Abstract
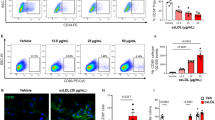
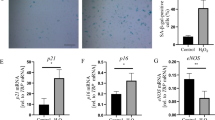
Endothelial dysfunction is an important factor in the development of vascular diseases such as atherosclerosis, hypertension and diabetes. Methylglyoxal (MG) is a highly reactive dicarbonyl metabolite that is an extremely toxic glucose degradation product with strong oxidative activity. MG is involved in the pathogenesis of vascular complications of diabetes. Several studies have reported increased MG levels in pathology models of vascular injury. The present study investigated the genome-wide transcriptional responses of human umbilical vein endothelial cells (HUVECs) exposed to MG by microarray gene expression profiling. As a result, we identified 1,624 genes that were 1.5-fold up-or down-regulated within 12 h of MG treatment. The differentially expressed genes that were dysregulated in many biological processes included inflammatory responses, cell cycle, apoptosis, and cell adhesion. These results demonstrate the MG induced genome-wide alterations in expression profile in human endothelial cells and indicate that MG may cause cytotoxicity and tissue injury in the human endothelium. The data supports the view that MG-stimulated changes in gene expression contribute to the development of vascular disease.
Similar content being viewed by others
References
Schalkwijk, C.G. & Stehouwer, C.D. Vascular complications in diabetes mellitus: the role of endothelial dysfunction. Clin. Sci. (Lond) 109, 143–159 (2005).
Victor, V.M. et al. Oxidative stress, endothelial dysfunction and atherosclerosis. Curr. Pharm. Des. 15, 2988–3002 (2009).
Ray, S. & Ray, M. Isolation of methylglyoxal synthase from goat liver. J. Biol. Chem. 256, 6230–6233 (1981).
Phillips, S.A. & Thornalley, P.J. The formation of methylglyoxal from triose phosphates. Investigation using a specific assay for methylglyoxal. Eur. J. Biochem. 212, 101–105 (1993).
Thornalley, P.J., Langborg, A. & Minhas, H.S. Formation of glyoxal, methylglyoxal and 3-deoxyglucosone in the glycation of proteins by glucose. Biochem. J. 344Pt 1, 109–116 (1999).
Casazza, J.P., Felver, M.E. & Veech, R.L. The metabolism of acetone in rat. J. Biol. Chem. 259, 231–236 (1984).
Yu, P.H., Wright, S., Fan, E.H., Lun, Z.R. & Gubisne-Harberle, D. Physiological and pathological implications of semicarbazide-sensitive amine oxidase. Biochim. Biophys. Acta 1647, 193–199 (2003).
Ahmed, M.U., Brinkmann Frye, E., Degenhardt, T.P., Thorpe, S.R. & Baynes, J.W. N-epsilon-(carboxyethyl) lysine, a product of the chemical modification of proteins by methylglyoxal, increases with age in human lens proteins. Biochem. J. 324(Pt 2), 565–570 (1997).
Brownlee, M. Lilly Lecture 1993. Glycation and diabetic complications. Diabetes 43, 836–841 (1994).
Bourajjaj, M., Stehouwer, C.D., van Hinsbergh, V.W. & Schalkwijk, C.G. Role of methylglyoxal adducts in the development of vascular complications in diabetes mellitus. Biochem. Soc. Trans. 31, 1400–1402 (2003).
Karachalias, N., Babaei-Jadidi, R., Ahmed, N. & Thornalley, P.J. Accumulation of fructosyl-lysine and advanced glycation end products in the kidney, retina and peripheral nerve of streptozotocin-induced diabetic rats. Biochem. Soc. Trans. 31, 1423–1425 (2003).
Schleicher, E.D., Wagner, E. & Nerlich, A.G. Increased accumulation of the glycoxidation product N(epsilon)-(carboxymethyl)lysine in human tissues in diabetes and aging. J. Clin. Invest. 99, 457–468 (1997).
Kilhovd, B.K. et al. Increased serum levels of the specific AGE-compound methylglyoxal-derived hydroimidazolone in patients with type 2 diabetes. Metabolism 52, 163–167 (2003).
Shinohara, M. et al. Overexpression of glyoxalase-I in bovine endothelial cells inhibits intracellular advanced glycation endproduct formation and prevents hyperglycemia-induced increases in macromolecular endocytosis. J. Clin. Invest. 101, 1142–1147 (1998).
Beisswenger, P.J. et al. Susceptibility to diabetic nephropathy is related to dicarbonyl and oxidative stress. Diabetes 54, 3274–3281 (2005).
Wang, X., Desai, K., Chang, T. & Wu, L. Vascular methylglyoxal metabolism and the development of hypertension. J. Hypertens. 23, 1565–1573 (2005).
Wang, X., Desai, K., Clausen, J.T. & Wu, L. Increased methylglyoxal and advanced glycation end products in kidney from spontaneously hypertensive rats. Kidney Int. 66, 2315–2321 (2004).
Zeng, J. & Davies, M.J. Evidence for the formation of adducts and S-(carboxymethyl)cysteine on reaction of alpha-dicarbonyl compounds with thiol groups on amino acids, peptides, and proteins. Chem. Res. Toxicol. 18, 1232–1241 (2005).
Lapolla, A. et al. Glyoxal and methylglyoxal levels in diabetic patients: quantitative determination by a new GC/MS method. Clin. Chem. Lab. Med. 41, 1166–1173 (2003).
Meng, Q.H. et al. Increased plasma methylglyoxal level, inflammation, and vascular endothelial dysfunction in diabetic nephropathy. Clin. Biochem. 44, 307–311 (2011).
