Abstract
The anti-aging protein Klotho has been associated with cardiovascular health protection. Nevertheless, the protective mechanism remains unknown. The present study is aimed at exploring the effect of Klotho on cardiac remodeling and its potential mechanism in mice with myocardial infarction (MI). We used left anterior coronary artery descending ligation to develop an MI model for in vivo analyses. In contrast, H9C2 cells and cardiac fibroblasts were used to establish the oxygen–glucose deprivation (OGD) model in in vitro analyses. In vivo and in vitro models were treated with Klotho. Compound C, an AMPK signaling inhibitor, was used to determine whether Klotho’s effects are mediated through the AMPK/mTOR signaling pathway. Echocardiography, Masson trichrome staining, immunofluorescence, immunohistochemistry, real-time polymerase chain reaction (RT-PCR), and western blot were used to detect the related indicators. The findings of the in vivo model indicate that Klotho treatment improved the mice’s cardiac function, reduced cardiac fibrosis, and attenuated myocardial inflammatory factors, ferroptosis, and oxidative stress. The results of the in vitro model were in line with the findings of in vivo modeling. An AMPK inhibitor, Compound C, reversed all these effects. In conclusion, Klotho potentially improves cardiac remodeling in MI mice by regulating AMPK/mTOR signaling, demonstrating Klotho as an effective MI therapeutic agent.






Similar content being viewed by others
Data Availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Wang K, Li Z, Sun Y et al (2021) Dapagliflozin improves cardiac function, remodeling, myocardial apoptosis, and inflammatory cytokines in mice with myocardial infarction. J Cardiovasc Transl Res, https://doi.org/10.1007/s12265-021-10192-y
Deng R, Liu Y, He H et al (2020) Haemin pre-treatment augments the cardiac protection of mesenchymal stem cells by inhibiting mitochondrial fission and improving survival. J Cell Mol Med 24:431–440. https://doi.org/10.1111/jcmm.14747
Talman V, Ruskoaho H (2016) Cardiac fibrosis in myocardial infarction-from repair and remodeling to regeneration. Cell Tissue Res 365:563–581. https://doi.org/10.1007/s00441-016-2431-9
Wang X, Guo Z, Ding Z et al (2018) Inflammation, autophagy, and apoptosis after myocardial infarction. J Am Heart Assoc 7: https://doi.org/10.1161/jaha.117.008024
Wu X, Li Y, Zhang S et al (2021) Ferroptosis as a novel therapeutic target for cardiovascular disease. Theranostics 11:3052–3059. https://doi.org/10.7150/thno.54113
Kurian GA, Rajagopal R, Vedantham S et al (2016) The role of oxidative stress in myocardial ischemia and reperfusion injury and remodeling: revisited. Oxid Med Cell Longev 2016:1656450. https://doi.org/10.1155/2016/1656450
Kuro-o M, Matsumura Y, Aizawa H et al (1997) Mutation of the mouse klotho gene leads to a syndrome resembling ageing. Nature 390:45–51. https://doi.org/10.1038/36285
Yamada S, Giachelli CM (2017) Vascular calcification in CKD-MBD: roles for phosphate, FGF23, and Klotho. Bone 100:87–93. https://doi.org/10.1016/j.bone.2016.11.012
Wang K, Mao Y, Lu M et al (2022) Association between serum klotho levels and the prevalence of diabetes among adults in the United States. Front Endocrinol (Lausanne) 13:1005553. https://doi.org/10.3389/fendo.2022.1005553
Chen K, Wang S, Sun QW et al (2021) Klotho deficiency causes heart aging via impairing the Nrf2-GR pathway. Circ Res 128:492–507. https://doi.org/10.1161/circresaha.120.317348
