Abstract
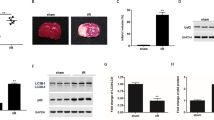
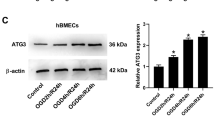
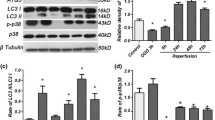
Autophagy has been described to be both protective and pathogenic in cerebral ischemia/reperfusion (I/R) injury. The underlying association between autophagy and ferroptosis in ischemic stroke has not yet been clearly investigated. The purpose of this study was to explore the role of autophagy-related gene 5 (ATG5) in experimental ischemic stroke. After injection of ATG5 shRNA lentivirus, mice underwent surgery for transient middle cerebral artery occlusion (MCAO)-induced focal cerebral ischemia. The infarct volume, neurological function, apoptosis, reactive oxygen species (ROS), autophagy, and ferroptosis levels were evaluated. After MCAO, ATG5-knockdown mice had a smaller infarct size and fewer neurological deficits than wild-type mice. The levels of apoptosis and ROS in ischemic mouse brains were alleviated through ATG5 knockdown. The expression of LC3 I/II was reduced through ATG5 knockdown after MCAO. Additionally, the expression of beclin1 and LC3 II was increased after I/R, but the increase was counteracted by preconditioning with ATG5 knockdown. After ischemic stroke, the levels of Fe2+ and malondialdehyde (MDA) were increased, but they were reduced by ATG5 knockdown. Similarly, the expression of glutathione peroxidase 4 (GPX4) and glutathione (GSH) was decreased by I/R but elevated by ATG5 knockdown. The present study shows that ATG5 knockdown attenuates autophagy-induced ferroptosis, which may offer a novel potential approach for ischemic stroke treatment.






Similar content being viewed by others
Data Availability
The data presented in the study are included in the article/Supplementary Material, and further inquiries can be directed to the corresponding authors.
References
Yang S, Wang H, Yang Y, Wang R, Wang Y, Wu C, et al. Baicalein administered in the subacute phase ameliorates ischemia-reperfusion-induced brain injury by reducing neuroinflammation and neuronal damage. Biomed Pharmacother. 2019;117:109102. https://doi.org/10.1016/j.biopha.2019.109102.
Sommer CJ. Ischemic stroke: experimental models and reality. Acta Neuropathol. 2017;133:245–61. https://doi.org/10.1007/s00401-017-1667-0.
Graham SH, Liu H. Life and death in the trash heap: the ubiquitin proteasome pathway and UCHL1 in brain aging, neurodegenerative disease and cerebral Ischemia. Ageing Res Rev. 2017;34:30–8. https://doi.org/10.1016/j.arr.2016.09.011.
Xie G, Yang S, Chen A, Lan L, Lin Z, Gao Y, et al. Electroacupuncture at Quchi and Zusanli treats cerebral ischemia-reperfusion injury through activation of ERK signaling. Exp Ther Med. 2013;5:1593–7. https://doi.org/10.3892/etm.2013.1030.
Wang MM, Zhang M, Feng YS, Xing Y, Tan ZX, Li WB, et al. Electroacupuncture inhibits neuronal autophagy and apoptosis via the PI3K/AKT pathway following ischemic stroke. Front Cell Neurosci. 2020;14:134. https://doi.org/10.3389/fncel.2020.00134.
Choi AM, Ryter SW, Levine B. Autophagy in human health and disease. N Engl J Med. 2013;368:651–62. https://doi.org/10.1056/NEJMra1205406.
Zhang X, Yan H, Yuan Y, Gao J, Shen Z, Cheng Y, et al. Cerebral ischemia-reperfusion-induced autophagy protects against neuronal injury by mitochondrial clearance. Autophagy. 2013;9:1321–33. https://doi.org/10.4161/auto.25132.
Luo C, Ouyang MW, Fang YY, Li SJ, Zhou Q, Fan J, et al. Dexmedetomidine protects mouse brain from ischemia-reperfusion injury via inhibiting neuronal autophagy through up-regulating HIF-1α. Front Cell Neurosci. 2017;11:197. https://doi.org/10.3389/fncel.2017.00197.
Zhang DM, Zhang T, Wang MM, Wang XX, Qin YY, Wu J, et al. TIGAR alleviates ischemia/reperfusion-induced autophagy and ischemic brain injury. Free Radic Biol Med. 2019;137:13–23. https://doi.org/10.1016/j.freeradbiomed.2019.04.002.
Wang L, Xiong X, Zhang X, Ye Y, Jian Z, Gao W, et al. Sodium tanshinone IIA sulfonate protects against cerebral ischemia-reperfusion injury by inhibiting autophagy and inflammation. Neuroscience. 2020;441:46–57. https://doi.org/10.1016/j.neuroscience.2020.05.054.
