020) Supervised machine learning for coronary artery lumen segmentation in intravascular ultrasound images. International Journal of Newmerical Methods in Biomedical Engineering 36:e3348
Bargsten L, Raschka S, Schlaefer A (2021) Capsule networks for segmentation of small intravascular ultrasound image datasets. International Journal of Computer Assisted Radiology and Surgery 16:1243–1254. https://doi.org/10.1007/s11548-021-02417-x
Eslamizadeh M, Attarodi G, Dabanloo NJ, et al (2017) The segmentation of lumen boundaries at intravascular ultrasound images using fuzzy approach. Computing in Cardiology 44:1–4. https://doi.org/10.22489/CinC.2017.288-285
Zheng S, Bing-Ru L (2016) Fast retrieval of calcification from sequential intravascular ultrasound gray-scale images. Bio-Medical Materials and Engineering 27:183–195. https://doi.org/10.3233/BME-161575
Anam S, Uchino E, Suetake N (2014) Coronary plaque boundary enhancement in IVUS image by using a modified Perona-Malik diffusion filter. International Journal of Biomedical Imaging 2014:. https://doi.org/10.1155/2014/740627
Gronningsaeter A, Angelsen BAJ, Torp HG, et al (1995) Blood Noise Reduction in Intravascular Ultrasound Imaging. IEEE Transactions on Ultrasonics, Ferroelectrics and Frequency Control 42:200–209. https://doi.org/10.1109/58.365234
Hibi K, Takagi A, Zhang X, et al (2000) Feasibility of a novel blood noise reduction algorithm to enhance reproducibility of ultra-high-frequency intravascular ultrasound images. Circulation 102:1657–1663. https://doi.org/10.1161/01.CIR.102.14.1657
China D, Mitra P, Chakraborty C, Mandana KM (2015) Wavelet based non local means filter for despeckling of intravascular ultrasound image. 2015 International Conference on Advances in Computing, Communications and Informatics, ICACCI 2015 1361–1365. https://doi.org/10.1109/ICACCI.2015.7275802
Katouzian A, Baseri B, Konofagou EE, Laine AF (2008) Automatic detection of blood versus non-blood regions on intravascular ultrasound (IVUS) images using wavelet packet signatures. In: SPIE 6920, Medical Imaging 2008: Ultrasonic Imaging and Signal Processing, 69200H
Yu Y, Acton ST (2002) Speckle reducing anisotropic diffusion. IEEE Transactions on Image Processing 11:1260–1270. https://doi.org/10.1109/TIP.2002.804276
Haralick, Robert M., Shanmugam. K A, Dinstein I (1973) Textural Features for Image Classification. IEEE Transactions on Systems, Man and Cybernetics SMC-3:610–621
Ojala T, Pietikäinen M, Mäenpää T (2002) Multiresolution Gray scale and rotation invariant texture classification with local binary patterns. IEEE Trans Pattern Analysis and Machine Intelligence 24:971–987. https://doi.org/10.1007/3-540-45054-8_27
Lo Vercio L, Del Fresno M, Larrabide I (2017) Detection of morphological structures for vessel wall segmentation in IVUS using random forests. In: 12th International Symposium on Medical Information Processing and Analysis
Huang G-B, Zhu Q-Y, Siew C-K (2006) Extreme learning machine: Theory and applications. Neurocomputing 79:489–501. https://doi.org/10.1016/j.neucom.2005.12.126
Couceiro MS, Rocha RP, Ferreira NMF, Machado JAT (2012) Introducing the fractional-order Darwinian PSO. Signal, Image and Video Processing 6:343–350. https://doi.org/10.1007/s11760-012-0316-2
Liu S, Neleman T, Hartman EMJ, et al (2020) Automated Quantitative Assessment of Coronary Calcification Using Intravascular Ultrasound. Ultrasound in Medicine and Biology 46:2801–2809. https://doi.org/10.1016/j.ultrasmedbio.2020.04.032
Wongwarn J, Rasmequan S (2019) Tunica Media Localization in Intravascular Image with Shadow Artifact Constraint using Circular-like Estimating Techniques. In: Proceedings of 2019 4th International Conference on Information Technology: Encompassing Intelligent Technology and Innovation Towards the New Era of Human Life, InCIT 2019. IEEE, Bangkok, Thailand, pp 83–88
Sinha P, Wu Y, Psaromiligkos I, Zilic Z (2020) Lumen Media Segmentation of IVUS Images via Ellipse Fitting Using a Wavelet-Decomposed Subband CNN. In: IEEE International Workshop on Machine Learning for Signal Processing, MLSP. ESPOO, Finland
Huang Y, Yan W, Xia M, et al (2020) Vessel membrane segmentation and calcification location in intravascular ultrasound images using a region detector and an effective selection strategy. Computer Methods and Programs in Biomedicine 189:105339. https://doi.org/10.1016/j.cmpb.2020.105339
Dong L, Jiang W, Lu W, et al (2021) Automatic segmentation of coronary lumen and external elastic membrane in intravascular ultrasound images using 8-layer U-Net. BioMedical Engineering Online 20:1–11. https://doi.org/10.1186/s12938-021-00852-0
Acknowledgements
The authors would like to acknowledge Guru Nanak Dev Engineering College, Ludhiana, Punjab (India) and IKG Punjab Technical University, Kapurthala, Punjab (India) for their support in this research work.
Funding
.
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Christine P. Hendon.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
Appendix I
Appendix I
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Arora, P., Singh, P., Girdhar, A. et al. A State-Of-The-Art Review on Coronary Artery Border Segmentation Algorithms for Intravascular Ultrasound (IVUS) Images. Cardiovasc Eng Tech 14, 264–295 (2023). https://doi.org/10.1007/s13239-023-00654-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13239-023-00654-6