Guo Y, Zhuang X, Huang Z et al (2018) Klotho protects the heart from hyperglycemia-induced injury by inactivating ROS and NF-κB-mediated inflammation both in vitro and in vivo. Biochim Biophys Acta Mol Basis Dis 1864:238–251. https://doi.org/10.1016/j.bbadis.2017.09.029
Wang K, Zhongming LI, Yansong LI et al (2022) Cardioprotection of klotho against myocardial infarction-induced heart failure through inducing autophagy. Mech Ageing Dev. https://doi.org/10.1016/j.mad.2022.111714111714
Zhang X, Wang Q, Wang X et al (2019) Tanshinone IIA protects against heart failure post-myocardial infarction via AMPKs/mTOR-dependent autophagy pathway. Biomed Pharmacother 112:108599. https://doi.org/10.1016/j.biopha.2019.108599
Lu Q, Liu J, Li X et al (2020) Empagliflozin attenuates ischemia and reperfusion injury through LKB1/AMPK signaling pathway. Mol Cell Endocrinol 501:110642. https://doi.org/10.1016/j.mce.2019.110642
He Z, Zeng X, Zhou D et al (2021) lncRNA Chaer prevents cardiomyocyte apoptosis from acute myocardial infarction through AMPK activation. Front Pharmacol 12:649398. https://doi.org/10.3389/fphar.2021.649398
Yan J, Yan JY, Wang YX et al (2019) Spermidine-enhanced autophagic flux improves cardiac dysfunction following myocardial infarction by targeting the AMPK/mTOR signalling pathway. Br J Pharmacol 176:3126–3142. https://doi.org/10.1111/bph.14706
Ding J, Tang Q, Luo B et al (2019) Klotho inhibits angiotensin II-induced cardiac hypertrophy, fibrosis, and dysfunction in mice through suppression of transforming growth factor-β1 signaling pathway. Eur J Pharmacol 859:172549. https://doi.org/10.1016/j.ejphar.2019.172549
Wang F, Liu Y, Yuan J et al (2019) Compound C protects mice from HFD-induced obesity and nonalcoholic fatty liver disease. Int J Endocrinol 2019:3206587. https://doi.org/10.1155/2019/3206587
Benavides-Vallve C, Corbacho D, Iglesias-Garcia O et al (2012) New strategies for echocardiographic evaluation of left ventricular function in a mouse model of long-term myocardial infarction. PLoS One 7: e41691. https://doi.org/10.1371/journal.pone.0041691
Xu S, Mao Y, Wu J et al (2020) TGF-β/Smad and JAK/STAT pathways are involved in the anti-fibrotic effects of propylene glycol alginate sodium sulphate on hepatic fibrosis. J Cell Mol Med 24:5224–5237. https://doi.org/10.1111/jcmm.15175
Ge C, Zhao Y, Liang Y et al (2022) Silencing of TLR4 inhibits atrial fibrosis and susceptibility to atrial fibrillation via downregulation of NLRP3-TGF-β in spontaneously hypertensive rats. Dis Markers 2022:2466150. https://doi.org/10.1155/2022/2466150
Zhang Y, Liu S, Ding L et al (2021) Circ_0030235 knockdown protects H9c2 cells against OGD/R-induced injury via regulation of miR-526b. PeerJ 9:e11482. https://doi.org/10.7717/peerj.11482
Yang F, Qin Y, Wang Y et al (2019) Metformin inhibits the NLRP3 inflammasome via AMPK/mTOR-dependent effects in diabetic cardiomyopathy. Int J Biol Sci 15:1010–1019. https://doi.org/10.7150/ijbs.29680
Zhang M, Lei YS, Meng XW et al (2021) Iguratimod alleviates myocardial ischemia/reperfusion injury through inhibiting inflammatory response induced by cardiac fibroblast pyroptosis via COX2/NLRP3 signaling pathway. Front Cell Dev Biol 9:746317. https://doi.org/10.3389/fcell.2021.746317
Feng H, Mou SQ, Li WJ et al (2020) Resveratrol inhibits ischemia-induced myocardial senescence signals and NLRP3 inflammasome activation. Oxid Med Cell Longev 2020:2647807. https://doi.org/10.1155/2020/2647807
