Abstract
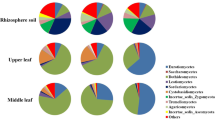
In this study, metagenomic sequencing technology was employed to analyze the ITS1 region sequence of the ITS rDNA gene of endophytic fungi and 16S sequence of endophytic bacteria in tea leaves with varying degrees of infection by tea blister blight disease as well as healthy tea leaves. Subsequently, a comparative analysis was conducted on the endophytic microbial diversity and the community structure in tea leaves. The findings of this investigation reveal a shift in the dominant endophytic fungal genera from Ascomycota to Basidiomycota as the disease progressed. Furthermore, a negative correlation was observed between Exobasidium and Talaromyce, with Talaromyce exhibiting potential as an antagonist against the disease. Meanwhile, our findings reveal that Proteobacteria, Firmicutes, and Actinobacteria were the three most abundant bacteria phyla in tea leaves. As the disease progressed, there was an increase in the relative abundance of Actinobacteria, while Variovorax, Sphingomonas, and Pseudomonas were found to have higher abundance in later stages. The diversity analysis results indicated that the endophytic microbial diversity and the community structure in tea leaves in the diseased group were lower than those in the healthy control group. In general, blister blight disease altered the community structure of endophytic microorganisms in tea leaves, resulting in a few species with high abundance. The study lays a foundation for investigating the pathogenic mechanism of tea blister disease and establishing a theoretical basis for controlling diseases in tea trees.







Similar content being viewed by others
References
Aghdam NMN, Baghaee-Ravari S, Shiri A (2022) Antimicrobial capacity of Pseudomonas brassicacearum strain EnPb against potato soft rot agent. Eur J Plant Pathol 165:215–231. https://doi.org/10.1007/s10658-022-02600-z
Bahramian D, Naraghi L, Heydari A (2016) Effectiveness of the chemical stabilizers of Talaromyces flavus in biological control of tomato and greenhouse cucumber vascular wilt disease. J Plant Prot Res 56(3):291–297. https://doi.org/10.1515/jppr-2016-0045
Barman A, Nath A, Thakur D (2020) Identification and characterization of fungi associated with blister blight lesions of tea (Camellia sinensis L. Microbiol Res, Kuntze) isolated from Meghalaya, India. https://doi.org/10.1016/j.micres.2020.126561
Biessy A, Novinscak A, St-Onge R, Léger G, Zboralski A, Filion M (2021) Inhibition of three potato pathogens by phenazine-producing Pseudomonas spp. is associated with multiple biocontrol-related traits. mSphere. https://doi.org/10.1128/mSphere.00427-21
Bokulich NA, Kaehler BD, Rideout JR, Dillon M, Bolyen E, Knight R, Huttley GA, Caporaso JG (2018) Optimizing taxonomic classification of marker-gene amplicon sequences with QIIME 2’s q2-feature-classifier plugin. Microbiome 6:90. https://doi.org/10.1186/s40168-018-0470-z
Bové JM (2006) Huanglongbing: A destructive, newly emerging, century-old disease of citrus. J Plant Pathol 88(1):7–37. https://www.jstor.org/stable/41998278
Chakraborty BN, Sharma M (2007) Serological detection and immunogold localization of cross-reactive antigens shared by Camellia sinensis and Exobasidium vexans. J Appl Microbiol 103:1669–1680
Chaliha C, Kalita E, Verma PK (2020) Optimizing in vitro culture conditions for the biotrophic fungi Exobasidium vexans through response surface methodology. Indian J Microbiol 60:167–174. https://doi.org/10.1007/s12088-019-00846-6
Chandra S, Chakraborty N, Panda K, Acharya K (2017) Chitosan-induced immunity in Camellia sinensis (L.) O. Kuntze against blister blight disease is mediated by nitric-oxide. Plant Physiol Biochem 115:298–307. https://doi.org/10.1016/j.plaphy.2017.04.008
