Abstract
Aim
MicroRNAs have been widely acknowledged as a diagnostic, prognostic, and/or therapeutic biomarker for the progression of OSCC, but the correlation of hsa-miR-101-5p and hsa-miR-155-3p is yet to be established with c-Fos in OSCC and OSMF.
Methodology
An observational study enrolled 40 patients divided into 2 groups: Group I—21 OSMF patients without malignant transformation, Group II—19 patients with locally advanced, large-operable, or metastatic OSCC, after applying inclusion and exclusion criteria. Both miRNAs were extracted and analyzed from the tissue sample excised from the involved site. The linear regression analysis of the expression of hsa-miR-155-3p, hsa-miR-101-5p, and levels of c-fos in OSMF and OSCC patients and its correlation for habits, age, and gender were evaluated.
Results
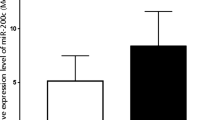
The expression of hsa-miR-101-5p was 0.81 times downregulated in OSCC tissue compared to OSMF, whereas hsa-miR-155-3p and c-fos were both upregulated 9.30 times and 1.75 times, respectively, in OSCC tissue. In Gutkha and tobacco chewers, the hsa-miR-155-3p expression could explain 12.3% (p = 0.031) for Gutkha chewers, whereas c-fos could explain 38.6% of the cases (p = 0.020) for tobacco chewers. The expression of hsa-miR-101-5p and hsa-miR-155-3p explained 43.7% and 59.5% of OSCC cases in alcoholics, respectively. Interestingly, in non-alcoholics, hsa-miR-155-3p and hsa-miR-101-5p were significant predictors of OSCC.
Conclusion
Downregulation of tumor-suppressor hsa-miR-101-5p and upregulation of proto-onco hsa-miR-155-3p is responsible for intricate regulation of the progression of OSMF to OSCC via deregulated expression of c-Fos and tobacco chewing and advancing age is significant contributors for OSCC.



Similar content being viewed by others
References
Bray F, Ferlay J, Soerjomataram I, Siegel RL et al (2018) Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. Ca Cancer J Clin 68:394–424
Chou H-C, Lin H-W, Yang J-H, Lin P-Y et al (2019) Clinical outcomes of oral cancer patients who survive for more than 5 years in Taiwan. J Formos Med Assoc 118(12):1616–1622
Phulari RGS, Dave EJ (2020) A systematic review on the mechanisms of malignant transformation of oral submucous fibrosis. Eur J Cancer Prev 29(5):470–473
Arora R, Adwani D, Naphade M, Bhagat B, Qureshi AQ (2014) Malignant conversion of oral submucous fibrosis in surgically treated case. J Clin Diagn Res 8(10):ZD31–ZD32
Ong TK et al (2017) Survival after surgery for oral cancer: a 30-year experience. Br J Oral Maxillofac Surg 55(9):911–916
Tsai WC, Kung PT, Wang ST, Hunag KW, Liu SA (2015) Beneficial impact of multidisciplinary team management on the survival in different stages of oral cavity cancer patients: results of a nationwide cohort study in Taiwan. Oral Oncol 51(2):105–111
Mathey-Andrews CA et al (2015) Small but mighty: microRNAs as novel signalling molecules in cancer. RNA Dis 2:e627. https://doi.org/10.14800/rd.627
Baba O, Hasegawa S, Nagai H et al (2016) MicroRNA-155-5p is associated with oral squamous cell carcinoma metastasis and poor prognosis. J Oral Pathol Med 45(4):248–255. https://doi.org/10.1111/jop.12351
Chakravarthi B, Goswami M, Pathi S et al (2016) MicroRNA-101 regulated transcriptional modulator SUB1 plays a role in prostate cancer. Oncogene 35:6330–6340. https://doi.org/10.1038/onc.2016.164
Wang J, Zeng H, Li H, Chen T, Wang L, Zhang K, Chen J, Wang R, Li Q, Wang S (2017) MicroRNA-101 inhibits growth, proliferation and migration and induces apoptosis of breast cancer cells by targeting sex-determining region Y-Box 2. Cell PhysiolBiochem 43:717–732. https://doi.org/10.1159/000481445
Iorio MV, Ferracin M, Liu C-G, Veronese A, Spizzo R, Sabbioni S, Magri E, Pedriali M, Fabbri M, Campiglio M, et al. (2005) MicroRNA gene expression deregulation in human breast cancer. Cancer Res 65:7065–7070. View Article : Google Scholar : PubMed/NCBI
Muhammad N, Bhattacharya S, Steele R, Phillips N, Ray RB (2017) Involvement of c-Fos in the promotion of cancer stem-like cell properties in head and neck squamous cell carcinoma. Clin Cancer Res 23(12):3120–3128. https://doi.org/10.1158/1078-0432.CCR-16-2811
