Abstract
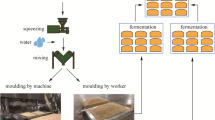
The microbial community structure and succession regularity of six key periods during high-temperature Daqu production were revealed using high-throughput sequencing to explore the factors affecting the flavor formation of Northern Jiang-flavored Baijiu technology. The results showed that among the six Daqu samples, the bacteria mainly included Firmicutes, Actinobacteriota, and Proteobacteria, of which Proteobacteria was the most dominant. The primary fungus was Ascomycota. At the genus level, the primary bacterial groups were Lactobacillus, Weissella, Bacillus, Delftia, Achromobacter, Saccharopolyspora, Thermoactinomyces, Scopulibacillus, Pseudomonas, and Stenotrophomonas. The main fungal groups in the Daqu were Wickerhamomyces, Saccharomycopsis, Thermoascus, and Thermomyces. During the initial stage of Daqu production, the dominant bacteria were Lactobacillus (20.07%) and Weissella (48.30%). As the fermentation temperature of the Daqu increased, Achromobacter, Stenotrophomonas, and Delftia became the dominant bacteria during the first Daqu flipping period, the second Daqu flipping period, and the dry-fire period. During these three periods, many bacteria were eliminated, decreasing the bacterial diversity, while a decline in temperature was evident during the Daqu exit period. After adapting to the high-temperature environment, the accumulation of Saccharopolyspora (22.07%), Thermoactinomyces (16.73%), Scopulibacillus (27.13%), Kroppenstedtia (9.03%), and Bacillus (6.97%) increased the bacterial diversity during the Daqu exit period. Wickerhamomyces (83.47%) represented the main dominant fungus during the initial production stage but were eliminated with increased temperature. Furthermore, a higher temperature increased the abundance of Saccharomycopsis and Thermoascus, while Thermomyces gradually accumulated in the D, E, and F samples. Thermomyces (79.90%) and Thermoascus (13.83%) became the dominant fungi during the Daqu exit period. In this study, high-throughput sequencing technology was used to reveal the microbial diversity during the high-temperature Daqu production process of Northern Jiang-flavored Baijiu. This provided a scientific basis for improving the production process of this product in the future. Therefore, understanding the formation of the flavor substances and the related microorganisms in Northern Jiang-flavored Baijiu can provide guidance for using them to manipulate the preparation process while implementing microbial control and improving the production procedures.






Similar content being viewed by others
Availability of data and materials
All data generated or analyzed during this study are included in this published article and its additional information files.
References
Bholay AD, Bhushan K, Rameez S, Gaur A, Malarvili G (2015) Novel chitinolytic potential of Achromobacter denitrificans isolated from fishery waste. IOSR JPBS 10(4):31–36. https://doi.org/10.9790/3008-10433136
Caporaso JG, Kuczynski J, Stombaugh J, Bittinger K, Bushman FD, Costello EK, Fierer N, Peña AG, Goodrich JK, Gordon JI, Huttley GA, Kelley ST, Knights D, Koenig JE, Ley RE, Lozupone CA, McDonald D, Muegge BD, Pirrung M, Reeder J, Sevinsky JR, Turnbaugh PJ, Walters WA, Widmann J, Yatsunenko T, Zaneveld J, Knight R (2010) QIIME allows analysis of high-throughput community sequencing data. Nat Methods 7(5):335–336. https://doi.org/10.1038/nmeth.f.303
Cole JR, Wang Q, Fish JA, Chai B, McGarrell DM, Sun Y, Brown CT, Porras-Alfaro A, Kuske CR, Tiedje JM (2014) Ribosomal database project: data and tools for high throughput rRNA analysis. Nucleic Acids Res 42(D1):D633–D642. https://doi.org/10.1093/nar/gkt1244
Fan G, Sun B, Fu Z, Xia Y, Li X (2018) Analysis of physicochemical indices, volatile flavor components, and microbial community of a light-flavor daqu. J Am Soc Brew Chem 76(3):209–218. https://doi.org/10.1080/03610470.2018.1424402
Gan SH, Yang F, Sahu SK, Luo RY, Liao SL, Wang HY, Jin T, Wang L, Zhang PF, Liu X, Xu J, Xu J, Wang YY, Liu H (2019) Deciphering the composition and functional profile of the microbial communities in Chinese Maotai liquor starters. Front Microbiol 10:1540. https://doi.org/10.3389/fmicb.2019.01540