Fukunaga, M. et al. Methylglyoxal induces apoptosis through activation of p38 MAPK in rat Schwann cells. Biochem. Biophys. Res. Commun. 320, 689–695 (2004).
Kim, J., Son, J.W., Lee, J.A., Oh, Y.S. & Shinn, S.H. Methylglyoxal induces apoptosis mediated by reactive oxygen species in bovine retinal pericytes. J. Korean Med. Sci. 19, 95–100 (2004).
Akhand, A.A. et al. Glyoxal and methylglyoxal trigger distinct signals for map family kinases and caspase activation in human endothelial cells. Free Radic. Biol. Med. 31, 20–30 (2001).
Huang, W.J. et al. Ras activation modulates methylglyoxal-induced mesangial cell apoptosis through superoxide production. Ren. Fail. 29, 911–921 (2007).
Du, J. et al. Superoxide-mediated early oxidation and activation of ASK1 are important for initiating methylglyoxal-induced apoptosis process. Free Radic. Biol. Med. 31, 469–478 (2001).
Amicarelli, F. et al. Scavenging system efficiency is crucial for cell resistance to ROS-mediated methylglyoxal injury. Free Radic. Biol. Med. 35, 856–871 (2003).
Berlanga, J. et al. Methylglyoxal administration induces diabetes-like microvascular changes and perturbs the healing process of cutaneous wounds. Clin. Sci. (Lond) 109, 83–95 (2005).
Huang da, W. et al. DAVID Bioinformatics Resources: expanded annotation database and novel algorithms to better extract biology from large gene lists. Nucleic Acids Res. 35, W169–175 (2007).
Ogawa, S. et al. Methylglyoxal is a predictor in type 2 diabetic patients of intima-media thickening and elevation of blood pressure. Hypertension 56, 471–476 (2010).
Beisswenger, P.J., Howell, S.K., Nelson, R.G., Mauer, M. & Szwergold, B.S. alpha-oxoaldehyde metabolism and diabetic complications. Biochem. Soc. Trans. 31, 1358–1363 (2003).
Abe, M. et al. Methylglyoxal augments intracellular oxidative stress in human aortic endothelial cells. Free Radic. Res. 44, 101–107 (2010).
Nakayama, M., Sakai, A., Numata, M. & Hosoya, T. Hyper-vascular change and formation of advanced glycation endproducts in the peritoneum caused by methylglyoxal and the effect of an anti-oxidant, sodium sulfite. Am. J. Nephrol. 23, 390–394 (2003).
Wang, X.X., Desai, K., Chang, T.J. & Wu, L.Y. Vascular methylglyoxal metabolism and the development of hypertension. J. Hypertens. 23, 1565–1573 (2005).
Chen, Y., Zhao, S. & Xiang, R. RTN3 and RTN4: Candidate modulators in vascular cell apoptosis and atherosclerosis. J. Cell. Biochem. 111, 797–800.
Millette, E., Rauch, B.H., Kenagy, R.D., Daum, G. & Clowes, A.W. Platelet-derived growth factor-BB transactivates the fibroblast growth factor receptor to induce proliferation in human smooth muscle cells. Trends Cardiovasc. Med. 16, 25–28 (2006).
Wagsater, D., Zhu, C., Bjorck, H.M. & Eriksson, P. Effects of PDGF-C and PDGF-D on monocyte migration and MMP-2 and MMP-9 expression. Atherosclerosis 202, 415–423 (2009).
Gigante, B., Bennet, A.M., Leander, K., Vikstrom, M. & de Faire, U. The interaction between coagulation factor 2 receptor and interleukin 6 haplotypes increases the risk of myocardial infarction in men. PLoS One 5 (2010).
Rosenberger, C. et al. Expression of hypoxia-inducible factor-1alpha and -2alpha in hypoxic and ischemic rat kidneys. J. Am. Soc. Nephrol. 13, 1721–1732 (2002).
Turanek, J. et al. Liposomal formulation of alpha-tocopheryl maleamide: in vitro and in vivo toxicological profile and anticancer effect against spontaneous breast carcinomas in mice. Toxicol. Appl. Pharmacol. 237, 249–257 (2009).
Forsythe, J.A. et al. Activation of vascular endothelial growth factor gene transcription by hypoxia-inducible factor 1. Mol. Cell. Biol. 16, 4604–4613 (1996).
Schutyser, E., Struyf, S. & Van Damme, J. The CC chemokine CCL20 and its receptor CCR6. Cytokine Growth Factor Rev. 14, 409–426 (2003).
Terao, Y. et al. Macrophage inflammatory protein-3alpha plays a key role in the inflammatory cascade in rat focal cerebral ischemia. Neurosci. Res. 64, 75–82 (2009).
Jeong, S.I. et al. Toxicologic evaluation of bacterial synthesized cellulose in endothelial cells and animals. Mol. Cell. Toxicol. 6, 373–380 (2010).
Lee, S.H. et al. Identification of atherosclerosis related gene expression profiles by treatment of benzo(a) pyrene in human umbilical vein endothelial cells. Mol. Cell. Toxicol. 5, 113–119 (2009).
Lee, N.J., Lee, S.E., Lee, S.H., Ryu, D.S. & Park, Y.S. Acrolein induces adaptive response through upregulate of HO-1 via activation of Nrf2 in RAW 264.7 macrophage. Mol. Cell. Toxicol. 5, 230–236 (2009).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lee, S.E., Yang, H., Jeong, S.I. et al. Methylglyoxal-mediated alteration of gene expression in human endothelial cells. BioChip J 5, 220–228 (2011). https://doi.org/10.1007/s13206-011-5305-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13206-011-5305-y