Abstract
Neurite plasticity is a critical aspect of brain functional recovery after stroke. Emerging data suggest that Ras-related C3 botulinum toxin substrate 1 (Rac1) plays a central role in axonal regeneration in the injured brain, specifically by stimulating neuronal intrinsic growth and counteracting the growth inhibitory signaling that leads to growth cone collapse. Therefore, we investigated the functional role of Rac1 in axonal regeneration after stroke.
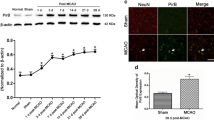
Delayed treatment with a specific Rac1 inhibitor, NSC 23766, worsened functional recovery, which was assessed by the pellet reaching test from day 14 to day 28 after stroke. It additionally reduced axonal density in the peri-infarct zone, assessed 28 days after stroke, with no effect on brain cavity size or on the number of newly formed cells. Accordingly, Rac1 overexpression using lentivirus promoted axonal regeneration and functional recovery after stroke from day 14 to day 28. Rac1 inhibition led to inactivation of pro-regenerative molecules, including mitogen-activated protein kinase kinase (p-MEK)1/2, LIM domain kinase (LIMK)1, and extracellular signal-regulated kinase (p-ERK)1/2 at 14 days after stroke. Inhibition of Rac1 reduced axonal length and number in cultured primary mouse cortical neurons using microfluidic chambers after oxygen-glucose deprivation (OGD) without affecting cell viability. In contrast, inhibition of Rac1 increased levels of glial fibrillary acidic protein, an extrinsic inhibitory signal for axonal growth, after stroke in vivo and in primary astrocytes after OGD.
In conclusion, Rac1 signaling enhances axonal regeneration and improve post-stroke functional recovery in experimental models of stroke.





Similar content being viewed by others
References
Liu L, McCullough L, Li J. Genetic deletion of calcium/calmodulin-dependent protein kinase kinase beta (CaMKK beta) or CaMK IV exacerbates stroke outcomes in ovariectomized (OVXed) female mice. BMC Neurosci. 2014;15:118. https://doi.org/10.1186/s12868-014-0118-2.
Benowitz LI, Carmichael ST. Promoting axonal rewiring to improve outcome after stroke. Neurobiol Dis. 2010;37(2):259–66. https://doi.org/10.1016/j.nbd.2009.11.009.
Lopez-Valdes HE, Clarkson AN, Ao Y, Charles AC, Carmichael ST, Sofroniew MV, et al. Memantine enhances recovery from stroke. Stroke. 2014;45(7):2093–100. https://doi.org/10.1161/STROKEAHA.113.004476.
Lorenzetto E, Ettorre M, Pontelli V, Bolomini-Vittori M, Bolognin S, Zorzan S, et al. Rac1 selective activation improves retina ganglion cell survival and regeneration. PLoS One. 2013;8(5):e64350. https://doi.org/10.1371/journal.pone.0064350.
Stankiewicz TR, Linseman DA. Rho family GTPases: key players in neuronal development, neuronal survival, and neurodegeneration. Front Cell Neurosci. 2014;8:314. https://doi.org/10.3389/fncel.2014.00314.
Posada-Duque RA, Velasquez-Carvajal D, Eckert GP, Cardona-Gomez GP. Atorvastatin requires geranylgeranyl transferase-I and Rac1 activation to exert neuronal protection and induce plasticity. Neurochem Int. 2013;62(4):433–45. https://doi.org/10.1016/j.neuint.2013.01.026.
Choi DH, Kim JH, Lee KH, Kim HY, Kim YS, Choi WS, et al. Role of neuronal NADPH oxidase 1 in the peri-infarct regions after stroke. PLoS One. 2015;10(1):e0116814. https://doi.org/10.1371/journal.pone.0116814.
Jiang X, Pu H, Hu X, Wei Z, Hong D, Zhang W, et al. A post-stroke therapeutic regimen with Omega-3 polyunsaturated fatty acids that promotes white matter integrity and beneficial microglial responses after cerebral ischemia. Transl Stroke Res. 2016;7(6):548–61. https://doi.org/10.1007/s12975-016-0502-6.
Pu H, Jiang X, Hu X, Xia J, Hong D, Zhang W, et al. Delayed docosahexaenoic acid treatment combined with dietary supplementation of Omega-3 fatty acids promotes long-term neurovascular restoration after ischemic stroke. Transl Stroke Res. 2016;7(6):521–34. https://doi.org/10.1007/s12975-016-0498-y.