Gou M, Wang HZ, Yuan HW, Zhang WX, Tang YQ, Kida K (2015) Characterization of the microbial community in three types of fermentation starters used for Chinese liquor production. J Inst Brew 121(4):620–627. https://doi.org/10.1002/jib.272
He G, Huang J, Zhou R, Wu C, Jin Y (2019) Effect of fortified Daqu on the microbial community and flavor in Chinese strong-flavor liquor brewing process. Front Microbiol 10:56. https://doi.org/10.3389/fmicb.2019.00056
Huang Y, Yi Z, Jin Y, Huang M, He K, Liu D, Luo H, Zhao D, He H, Fang Y, Zhao H (2017) Metatranscriptomics reveals the functions and enzyme profiles of the microbial community in Chinese Nong-flavor liquor starter. Front Microbiol 8:1747. https://doi.org/10.3389/fmicb.2017.01747
Huang X, Fan Y, Lu T, Kang J, Pang X, Han B, Chen J (2020) Composition and metabolic functions of the microbiome in fermented grain during light-flavor Baijiu fermentation. Microorganisms 8(9):1281. https://doi.org/10.3390/microorganisms8091281
Jin GY, Zhu Y, Xu Y (2017) Mystery behind Chinese liquor fermentation. Trends Food Sci Technol 63:18–28. https://doi.org/10.1016/j.tifs.2017.02.016
Klindworth A, Pruesse E, Schweer T, Peplies J, Quast C, Horn M, Glöckner FO (2013) Evaluation of general 16S ribosomal RNA gene PCR primers for classical and next-generation sequencing-based diversity studies. Nucleic Acids Res 41(1):e1. https://doi.org/10.1093/nar/gks808
Kozik AJ, Nakatsu CH, Chun H, Jones-Hall YL (2017) Age, sex, and TNF associated differences in the gut microbiota of mice and their impact on acute TNBS colitis. Exp Mol Pathol 103:311–319. https://doi.org/10.1016/j.yexmp.2017.11.014
Kuczynski J, Stombaugh J, Walters WA, González A, Caporaso JG, Knight R (2012) Using QIIME to analyze 16S rRNA gene sequences from microbial communities. Curr Protoc Microbiol Chapter 1: Unit 1E.5. https://doi.org/10.1002/9780471729259.mc01e05s27
Li Z, Bai Z, Wang D, Zhang W, Zhang M, Lin F, Gao L, Zhang H (2014) Cultivable bacterial diversity and amylase production in three typical Daqus of Chinese spirits. Int J Food Sci Technol 49(3):776–786. https://doi.org/10.1111/ijfs.12365
Liu P, Miao L (2020) Multiple batches of fermentation promote the formation of functional microbiota in Chinese miscellaneous-flavor Baijiu fermentation. Front Microbiol 11:75. https://doi.org/10.3389/fmicb.2020.00075
Liu H, Sun B (2018) Effect of fermentation processing on the flavor of Baijiu. J Agric Food Chem 66(22):5425–5432. https://doi.org/10.1021/acs.jafc.8b00692
Liu SP, Mao J, Liu YY, Meng XY, Ji ZW, Zhou ZL, Ai-lati A (2015) Bacterial succession and the dynamics of volatile compounds during the fermentation of Chinese rice wine from Shaoxing region. World J Microbiol Biotechnol 31(12):1907–1921. https://doi.org/10.1007/s11274-015-1931-1
Magoč T, Salzberg SL (2011) FLASH: fast length adjustment of short reads to improve genome assemblies. Bioinformatics 27(21):2957–2963. https://doi.org/10.1093/bioinformatics/btr507
McClendon SD, Batth T, Petzold CJ, Adams PD, Simmons BA, Singer SW (2012) Thermoascus aurantiacus is a promising source of enzymes for biomass deconstruction under thermophilic conditions. Biotechnol Biofuels 5:54. https://doi.org/10.1186/1754-6834-5-54
Pang XN, Han BZ, Huang XN, Zhang X, Hou LF, Cao M, Gao LJ, Hu GH, Chen JY (2018) Effect of the environment microbiota on the flavour of light-flavour Baijiu during spontaneous fermentation. Sci Rep 8(1):3396. https://doi.org/10.1038/s41598-018-21814-y
Patel RK, Jain M (2012) NGS QC Toolkit: a toolkit for quality control of next generation sequencing data. PLoS ONE 7(2):e30619. https://doi.org/10.1371/journal.pone.0030619
Perry BJ, Bergsveinson J, Tambalo DD, Yost CK, Khan NH, Whiting M (2017) Complete genome sequence of Delftia acidovorans RAY209, a plant growth-promoting rhizobacterium for canola and soybean. Genome Announc 5(44):e01224-e1317. https://doi.org/10.1128/genomeA.01224-17
Rognes T, Flouri T, Nichols B, Quince C, Mahé F (2016) VSEARCH: a versatile opensource tool for metagenomics. PeerJ 4:e2584. https://doi.org/10.7717/peerj.2584