Chao A (1984) Nonparametric estimation of the number of classes in a population. Scand J Stat 11(4): 265–270. https://www.jstor.org/stable/4615964
Chen YY, Zhou B, Li JL, Tang H, Cui YY, Wu WH, Liu JY, Tang JC (2023) Blister blight lesions of tea (Camellia sinensis L. Kuntze) leaves: Microbial diversity analysis and identification of the disease fungi. Chin Agric Sci Bull 39(6):116–123
Dethoup T, Klaram R, Pankaew T, Jantasorn A (2022) Impact of fungicides and plant extracts on biocontrol agents and side-effects of Trichoderma spp. on rice growth. Eur J Plant Pathol 164:567–582. https://doi.org/10.1007/s10658-022-02581-z
Di C, Han Z, Chai C, Sun J, Wu F, Pan K (2023) Improvement of Straw Changed Soil Microbial Flora Composition and Suppressed Chinese Cabbage (Brassica rapa L. ssp. pekinensis) Clubroot Disease. Agronomy. https://doi.org/10.3390/agronomy13071688
Dong LP, Rong ZZ, Yunzeng Z, Jianping X, Hongkai W, Zhengyi W, Hongye L (2022) The phyllosphere microbiome shifts toward combating melanose pathogen. Microbiome. https://doi.org/10.1186/s40168-022-01234-x
Finkel OM, Salas-González I, Castrillo G, Conway JM, Law TF, Teixeira PJPL, Wilson ED, Fitzpatrick CR, Jones CD, Dangl JL (2020) A single bacterial genus maintains root growth in a complex microbiome. Nature 587:103–108. https://doi.org/10.1038/s41586-020-2778-7
Gupta S, Chaturvedi P, Kulkarni MG, Staden JV (2020) A critical review on exploiting the pharmaceutical potential of plant endophytic fungi. Biotechnol Adv. https://doi.org/10.1016/j.biotechadv.2019.107462
Gustavsen JA, Pai S, Isserlin R, Demchak B, Pico AR (2019) RCy3: Network biology using cytoscape from within R. Research. https://doi.org/10.1101/793166
Hadian S, Supronienė S, Kulaitienė J, Hasanzadeh N (2023) Effect of epiphytic bacteria from citrus against green mold post-harvest diseases of citrus. Horticulturae 9(7):764. https://doi.org/10.3390/horticulturae9070764
Hazarika SN, Saikia K, Borah A, Thakur D (2021) Prospecting endophytic bacteria endowed with plant growth promoting potential isolated from Camellia sinensis. Front Microbiol. https://doi.org/10.3389/fmicb.2021.738058
Hong CE, Jo SH, Jo IH, Jeong H, Park JM (2017) Draft genome sequence of the endophytic bacterium Variovorax paradoxus KB5, which has antagonistic activity against a phytopathogen, Pseudomonas syringae pv. tomato DC3000. Gen Announc. https://doi.org/10.1128/genomea.00950-17
Huong NTM, Hoai PTT, Thao PTH, Huong TT, Chinh VD (2022) Growth stimulation, phosphate resolution, and resistance to fungal pathogens of some endogenous fungal strains in the rhizospheres of medicinal plants in vietnam. Molecules 27(16):5051. https://doi.org/10.3390/molecules27165051
Jayaswall K, Mahajan P, Singh G, Parmar R, Seth R, Raina A, Swarnkar MK, Singh AK, Sharma SR, RK, (2016) Transcriptome analysis reveals candidate genes involved in blister blight defense in tea (Camellia sinensis (L) Kuntze). Sci Rep 6:30412. https://doi.org/10.1038/srep30412
Jia HY, Xi ZG, Ma JQ, Li YF, Hao CH, LU MQ, Zhang ZZ, Deng WW, (2022) Endophytic bacteria from the leaves of two types of albino tea plants, indicating the plant growth promoting properties. Plant Growth Regul 96:331–343. https://doi.org/10.1007/s10725-021-00779-5
Kabir MH, Unban K, Kodchasee P, Govindarajan RK, Lumyong S, Suwannarach N, Wongputtisin P, Shetty K, Khanongnuch C (2023) Endophytic bacteria isolated from tea leaves (Camellia sinensis var. assamica) enhanced plant-growth-promoting activity. Agriculture. https://doi.org/10.3390/agriculture13030533
Kerr A, Rodrigo WRF (1967) Epidemiology of tea blister blight (Exobasidium vexans): IV Disease Forecasting. Trans Brit Mycol Soc 50(4):609–614. https://doi.org/10.1016/S0007-1536(67)80092-5
Liu SJ, Moon CD, Zheng N, Huws S, Zhao SG, Wang JQ (2022) Opportunities and challenges of using metagenomic data to bring uncultured microbes into cultivation. Microbiome 10:76. https://doi.org/10.1186/s40168-022-01272-5