Xu C, Gao X, Wei Q et al (2018) Stem cell factor is selectively secreted by arterial endothelial cells in bone marrow. Nat Commun 9:2449. https://doi.org/10.1038/s41467-018-04726-3
Wang K, Li Z, Ma W et al (2021) Construction of miRNA-mRNA network reveals crucial miRNAs and genes in acute myocardial infarction. J Biomed Res 35:425–435. https://doi.org/10.7555/jbr.35.20210088
Zhang L, Cao M, Yu Z et al (2020) The restoration of Wnt/β-catenin signalling activity by a tuna backbone-derived peptide ameliorates hypoxia-induced cardiomyocyte injury. Am J Transl Res 12:5221–5236
Xu JP, Zeng RX, He MH et al (2022) Associations between serum soluble α-klotho and the prevalence of specific cardiovascular disease. Front Cardiovasc Med 9:899307. https://doi.org/10.3389/fcvm.2022.899307
Hongwei Y, Ruiping C, Yingyan F et al (2019) Effect of irbesartan on AGEs-RAGE and MMPs systems in rat type 2 diabetes myocardial-fibrosis model. Exp Biol Med (Maywood) 244:612–620. https://doi.org/10.1177/1535370219840981
Błyszczuk P, Zuppinger C, Costa A et al (2020) Activated cardiac fibroblasts control contraction of human fibrotic cardiac microtissues by a β-adrenoreceptor-dependent mechanism. Cells 9: https://doi.org/10.3390/cells9051270
Russo I, Frangogiannis NG (2016) Diabetes-associated cardiac fibrosis: cellular effectors, molecular mechanisms and therapeutic opportunities. J Mol Cell Cardiol 90:84–93. https://doi.org/10.1016/j.yjmcc.2015.12.011
Geelen T, Yeo SY, Paulis LE et al (2012) Internalization of paramagnetic phosphatidylserine-containing liposomes by macrophages. J Nanobiotechnol 10:37. https://doi.org/10.1186/1477-3155-10-37
Li A, Yu Y, Ding X et al (2020) miR-135b protects cardiomyocytes from infarction through restraining the NLRP3/caspase-1/IL-1β pathway. Int J Cardiol 307:137–145. https://doi.org/10.1016/j.ijcard.2019.09.055
Groot HE, Al Ali L, van der Horst ICC et al (2019) Plasma interleukin 6 levels are associated with cardiac function after ST-elevation myocardial infarction. Clin Res Cardiol 108:612–621. https://doi.org/10.1007/s00392-018-1387-z
Bradham WS, Moe G, Wendt KA et al (2002) TNF-alpha and myocardial matrix metalloproteinases in heart failure: relationship to LV remodeling. Am J Physiol Heart Circ Physiol 282:H1288-1295. https://doi.org/10.1152/ajpheart.00526.2001
Chen Q, Zhang D, Bi Y et al (2020) The protective effects of liguzinediol on congestive heart failure induced by myocardial infarction and its relative mechanism. Chin Med 15:63. https://doi.org/10.1186/s13020-020-00345-7
Chaar D, Dumont B, Vulesevic B et al (2021) Neutrophils pro-inflammatory and anti-inflammatory cytokine release in patients with heart failure and reduced ejection fraction. ESC Heart Fail 8:3855–3864. https://doi.org/10.1002/ehf2.13539
Li RL, Fan CH, Gong SY et al (2021) Effect and mechanism of LRP6 on cardiac myocyte ferroptosis in myocardial infarction. Oxid Med Cell Longev 2021:8963987. https://doi.org/10.1155/2021/8963987
Liu XJ, Lv YF, Cui WZ et al (2021) Icariin inhibits hypoxia/reoxygenation-induced ferroptosis of cardiomyocytes via regulation of the Nrf2/HO-1 signaling pathway. FEBS Open Bio 11:2966–2976. https://doi.org/10.1002/2211-5463.13276
Golej DL, Askari B, Kramer F et al (2011) Long-chain acyl-CoA synthetase 4 modulates prostaglandin E2 release from human arterial smooth muscle cells. J Lipid Res 52:782–793. https://doi.org/10.1194/jlr.M013292
Kagan VE, Mao G, Qu F et al (2017) Oxidized arachidonic and adrenic PEs navigate cells to ferroptosis. Nat Chem Biol 13:81–90. https://doi.org/10.1038/nchembio.2238