Sánchez-Castro I, Ruiz-Fresneda MA, Bakkali M, Kämpfer P, Glaeser SP, Busse HJ, López-Fernández M, Martínez-Rodríguez P, Merroun ML (2017) Stenotrophomonas bentonitica sp. nov., isolated from bentonite formations. Int J Syst Evol Microbiol 67(8):2779–2786. https://doi.org/10.1099/ijsem.0.002016
Schloss PD, Westcott SL, Ryabin T, Hall JR, Hartmann M, Hollister EB, Lesniewski RA, Oakley BB, Parks DH, Robinson CJ, Sahl JW, Stres B, Thallinger GG, Van Horn DJ, Weber CF (2009) Introducing mothur: open-source, platform-independent, community-supported software for describing and comparing microbial communities. Appl Environ Microbiol 75(23):7537–7541. https://doi.org/10.1128/AEM.01541-09
Song Z, Du H, Zhang Y, Xu Y (2017) Unraveling core functional microbiota in traditional solid-state fermentation by high-throughput amplicons and metatranscriptomics sequencing. Front Microbiol 8:1294. https://doi.org/10.3389/fmicb.2017.01294
Usyk M, Zolnik CP, Patel H, Levi MH, Burk RD (2017) Novel ITS1 fungal primers for characterization of the mycobiome. mSphere 2(6):e00488-17. https://doi.org/10.1128/mSphere.00488-17
Wang H, Xu Y (2018) Microbial succession and metabolite changes during the fermentation of Chinese light aroma-style liquor. J Inst Brew 125(1):162–170. https://doi.org/10.1002/jib.544
Wang HY, Gao YB, Fan QW, Xu Y (2011) Characterization and comparison of microbial community of different typical Chinese liquor Daqus by PCR-DGGE. Lett Appl Microbiol 53(2):134–140. https://doi.org/10.1111/j.1472-765X.2011.03076.x
Wang L, Wang YY, Wang DQ, Xu J, Yang F, Liu G, Zhang DY, Feng Q, Xiao L, Xue WB, Guo J, Li YZ, Jin T (2015) Dynamic changes in the bacterial community in Maotai liquor fermentation process characterized by deep sequencing. J Inst Brew 121(4):603–608. https://doi.org/10.1002/jib.259
Wang Q, Zhang H, Liu X (2016) Microbial community composition associated with Maotai liquor fermentation. J Food Sci 81(6):M1485–M1494. https://doi.org/10.1111/1750-3841.13319
Wang P, Wu Q, Jiang X, Wang Z, Tang J, Xu Y (2017a) Bacillus licheniformis affects the microbial community and metabolic profile in the spontaneous fermentation of Daqu starter for Chinese liquor making. Int J Food Microbiol 250:59–67. https://doi.org/10.1016/j.ijfoodmicro.2017.03.010
Wang XD, Ban SD, Qiu SY (2017b) Analysis of the mould microbiome and exogenous enzyme production in Maotai-flavor Daqu. J Inst Brew 124(1):91–99. https://doi.org/10.1002/jib.467
Wang X, Du H, Zhang Y, Xu Y (2018) Environmental microbiota drives microbial succession and metabolic profiles during Chinese liquor fermentation. Appl Environ Microbiol 84(4):e02369-e2417. https://doi.org/10.1128/AEM.02369-17
Wang MY, Yang JG, Zhao QS, Zhang KZ, Su C (2019a) Research progress on flavor compounds and microorganisms of Maotai flavor Baijiu. J Food Sci 84(1):6–18. https://doi.org/10.1111/1750-3841.14409
Wang S, Wu Q, Nie Y, Wu J, Xu Y (2019b) Construction of synthetic microbiota for reproducible flavor compound metabolism in Chinese light-aroma-type liquor produced by solid-state fermentation. Appl Environ Microbiol 85(10):e03090-e3118. https://doi.org/10.1128/AEM.03090-18
Wei Y, Zou W, Shen CH, Yang JG (2020) Basic flavor types and component characteristics of Chinese traditional liquors: a review. J Food Sci 85(12):4096–4107. https://doi.org/10.1111/1750-3841.15536
Winger AM, Heazlewood JL, Chan LJ, Petzold CJ, Permaul K, Singh S (2014) Secetome analysis of the thermophilic xylanase hyper-producer Thermomyces lanuginosus SSBP cultivated on corn cobs. J Ind Microbiol Biotechnol 41(11):1687–1696. https://doi.org/10.1007/s10295-014-1509-1
Wu X, Jing R, Chen W, Geng X, Li M, Yang F, Yan Y, Liu Y (2020) High-throughput sequencing of the microbial diversity of roasted-sesame-like flavored Daqu with different characteristics. 3 Biotech 10(11):502. https://doi.org/10.1007/s13205-020-02500-1
Xiao C, Lu ZM, Zhang XJ, Wang ST, Ao L, Shen CH, Shi JS, Xu ZH (2017) Bio-heat is a key environmental driver shaping the microbial community of medium-temperature Daqu. Appl Environ Microbiol 83(23):e01550-e1617. https://doi.org/10.1128/AEM.01550-17
