Abstract
Background and Aims
In chronic pancreatitis, fully covered self-expanding metal stents (FCSEMS) are used to treat refractory pancreatic duct strictures. However, the FCSEMS design, effectiveness, safety, optimal stent indwelling time and patient selection remain unclear. This study aimed at evaluating technical success, clinical success and adverse events with FCSEMS in patients with symptomatic pancreatic duct stricture.
Methods
The prospective study was conducted between May 2017 and May 2021 at a tertiary care center for chronic pancreatitis with refractory pancreatic duct stricture using controlled radial expansion (CRE) endoscopic retrograde cholangiopancreatography (ERCP) with FCSEMS (Niti-S, Bumpy stent, Taewoong Medical, Gimpo-Si, South Korea).
Results
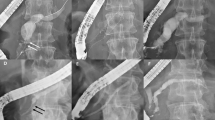
During the study period, a total of 11 patients underwent ERCP with FCSEMS for refractory pancreatic duct stricture. The mean age (± standard deviation, [SD]) was 32.36 ± 10.98 years and nine patients (81%) were male. Technical and clinical success rates were 100% and 90.9%, respectively. All patients had a history of prior pancreatic endotherapy. The median (inter quartile range, [IQR]) stent indwell time was seven (6–10) months. The median visual analogue scale (VAS) pain score pre and post-FCSEMS was 8 (5–8) and 1 (0–2), respectively (p-value 0.003). Median (IQR) follow-up after stent removal was 48 (40–60) months. One patient (9%) developed de novo main pancreatic duct (MPD) stricture, which was asymptomatic. None of the patients had cholangitis, pancreatitis, perforation, proximal migration or stent fracture.
Conclusion
The FCSEMS treatment appears to be safe, feasible and possibly an effective option for patients who have not responded to endoscopic plastic stenting.
Graphical Abstract



Similar content being viewed by others
References
Löhr JM, Dominguez-Munoz E, Rosendahl J, et al. United European Gastroenterology evidence-based guidelines for the diagnosis and therapy of chronic pancreatitis (HaPanEU). United Eur Gastroenterol J. 2017;5:153–99.
Testoni PA. Endoscopic stenting in benign pancreatic diseases. JOP J Pancreas. 2007;8:141–50.
Delhaye M, Arvanitakis M, Bali M, Matos C, Devière J. Endoscopic therapy for chronic pancreatitis. Scand J Surg. 2005;94:143–53. https://doi.org/10.1177/145749690509400211.
Topazian M, Aslanian H, Andersen D. Outcome following endoscopic stenting of pancreatic duct strictures in chronic pancreatitis. J Clin Gastroenterol. 2005;39:908–11.
Seza K, Yamaguchi T, Ishihara T, et al. A long-term controlled trial of endoscopic pancreatic stenting for treatment of main pancreatic duct stricture in chronic pancreatitis. Hepatogastroenterology. 2011;58:2128–31.
Adler DG, Lichtenstein D, Baron TH, et al. The role of endoscopy in patients with chronic pancreatitis. Gastrointest Endosc. 2006;63:933–7.
Gabbrielli A, Pandolfi M, Mutignani M, et al. Efficacy of main pancreatic-duct endoscopic drainage in patients with chronic pancreatitis, continuous pain, and dilated duct. Gastrointest Endosc. 2005;61:576–81.
Grimm H, Meyer WH, Nam VC, Soehendra N. New modalities for treating chronic pancreatitis. Endoscopy. 1989;21:70–4.
Cremer M, Devière J, Delhaye M, Baize M, Vandermeeren A. Stenting in severe chronic pancreatitis: results of medium-term follow-up in seventy-six patients. Endoscopy. 1991;23:171–6.
Smits ME, Badiga SM, Rauws EA, Tytgat GN, Huibregtse K. Long-term results of pancreatic stents in chronic pancreatitis. Gastrointest Endosc. 1995;42:461–7.
Binmoeller KF, Jue P, Seifert H, Nam WC, Izbicki J, Soehendra N. Endoscopic pancreatic stent drainage in chronic pancreatitis and a dominant stricture: long-term results. Endoscopy. 1995;27:638–44.
Ponchon T, Bory RM, Hedelius F, et al. Endoscopic stenting for pain relief in chronic pancreatitis: results of a standardized protocol. Gastrointest Endosc. 1995;42:452–6.
Rösch T, Daniel S, Scholz M, et al. Endoscopic treatment of chronic pancreatitis: a multicenter study of 1000 patients with long-term follow-up. Endoscopy. 2002;34:765–71.
Delhaye M, Arvanitakis M, Verset G, Cremer M, Devière J. Long-term clinical outcome after endoscopic pancreatic ductal drainage for patients with painful chronic pancreatitis. Clin Gastroenterol Hepatol. 2004;2:1096–106.
Eleftherladis N, Dinu F, Delhaye M, et al. Long-term outcome after pancreatic stenting in severe chronic pancreatitis. Endoscopy. 2005;37:223–30.