Yang B, Parsha K, Schaar K, Xi X, Aronowski J, Savitz SI. Various cell populations within the mononuclear fraction of bone marrow contribute to the beneficial effects of autologous bone marrow cell therapy in a rodent stroke model. Transl Stroke Res. 2016;7(4):322–30. https://doi.org/10.1007/s12975-016-0462-x.
Manwani B, Liu F, Xu Y, Persky R, Li J, McCullough LD. Functional recovery in aging mice after experimental stroke. Brain Behav Immun. 2011;25(8):1689–700. https://doi.org/10.1016/j.bbi.2011.06.015.
Yuan H, Denton K, Liu L, Li XJ, Benashski S, McCullough L, et al. Nuclear translocation of histone deacetylase 4 induces neuronal death in stroke. Neurobiol Dis. 2016;91:182–93. https://doi.org/10.1016/j.nbd.2016.03.004.
Hou H, Chavez AE, Wang CC, Yang H, Gu H, Siddoway BA, et al. The Rac1 inhibitor NSC23766 suppresses CREB signaling by targeting NMDA receptor function. J Neurosci. 2014;34(42):14006–12. https://doi.org/10.1523/JNEUROSCI.1659-14.2014.
Veluthakal R, Sidarala V, Kowluru A. NSC23766, a known inhibitor of Tiam1-Rac1 signaling module, prevents the onset of type 1 diabetes in the NOD mouse model. Cell Physiol Biochem. 2016;39(2):760–7. https://doi.org/10.1159/000445666.
Akbar H, Cancelas J, Williams DA, Zheng J, Zheng Y. Rational design and applications of a Rac GTPase-specific small molecule inhibitor. Methods Enzymol. 2006;406:554–65. https://doi.org/10.1016/S0076-6879(06)06043-5.
Li J, Siegel M, Yuan M, Zeng Z, Finnucan L, Persky R, et al. Estrogen enhances neurogenesis and behavioral recovery after stroke. J Cereb Blood Flow Metab. 2011;31(2):413–25. https://doi.org/10.1038/jcbfm.2010.181.
Linden J, Fassotte L, Tirelli E, Plumier JC, Ferrara A. Assessment of behavioral flexibility after middle cerebral artery occlusion in mice. Behav Brain Res. 2014;258:127–37. https://doi.org/10.1016/j.bbr.2013.10.028.
Liu Z, Li Y, Cui Y, Roberts C, Lu M, Wilhelmsson U, et al. Beneficial effects of gfap/vimentin reactive astrocytes for axonal remodeling and motor behavioral recovery in mice after stroke. Glia. 2014;62(12):2022–33. https://doi.org/10.1002/glia.22723.
Alaverdashvili M, Whishaw IQ. A behavioral method for identifying recovery and compensation: hand use in a preclinical stroke model using the single pellet reaching task. Neurosci Biobehav Rev. 2013;37(5):950–67. https://doi.org/10.1016/j.neubiorev.2013.03.026.
Ueno Y, Chopp M, Zhang L, Buller B, Liu Z, Lehman NL, et al. Axonal outgrowth and dendritic plasticity in the cortical peri-infarct area after experimental stroke. Stroke. 2012;43(8):2221–8. https://doi.org/10.1161/STROKEAHA.111.646224.
Yu IC, Kuo PC, Yen JH, Paraiso HC, Curfman ET, Hong-Goka BC, et al. A combination of three repurposed drugs administered at reperfusion as a promising therapy for postischemic brain injury. Transl Stroke Res. 2017;8(6):560–77. https://doi.org/10.1007/s12975-017-0543-5.
Liu W, Wu J, Huang J, Zhuo P, Lin Y, Wang L, et al. Electroacupuncture regulates hippocampal synaptic plasticity via miR-134-mediated LIMK1 function in rats with ischemic stroke. Neural Plasticity. 2017;2017:9545646. https://doi.org/10.1155/2017/9545646.
Wang ZQ, Wu DC, Huang FP, Yang GY. Inhibition of MEK/ERK 1/2 pathway reduces pro-inflammatory cytokine interleukin-1 expression in focal cerebral ischemia. Brain Res. 2004;996(1):55–66. https://doi.org/10.1016/j.brainres.2003.09.074.
Liu HQ, Li WB, Li QJ, Zhang M, Sun XC, Feng RF, et al. Nitric oxide participates in the induction of brain ischemic tolerance via activating ERK1/2 signaling pathways. Neurochem Res. 2006;31(7):967–74. https://doi.org/10.1007/s11064-006-9102-2.
Fujita Y, Yamashita T. Axon growth inhibition by RhoA/ROCK in the central nervous system. Front Neurosci. 2014;8:338. https://doi.org/10.3389/fnins.2014.00338.