Xiang T, Luo X, Zeng C et al (2021) Klotho ameliorated cognitive deficits in a temporal lobe epilepsy rat model by inhibiting ferroptosis. Brain Res 1772:147668. https://doi.org/10.1016/j.brainres.2021.147668
Bugger H, Pfeil K (2020) Mitochondrial ROS in myocardial ischemia reperfusion and remodeling. Biochim Biophys Acta Mol Basis Dis 1866:165768. https://doi.org/10.1016/j.bbadis.2020.165768
Ma Z, Liu J, Li J et al (2020) Klotho ameliorates the onset and progression of cataract via suppressing oxidative stress and inflammation in the lens in streptozotocin-induced diabetic rats. Int Immunopharmacol 85:106582. https://doi.org/10.1016/j.intimp.2020.106582
Li MY, Zhu XL, Zhao BX et al (2019) Adrenomedullin alleviates the pyroptosis of Leydig cells by promoting autophagy via the ROS-AMPK-mTOR axis. Cell Death Dis 10:489. https://doi.org/10.1038/s41419-019-1728-5
Mihaylova MM, Shaw RJ (2011) The AMPK signalling pathway coordinates cell growth, autophagy and metabolism. Nat Cell Biol 13:1016–1023. https://doi.org/10.1038/ncb2329
Zhang J, Wang E, Zhang L et al (2021) PSPH induces cell autophagy and promotes cell proliferation and invasion in the hepatocellular carcinoma cell line Huh7 via the AMPK/mTOR/ULK1 signaling pathway. Cell Biol Int 45:305–319. https://doi.org/10.1002/cbin.11489
Ren PH, Zhang ZM, Wang P et al (2020) Yangxinkang tablet protects against cardiac dysfunction and remodelling after myocardial infarction in rats through inhibition of AMPK/mTOR-mediated autophagy. Pharm Biol 58:321–327. https://doi.org/10.1080/13880209.2020.1748662
Wang Z, Yao M, Jiang L et al (2022) Dexmedetomidine attenuates myocardial ischemia/reperfusion-induced ferroptosis via AMPK/GSK-3β/Nrf2 axis. Biomed Pharmacother 154:113572. https://doi.org/10.1016/j.biopha.2022.113572
Xue M, Yang F, Le Y et al (2021) Klotho protects against diabetic kidney disease via AMPK- and ERK-mediated autophagy. Acta Diabetol 58:1413–1423. https://doi.org/10.1007/s00592-021-01736-4
Funding
This work was supported by the National Natural Science Foundation of China (Grant No. 81871359 and No. 82071944).
Author information
Authors and Affiliations
Contributions
Kai WANG designed the study and completed the experiment together with Zhongming LI and Yinzhang DING. The first draft was written by Kai WANG, and the other authors participated in the revision and polishing of the draft. The authors declare that all data were generated in-house and that no paper mill was used.
Corresponding author
Ethics declarations
Ethics approval
The protocols for animal experiments followed NIH guidelines. Nanjing Medical University’s ethics committee has approved the study (IACUC-2102002). ARRIVE guidelines were followed in the design of animal experiments.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Keypoints.
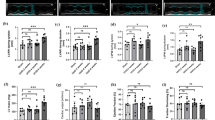
Klotho ameliorates cardiac function, reduces cardiac fibrosis, and attenuates myocardial inflammatory factors in mice with myocardial infarction.
Klotho improves ferroptosis and oxidative stress in mice with myocardial infarction.
Klotho potentially improves cardiac remodeling in MI mice by regulating AMPK/mTOR signaling.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
WANG, K., LI, Z., DING, Y. et al. Klotho improves cardiac fibrosis, inflammatory cytokines, ferroptosis, and oxidative stress in mice with myocardial infarction. J Physiol Biochem 79, 341–353 (2023). https://doi.org/10.1007/s13105-023-00945-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13105-023-00945-5