Weber A, Schneider J, Neu B, et al. Endoscopic stent therapy for patients with chronic pancreatitis: results from a prospective follow-up study. Pancreas. 2007;34:287–94.
Dumonceau J-M, Delhaye M, Tringali A, et al. Endoscopic treatment of chronic pancreatitis: European Society of Gastrointestinal Endoscopy (ESGE) Guideline - Updated August 2018. Endoscopy. 2019;51:179–93.
Costamagna G, Bulajic M, Tringali A, et al. Multiple stenting of refractory pancreatic duct strictures in severe chronic pancreatitis: long-term results. Endoscopy. 2006;38:254–9.
Tringali A, Bove V, Vadalà di Prampero SF, et al. Long-term follow-up after multiple plastic stenting for refractory pancreatic duct strictures in chronic pancreatitis. Endoscopy. 2019;51:930–5.
Tringali A, Vadalà di Prampero SF, Landi R, et al. Fully covered self-expandable metal stents to dilate persistent pancreatic strictures in chronic pancreatitis: long-term follow-up from a prospective study. Gastrointest Endosc. 2018;88:939–46.
Shah BB, Rodge GA, Goenka U, Afzalpurkar S, Goenka MK. A prospective study of fully covered self-expandable metal stents for refractory benign pancreatic duct strictures. Clin Endosc. 2022;55:793–800.
Sharaiha RZ, Novikov A, Weaver K, et al. Fully covered self-expanding metal stents for refractory pancreatic duct strictures in symptomatic chronic pancreatitis. US experience Endosc Int Open. 2019;7:E1419-23.
Cotton PB, Eisen GM, Aabakken L, et al. A lexicon for endoscopic adverse events: report of an ASGE workshop. Gastrointest Endosc. 2010;71:446–54.
Eisendrath P, Devière J. Expandable metal stents for benign pancreatic duct obstruction. Gastrointest Endosc Clin N Am. 1999;9:547–54.
Park DH, Kim M-H, Moon S-H, Lee SS, Seo D-W, Lee S-K. Feasibility and safety of placement of a newly designed, fully covered self-expandable metal stent for refractory benign pancreatic ductal strictures: a pilot study (with video). Gastrointest Endosc. 2008;68:1182–9.
Moon S-H, Kim M-H, Park DH, et al. Modified fully covered self-expandable metal stents with antimigration features for benign pancreatic-duct strictures in advanced chronic pancreatitis, with a focus on the safety profile and reducing migration. Gastrointest Endosc. 2010;72:86–91.
Sauer B, Talreja J, Ellen K, Ku J, Shami VM, Kahaleh M. Temporary placement of a fully covered self-expandable metal stent in the pancreatic duct for management of symptomatic refractory chronic pancreatitis: preliminary data (with videos). Gastrointest Endosc. 2008;68:1173–8.
Shen Y, Liu M, Chen M, Li Y, Lu Y, Zou X. Covered metal stent or multiple plastic stents for refractory pancreatic ductal strictures in chronic pancreatitis: a systematic review. Pancreatology. 2014;14:87-90.
Giacino C, Grandval P, Laugier R. Fully covered self-expanding metal stents for refractory pancreatic duct strictures in chronic pancreatitis. Endoscopy. 2012;44:874–7.
Matsubara S, Sasahira N, Isayama H, et al. Prospective pilot study of fully covered self-expandable metal stents for refractory benign pancreatic duct strictures: long-term outcomes. Endosc Int Open. 2016;4:E1215–22
Korpela T, Udd M, Lindström O, Kylänpää L. Fully covered self-expanding metal stents for benign refractory pancreatic duct strictures in chronic pancreatitis. Scand J Gastroenterol. 2019;54:365–70.
Acknowledgements
We thank the endoscopy staff for their cooperation in the completion of the study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
PR, PK, AK, SA, KA, M, RVKK and PM declare that they have no conflict of interest. This is an independent study with independently drawn protocols and conclusions. It has not been authorized or sponsored by Boston Scientific Corporation or its Affiliates.
Ethics statement
The study was performed conforming to the Helsinki Declaration of 1975, as revised in 2000 and 2008 concerning human and animal rights, and the authors followed the policy concerning informed consent as shown on Springer.com.
Disclaimer
The authors are solely responsible for the data and the contents of the paper. In no way, the Honorary Editor-in-Chief, Editorial Board Members, the Indian Society of Gastroenterology or the printer/publishers are responsible for the results/findings and content of this article.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
12664_2023_1445_MOESM1_ESM.png
Supplementary Fig. 1. (PNG 94 KB) Box plot showing the significant decrease in VAS pain score between pre-and post-FCSEMS intervention. FCSEMS fully covered self-expandable metal stent, VAS visual analogue scale
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Rai, P., Kumar, P., Kumar, A. et al. Self-expanding metallic stent for refractory pancreatic duct stricture in chronic pancreatitis: A prospective follow-up study. Indian J Gastroenterol (2023). https://doi.org/10.1007/s12664-023-01445-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12664-023-01445-6