Xie MW, Lv FX, Ma GX, Asim F, Li HH, Du Y, Liu Y (2020) High throughput sequencing of the bacterial composition and dynamic succession in Daqu for Chinese sesame favour liquor. J Inst Brew 126:98–104. https://doi.org/10.1002/jib.592
Yan Z, Zheng X, Chen J, Han J, Han B (2013) Effect of different bacillus strains on the profile of organic acids in a liquid culture of Daqu. J Inst Brew 119(1–2):78–83. https://doi.org/10.1002/jib.58
Yao S, Liu Y, Li H, Ge YY, Zhang MJ, Xin Ci XuL, Cheng C (2015) Bacterial communities during the process of high-temperature Daqu production of roasted sesame-like flavour liquor. J Inst Brew 121(3):440–448. https://doi.org/10.1002/jib.235
Yi Z, Jin Y, Xiao Y, Chen L, Tan L, Du A, He K, Liu D, Luo H, Fang Y, Zhao H (2019) Unraveling the contribution of high temperature stage to Jiang-flavor Daqu, a liquor starter for production of Chinese Jiang-flavor Baijiu with special reference to metatranscriptomics. Front Microbiol 10:472. https://doi.org/10.3389/fmicb.2019.00472
You L, Wang S, Zhou R, Hu X, Chu Y, Wang T (2016) Characteristics of yeast flora in Chinese strong flavour spirit fermentation in Yibin region of China. J Inst Brew 122(7):517–523. https://doi.org/10.1002/jib.352
Yue JC, Clayton MK (2005) A similarity measure based on species proportions. Commun Stat 34:2123–2131. https://doi.org/10.1080/STA-200066418
Zhang Y, Zhu X, Li X, Tao Y, Jia J, He X (2017) The process-related dynamics of microbial community during a simulated fermentation of Chinese strong-flavored liquor. BMC Microbiol 17(1):196. https://doi.org/10.1186/s12866-017-1106-3
Zhang H, Wang L, Tan Y, Wang H, Yang F, Chen L, Hao F, Lv X, Du H, Xu Y (2021) Effect of Pichia on shaping the fermentation microbial community of sauce-flavor Baijiu. Int J Food Microbiol 336:108898. https://doi.org/10.1016/j.ijfoodmicro.2020.108898
Zhao X, Liu Y, Shu L, He Y (2019) Study on metabolites of Bacillus producing soy sauce-like aroma in Jiang-flavor Chinese spirits. Food Sci Nutr 8(1):97–103. https://doi.org/10.1002/fsn3.1266
Zou W, Ye G, Zhang K (2018a) Diversity, function, and application of Clostridium in Chinese strong flavor Baijiu ecosystem: a review. J Food Sci 83(5):1193–1199. https://doi.org/10.1111/1750-3841.14134
Zou W, Zhao C, Luo H (2018b) Diversity and function of microbial community in Chinese strong-flavor Baijiu ecosystem: a review. Front Microbiol 9:671. https://doi.org/10.3389/fmicb.2018.00671
Zuo QC, Huang YG, Guo M (2020) Evaluation of bacterial diversity during fermentation process: a comparison between handmade and machine-made high-temperature Daqu of Maotai-flavor liquor. Ann Microbiol 70:57. https://doi.org/10.1186/s13213-020-01598-1
Acknowledgements
I would like to thank the research team of Manager Pengpeng Xu at Nanjing Puwikon, Co., Ltd. for their technical support and assistance in high-throughput sequencing and bioinformatics analysis.
Funding
The research was supported by the Open Project Program of the Key Laboratory of Brewing Molecular Engineering of China Light Industry (BME-202001) and the Fundamental Research Funds for the Central Universities (No. FRF-TP-18-012A1; FRF-BR-19-003B).
Author information
Authors and Affiliations
Contributions
QEJ and XYW designed and participated in all experimental procedures, performed data analysis, and drafted the manuscript. YQX and ZSW participated in data analysis and draft writing. YLZ, LYS and WJY participated in the Daqu samples preparation and collection. JYS and YL supervised the study and critically revised the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Consent for publication
Not applicable.
Conflict of interest
The authors declare that they have no competing interests.
Ethics approval and consent to participate
Not applicable.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Jiang, Q., Wu, X., Xu, Y. et al. Microbial composition and dynamic succession during the Daqu production process of Northern Jiang-flavored liquor in China. 3 Biotech 11, 224 (2021). https://doi.org/10.1007/s13205-021-02779-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13205-021-02779-8