Lozupone CA, Hamady M, Kelley ST, Knight R (2007) Quantitative and qualitative β diversity measures lead to different insights into factors that structure microbial communities. Appl Environ Microbiol 73(5):1576–1585. https://doi.org/10.1128/AEM.01996-06
Lu SL, Wang JM, Sheng RL, Fang YW, Guo RH (2020) Novel bioactive polyketides isolated from marine actinomycetes: An update review from 2013 to 2019. Chem Biodivers. https://doi.org/10.1002/cbdv.202000562
Lundberg D, Yourstone S, Mieczkowski P, Jones CD, Dangl JL (2013) Practical innovations for high-throughput amplicon sequencing. Nat Methods 10:999–1002. https://doi.org/10.1038/nmeth.2634
Lv NN, Tao CY, Ou YN, Wang JB, Deng XH, Liu HJ, Shen ZZ, Li R, Shen QR (2023) Root-associated antagonistic Pseudomonas spp. contribute to soil suppressiveness against banana Fusarium wilt disease of banana. Microbiol Spectr. https://doi.org/10.1128/spectrum.03525-22
Maki KA, Wolff B, Varuzza L, Green SJ, Barb JJ (2023) Multi-amplicon microbiome data analysis pipelines for mixed orientation sequences using QIIME2: Assessing reference database, variable region and pre-processing bias in classification of mock bacterial community samples. PLoS ONE 18(1):e0280293. https://doi.org/10.1371/journal.pone.0280293
Mu TC, Liu RY, Zhang XG, Li Z, Xia JW (2022) Diaporthe cha sp. nov on Camellia sinensis in China. Nova Hedwigia 115(3–4):473–485. https://doi.org/10.1127/nova_hedwigia/2022/0713
Martino C, Morton JT, Marotz CA, Thompson LR, Tripathi A, Knight R, Zengler K (2019) A novel sparse compositional technique reveals microbial perturbations. mSystems. https://doi.org/10.1128/msystems.00016-19
Niu S, Gao Y, Zi HX, Liu Y, Liu XM, Xiong XQ, Yao QQ, Qin ZW, Chen N, Guo L, Yang YZ, Qin P, Lin JZ, Zhu YH (2022) The osmolyte-producing endophyte Streptomyces albidoflavus OsiLf-2 induces drought and salt tolerance in rice via a multi-level mechanism. Crop J 10:375–386. https://doi.org/10.1016/j.cj.2021.06.008
Pandey AK, Sinniah GD, Babu A, Tanti A (2021) How the global tea industry copes with fungal diseases-challenges and opportunities. Plant Dis 105(7):1868–1879. https://doi.org/10.1094/PDIS-09-20-1945-FE
Premkumar R, Ponmurugan P, Manian S (2008) Growth and photosynthetic and biochemical responses of tea cultivars to blister blight infection. Photosynthetica 46(1):135–138. https://doi.org/10.1007/s11099-008-0021-0
Rachmad FD, Priyatmojo A, Widiastuti A (2022) Genetic diversity analysis of Exobasidium vexans causing tea blister blight in Wonosobo, Central Java, Indonesia using RAPD markers. Arch Phytopathol Plant Prot 55(10):1234–1249. https://doi.org/10.1080/03235408.2022.2086026
Rahman SFSA, Singh E, Pieterse CMJ, Schenk PM (2018) Emerging microbial biocontrol strategies for plant pathogens. Plant Sci 267:102–111. https://doi.org/10.1016/j.plantsci.2017.11.012
Ramette A (2007) Multivariate analyses in microbial ecology. FEMS Microbiol Ecol 62(2):142–160. https://doi.org/10.1111/j.1574-6941.2007.00375.x
Rateb ME, Ebel R, Jaspars M (2018) Natural product diversity of actinobacteria in the Atacama Desert. Antonie Van Leeuwenhoek 111:1467–1477. https://doi.org/10.1007/s10482-018-1030-z
Ruiz-Bedoya T, Wang PW, Desveaux D, Guttman DS (2023) Cooperative virulence via the collective action of secreted pathogen effectors. Nat Microbiol. https://doi.org/10.1038/s41564-023-01328-8
Sen S, Rai M, Das D, Chandra S, Acharya K (2020) Blister blight a threatened problem in tea industry: A review. J King Saud Univ Sci 32(8):3265–3272. https://doi.org/10.1016/j.jksus.2020.09.008
Shabani MH, Naraghi L, Maleki M, Negahban M (2024) Evaluation of the efficacy of different concentrations of nano-capsules containing Talaromyces flavus with two forms of powder and suspension in reducing the incidence of cotton Verticillium wilt. Braz J Biol. https://doi.org/10.1590/1519-6984.262480