Di Giovanni S. Molecular targets for axon regeneration: focus on the intrinsic pathways. Expert Opin Ther Targets. 2009;13(12):1387–98. https://doi.org/10.1517/14728220903307517.
Hill JJ, Jin K, Mao XO, Xie L, Greenberg DA. Intracerebral chondroitinase ABC and heparan sulfate proteoglycan glypican improve outcome from chronic stroke in rats. Proc Natl Acad Sci U S A. 2012;109(23):9155–60. https://doi.org/10.1073/pnas.1205697109.
Jain A, McKeon RJ, Brady-Kalnay SM, Bellamkonda RV. Sustained delivery of activated rho GTPases and BDNF promotes axon growth in CSPG-rich regions following spinal cord injury. PLoS One. 2011;6(1):e16135. https://doi.org/10.1371/journal.pone.0016135.
Koth AP, Oliveira BR, Parfitt GM, Buonocore Jde Q, Barros DM. Participation of group I p21-activated kinases in neuroplasticity. J Physiol Paris. 2014;108(4–6):270–7. https://doi.org/10.1016/j.jphysparis.2014.08.007.
Stern S, Knoll B. CNS axon regeneration inhibitors stimulate an immediate early gene response via MAP kinase-SRF signaling. Mol Brain. 2014;7(1):86. https://doi.org/10.1186/s13041-014-0086-6.
Ciccarelli A, Giustetto M. Role of ERK signaling in activity-dependent modifications of histone proteins. Neuropharmacology. 2014;80:34–44. https://doi.org/10.1016/j.neuropharm.2014.01.039.
Wiegert JS, Bading H. Activity-dependent calcium signaling and ERK-MAP kinases in neurons: a link to structural plasticity of the nucleus and gene transcription regulation. Cell Calcium. 2011;49(5):296–305. https://doi.org/10.1016/j.ceca.2010.11.009.
Liao J, Ye Z, Huang G, Xu C, Guo Q, Wang E. Delayed treatment with NSC23766 in streptozotocin-induced diabetic rats ameliorates post-ischemic neuronal apoptosis through suppression of mitochondrial p53 translocation. Neuropharmacology. 2014;85:508–16. https://doi.org/10.1016/j.neuropharm.2014.06.008.
Liu F, Schafer DP, McCullough LD. TTC, fluoro-jade B and NeuN staining confirm evolving phases of infarction induced by middle cerebral artery occlusion. J Neurosci Methods. 2009;179(1):1–8. https://doi.org/10.1016/j.jneumeth.2008.12.028.
Silver J, Miller JH. Regeneration beyond the glial scar. Nat Rev Neurosci. 2004;5(2):146–56. https://doi.org/10.1038/nrn1326.
Kuijk LM, Beekman JM, Koster J, Waterham HR, Frenkel J, Coffer PJ. HMG-CoA reductase inhibition induces IL-1beta release through Rac1/PI3K/PKB-dependent caspase-1 activation. Blood. 2008;112(9):3563–73. https://doi.org/10.1182/blood-2008-03-144667.
Hwaiz R, Hasan Z, Rahman M, Zhang S, Palani K, Syk I, et al. Rac1 signaling regulates sepsis-induced pathologic inflammation in the lung via attenuation of mac-1 expression and CXC chemokine formation. J Surg Res. 2013;183(2):798–807. https://doi.org/10.1016/j.jss.2013.02.045.
Taglieri DM, Ushio-Fukai M, Monasky MM. P21-activated kinase in inflammatory and cardiovascular disease. Cell Signal. 2014;26(9):2060–9. https://doi.org/10.1016/j.cellsig.2014.04.020.
Acknowledgements
This project was generously supported by National Institutes of Health grants R01 NS099628 (J.Li) and R01 NS078446 (J.Li). This project was generously supported by National Institutes of Health grants R01 NS099628 (J.Li) and R01 NS078446 (J.Li).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Ethical Approval
All procedures were approved by the Center for Laboratory Animal Care of the University of Connecticut Health Center and University of Texas Health Science Center and performed following the National Institutes of Health Guide for the Care and Use of Laboratory Animals.
Rights and permissions
About this article
Cite this article
Liu, L., Yuan, H., Yi, Y. et al. Ras-Related C3 Botulinum Toxin Substrate 1 Promotes Axonal Regeneration after Stroke in Mice. Transl. Stroke Res. 9, 506–514 (2018). https://doi.org/10.1007/s12975-018-0611-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12975-018-0611-5