Zhang M, Qiu H, Mao L, Wang B, Li N, Fan Y, et al. Ammonium tetrathiomolybdate triggers autophagy-dependent NRF2 activation in vascular endothelial cells. Cell Death Dis. 2022;13:733. https://doi.org/10.1038/s41419-022-05183-z.
Gao M, Monian P, Pan Q, Zhang W, Xiang J, Jiang X. Ferroptosis is an autophagic cell death process. Cell Res. 2016;26:1021–32. https://doi.org/10.1038/cr.2016.95.
Hou W, Xie Y, Song X, Sun X, Lotze MT, Zeh HJ 3rd, et al. Autophagy promotes ferroptosis by degradation of ferritin. Autophagy. 2016;12:1425–8. https://doi.org/10.1080/15548627.2016.1187366.
Zhou B, Liu J, Kang R, Klionsky DJ, Kroemer G, Tang D. Ferroptosis is a type of autophagy-dependent cell death. Semin Cancer Biol. 2020;66:89–100. https://doi.org/10.1016/j.semcancer.2019.03.002.
Hirschhorn T, Stockwell BR. The development of the concept of ferroptosis. Free Radic Biol Med. 2019;133:130–43. https://doi.org/10.1016/j.freeradbiomed.2018.09.043.
Tuo QZ, Lei P, Jackman KA, Li XL, Xiong H, Li XL, et al. Tau-mediated iron export prevents ferroptotic damage after ischemic stroke. Mol Psychiatry. 2017;22:1520–30. https://doi.org/10.1038/mp.2017.171.
Alim I, Caulfield JT, Chen Y, Swarup V, Geschwind DH, Ivanova E, et al. Selenium drives a transcriptional adaptive program to block ferroptosis and treat stroke. Cell. 2019;177:1262-1279.e25. https://doi.org/10.1016/j.cell.2019.03.032.
Liang T, Qiang T, Ren L, Cheng F, Wang B, Li M, et al. Near-infrared fluorescent probe for hydrogen sulfide: high-fidelity ferroptosis evaluation in vivo during stroke. Chem Sci. 2022;13:2992–3001. https://doi.org/10.1039/d1sc05930k.
Liu J, Kuang F, Kroemer G, Klionsky DJ, Kang R, Tang D. Autophagy-dependent ferroptosis: machinery and regulation. Cell Chem Biol. 2020;27:420–35. https://doi.org/10.1016/j.chembiol.2020.02.005.
Kang R, Tang D. Autophagy and Ferroptosis - What’s the connection? Curr Pathobiol Rep. 2017;5:153–9. https://doi.org/10.1007/s40139-017-0139-5.
Liu J, Guo ZN, Yan XL, Huang S, Ren JX, Luo Y, et al. Crosstalk between autophagy and ferroptosis and its putative role in ischemic stroke. Front Cell Neurosci. 2020;14:577403. https://doi.org/10.3389/fncel.2020.577403.
Kang R, Zhu S, Zeh HJ, Klionsky DJ, Tang D. BECN1 is a new driver of ferroptosis. Autophagy. 2018;14:2173–5. https://doi.org/10.1080/15548627.2018.1513758.
Bai Y, Meng L, Han L, Jia Y, Zhao Y, Gao H, et al. Lipid storage and lipophagy regulates ferroptosis. Biochem Biophys Res Commun. 2019;508:997–1003. https://doi.org/10.1016/j.bbrc.2018.12.039.
Zhou L, Li F, Xu HB, Luo CX, Wu HY, Zhu MM, et al. Treatment of cerebral ischemia by disrupting ischemia-induced interaction of nNOS with PSD-95. Nat Med. 2010;16:1439–43. https://doi.org/10.1038/nm.2245.
Guo Z, Cao G, Yang H, Zhou H, Li L, Cao Z, et al. A combination of four active compounds alleviates cerebral ischemia-reperfusion injury in correlation with inhibition of autophagy and modulation of AMPK/mTOR and JNK pathways. J Neurosci Res. 2014;92:1295–306. https://doi.org/10.1002/jnr.23400.
Zhu H, Jian Z, Zhong Y, Ye Y, Zhang Y, Hu X, et al. Janus kinase inhibition ameliorates ischemic stroke injury and neuroinflammation through reducing NLRP3 inflammasome activation via JAK2/STAT3 pathway inhibition. Front Immunol. 2021;12:714943. https://doi.org/10.3389/fimmu.2021.714943.
Li M, Gao WW, Liu L, Gao Y, Wang YF, Zhao B, et al. The Akt/glycogen synthase kinase-3β pathway participates in the neuroprotective effect of interleukin-4 against cerebral ischemia/reperfusion injury. Neural Regen Res. 2020;15:1716–23. https://doi.org/10.4103/1673-5374.276343.
Xiong X, Ye Y, Gao X, Zhu H, Hu W, Li C, et al. An ultrasensitive fluorescent platform for monitoring GSH variation during ischemic stroke. Chem Eng J. 2022;450:137931. https://doi.org/10.1016/j.cej.2022.137931.