Simpson EH (1949) Measurement of diversity. Nature. https://doi.org/10.1038/163688a0
Shannon CE (2001) A mathematical theory of communication. GetMobile-Mob Compu Commun Rev 27(1):23–27. https://doi.org/10.1145/3599184.3599192
Sharma P, Kumar S (2021) Bioremediation of heavy metals from industrial effluents by endophytes and their metabolic activity: Recent advances. Bioresource Technol 339:125589. https://doi.org/10.1016/j.biortech.2021.125589
Sun K, Lu F, Huang PW, Tang MJ, Xu FJ, Zhang W, Zhou JY, Zhao P, Jia Y, Dai CC (2022) Root endophyte differentially regulates plant response to NO3- and NH4+ nutrition by modulating N fluxes at the plant-fungal interface. Plant Cell Environ 45(6):1813–1828. https://doi.org/10.1111/pce.14304
Tamošiūnė I, Andriūnaitė E, Stanys V, Baniulis D (2019) Exploring diversity of bacterial endophyte communities using advanced sequencing technology. In: Kumar, V., Prasad, R., Kumar, M., Choudhary, D. (eds) Microbiome in Plant Health and Disease. Springer, Singapore. https://doi.org/10.1007/978-981-13-8495-0_20
Tibpromma S, Karunarathna SC, Bhat JD, Suwannarach N, Stephenson SL, Elgorban AM, Al-Rejaie S, Xu J, Mortimer PE (2022) Using culture-dependent and molecular techniques to identify endophytic fungi associated with tea leaves (Camellia spp) in Yunnan Province. Diversity, China. https://doi.org/10.3390/d14040287
Trivedi P, Leach JE, Tringe SG, Sa TM, Singh BK (2020) Plant-microbiome interactions: from community assembly to plant health. Nat Rev Microbiol 18:607–621. https://doi.org/10.1038/s41579-020-0412-1
Whittaker RH (1972) Evolution and measurement of species diversity. Taxon 21(2–3):213–251. https://doi.org/10.2307/1218190
Xin JJ, Liu Y, Li HY, Chen SM, Jiang JF, Song AP, Fang WM, Chen FD (2021) CmMLO17 and its partner CmKIC potentially support Alternaria alternata growth in Chrysanthemum morifolium. Hortic Res 8:101. https://doi.org/10.1038/s41438-021-00534-x
Xu Z, Song N, Song N, Ma L, Wu JS (2019) IRE1-bZIP60 pathway is required for Nicotiana attenuata resistance to fungal pathogen Alternaria alternata. Front Plant Sci 10:263. https://doi.org/10.3389/fpls.2019.00263
Yin QX, An XL, Wu X, Dharmasena DSP, Li DX, Jiang SL, Wang Y, Wang DL, Chen Z (2021) First report of Alternaria longipes causing leaf spot on tea in China. 105(12): 4167. https://doi.org/10.1094/PDIS-07-20-1583-PDN
Yu HR, Yan FF, Wang YL, Tong XY, Chen D, Ye Q, Piao RZ, Zhao HY (2022) Antagonistic effects of Sphingomonas and Pseudomonas aeruginosa on 4 kinds of pathogenic bacteria of Giaseng. Asian Agric Res 14(05):31–35. https://doi.org/10.19601/j.enki.issn1943-9903.2022.05.008
Zhang J, Wang ZB, Guo HW, Sun XL, Xiao Q (2023) Research progresses on blister blight in tea plant. Acta Phytopathol Sin. https://doi.org/10.13926/j.cnki.apps.001016(inChinese)
Zhou XL, Hu LH, Huy HN, Thanh TL, Zhou CB, Mei X, Buensanteai K (2023) The changes of metabolites, quality components and antioxidant activity of tea (Camellia sinensis L.) infected with Exobasidium vexans by applying UPLC-MS/MS based widely targeted metabolome and biochemical analysis. Phytopathology. https://doi.org/10.1094/PHYTO-03-23-0105-R
Funding
This work was supported by the Science and Technology Support Program of Guizhou, China (Nos. [2023]085 and [2020]1Y001) and the National Natural Science Foundation of China (No. 32160077).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Cao, R., Dong, X., Zhao, Y. et al. Effects of blister blight disease on endophytic microbial diversity and community structure in tea (Camellia sinensis) leaves. 3 Biotech 13, 421 (2023). https://doi.org/10.1007/s13205-023-03846-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13205-023-03846-y