Li R, Liang T, Chen Z, Zhang S, Lin X, Huang R. L-dopa methyl ester attenuates amblyopia-induced neuronal injury in visual cortex of amblyopic cat. Gene. 2013;527:115–22. https://doi.org/10.1016/j.gene.2013.05.072.
Zhong Y, Gu L, Ye Y, Zhu H, Pu B, Wang J, et al. JAK2/STAT3 axis intermediates microglia/macrophage polarization during cerebral ischemia/reperfusion injury. Neuroscience. 2022;496:119–28. https://doi.org/10.1016/j.neuroscience.2022.05.016.
Xu Q, Ye Y, Wang Z, Zhu H, Li Y, Wang J, et al. NLRP3 knockout protects against lung injury induced by cerebral ischemia-reperfusion. Oxid Med Cell Longev. 2022;2022:6260102. https://doi.org/10.1155/2022/6260102.
Li Y, Zhu H, Wang X, Cui Y, Gu L, Hou X, et al. Small-molecule fluorophores for near-infrared iib imaging and image-guided therapy of vascular diseases. CCS Chem 2022;0:1–16 https://doi.org/10.31635/ccschem.022.202101547
Yingze Y, Zhihong J, Tong J, Yina L, Zhi Z, Xu Z, et al. NOX2-mediated reactive oxygen species are double-edged swords in focal cerebral ischemia in mice. J Neuroinflammation. 2022;19:184. https://doi.org/10.1186/s12974-022-02551-6.
Han B, Jiang W, Liu H, Wang J, Zheng K, Cui P, et al. Upregulation of neuronal PGC-1α ameliorates cognitive impairment induced by chronic cerebral hypoperfusion. Theranostics. 2020;10:2832–48. https://doi.org/10.7150/thno.37119.
Wu J, Dou Y, Liu W, Zhao Y, Liu X. Osteocalcin improves outcome after acute ischemic stroke. Aging (Albany NY). 2020;12:387–96. https://doi.org/10.18632/aging.102629.
Wang P, Shao BZ, Deng Z, Chen S, Yue Z, Miao CY. Autophagy in ischemic stroke. Prog Neurobiol. 2018;163–164:98–117. https://doi.org/10.1016/j.pneurobio.2018.01.001.
Wang P, Guan YF, Du H, Zhai QW, Su DF, Miao CY. Induction of autophagy contributes to the neuroprotection of nicotinamide phosphoribosyltransferase in cerebral ischemia. Autophagy. 2012;8:77–87. https://doi.org/10.4161/auto.8.1.18274.
Wang P, Xu TY, Wei K, Guan YF, Wang X, Xu H, et al. ARRB1/β-arrestin-1 mediates neuroprotection through coordination of BECN1-dependent autophagy in cerebral ischemia. Autophagy. 2014;10:1535–48. https://doi.org/10.4161/auto.29203.
Liu Y, Xue X, Zhang H, Che X, Luo J, Wang P, et al. Neuronal-targeted TFEB rescues dysfunction of the autophagy-lysosomal pathway and alleviates ischemic injury in permanent cerebral ischemia. Autophagy. 2019;15:493–509. https://doi.org/10.1080/15548627.2018.1531196.
Tian F, Deguchi K, Yamashita T, Ohta Y, Morimoto N, Shang J, et al. In vivo imaging of autophagy in a mouse stroke model. Autophagy. 2010;6:1107–14. https://doi.org/10.4161/auto.6.8.13427.
Garg AD, Dudek AM, Ferreira GB, Verfaillie T, Vandenabeele P, Krysko DV, et al. ROS-induced autophagy in cancer cells assists in evasion from determinants of immunogenic cell death. Autophagy. 2013;9:1292–307. https://doi.org/10.4161/auto.25399.
Acknowledgements
This work was supported by the National Natural Science Foundation of China (Nos. 81870939 and 82171336 to Xiaoxing Xiong; Nos. 82071339 and 82271370 to Lijuan Gu).
Author information
Authors and Affiliations
Contributions
Xiaoxing Xiong and Lijuan Gu designed this work. Hua Zhu and Yi Zhong wrote the manuscript. Hua Zhu, Ran Chen, Yuntao Li, Ran Chen, Lei Wang, Zhihong Jian, and Yi Zhong performed the experiments and analyzed the data. All authors contributed to manuscript revision and read and approved the submitted version.
Corresponding authors
Ethics declarations
Ethics Statement
The animal study was reviewed and approved by the Ethics Committee of Renmin Hospital of Wuhan University.
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhu, H., Zhong, Y., Chen, R. et al. ATG5 Knockdown Attenuates Ischemia‒Reperfusion Injury by Reducing Excessive Autophagy-Induced Ferroptosis. Transl. Stroke Res. 15, 153–164 (2024). https://doi.org/10.1007/s12975-022-01118-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12975-022-01118-0