Krishna A, Bhatt MLB, Singh V, et al. (2018) Differential expression of c-fos proto-oncogene in normal oral mucosa versus squamous cell carcinoma. Asian Pac J Cancer Prev 19(3):867–874. Published 2018 Mar 27. https://doi.org/10.22034/APJCP.2018.19.3.867
Dong C, Ye DX, Zhang WB, Pan HY, Zhang ZY, Zhan L (2015) Overexpression of c-fos promotes cell invasion and migration via CD44 pathway in oral squamous cell carcinoma. J Oral Pathol Med 44:353–360
Lessa AM, Valverde LF, Dias RB, Machado MCM, Santos JN, Rocha CAG (2013) The role of microRNAS in oral squamous cell carcinoma pathogenesis: a literature review. Appl Cancer Res 33(4):198–205
Buechner J, Tomte E, Haug BH, Henriksen JR, Lokke C, Flaegstad T, Einvik C (2011) Tumour-suppressor microRNAs let-7 and mir-101 target the proto-oncogene MYCN and inhibit cell proliferation in MYCN-amplified neuroblastoma. Br J Cancer 105:296–303
Bartel DP (2004) MicroRNAs: genomics, biogenesis, mechanism, and function. Cell 116:281–297. https://doi.org/10.1016/S0092-8674(04)00045-5
Calin GA, Croce CM (2006) MicroRNA signatures in human cancers. Nat Rev Cancer 6:857–866. https://doi.org/10.1038/nrc1997
He B, Lin X, Tian F, Yu W, Qiao B (2018) MiR-133a-3p inhibits oral squamous cell carcinoma (OSCC) proliferation and invasion by suppressing COL1A1. J Cell Biochem 119(1):338–346. https://doi.org/10.1002/jcb.26182
Gissi DB, Morandi L, Gabusi A, et al. (2018) A noninvasive test for MicroRNA expression in oral squamous cell carcinoma. Int J Mol Sci 19(6):1789. Published 2018 Jun 16. https://doi.org/10.3390/ijms19061789
Liu CJ, Kao SY, Tu HF, Tsai MM, Chang KW, Lin SC (2010) Increase of microRNA miR-31 level in plasma could be a potential marker of oral cancer. Oral Dis 16(4):360–364. https://doi.org/10.1111/j.1601-0825.2009.01646.x
Hi LJ, Zhang CY, Zhou ZT, Ma JY, Liu Y, Bao ZX, Jiang WW (2014) MicroRNA-155 in oral squamous cell carcinoma: overexpression, localization, and prognostic potential. Head Neck 37:970–976. https://doi.org/10.1002/hed.23700
Zhao XD, Zhang W, Liang HJ, Ji WY (2013) Overexpression of miR-155 promotes proliferation and invasion of human laryngeal squamous cell carcinoma via targeting SOCS1 and STAT3. PLoS ONE 8:e56395. https://doi.org/10.1371/journal.pone.0056395
Zargar S, Tomar V, Shyamsundar V, Vijayalakshmi R, Somasundaram K, Karunagaran D (2019) A feedback loop between microRNA 155 (miR-155), programmed cell death 4, and activation protein 1 modulates the expression of miR-155 and tumorigenesis in tongue cancer. Mol Cell Biol 39:e00410-e418
Yin Q, Wang X, McBride J, Fewell C, Flemington E (2008) B-cell receptor activation induces BIC/miR-155 expression through a con- served AP-1 element. J Biol Chem 283:2654–2662
Manikandan M, Deva Magendhra Rao AK, Arunkumar G, et al. (2016) Oral squamous cell carcinoma: microRNA expression profiling and integrative analyses for elucidation of tumourigenesis mechanism. Mol Cancer 15:28. Published 2016 Apr 7. https://doi.org/10.1186/s12943-016-0512-8
Loh HY, Norman BP, Lai KS, Rahman NMANA, Alitheen NBM, Osman MA (2019) The regulatory role of MicroRNAs in breast cancer. Int J Mol Sci 20(19):4940. Published 2019 Oct 6. https://doi.org/10.3390/ijms20194940
Wang HJ, Ruan HJ, He XJ, Ma YY, Jiang XT, Xia YJ, Ye ZY, Tao HQ (2010) MicroRNA-101 is down-regulated in gastric cancer and involved in cell migration and invasion. Eur J Cancer 46:2295–2303
Long Y, Wu Z, Yang X, Chen L, Han Z, Zhang Y, Liu J, Liu W, Liu X (2016) MicroRNA-101 inhibits the proliferation and invasion of bladder cancer cells via targeting c-FOS. Mol Med Rep 14:2651–2656
Liu J-J, Lin X-J, Yang X-J, Zhou L, He S, Zhuang S-M, Yang J (2014) A novel AP-1/miR-101 regulatory feedback loop and its implication in the migration and invasion of hepatoma cells. Nucleic Acids Res 42(19):12041–12051. https://doi.org/10.1093/nar/gku87
Acknowledgements
We would like to extend our thanks to our study participants for participating in the study. We also like to thank our parent Institute for funding this study.
Funding
This study received funding from the parent institute (Grant Number AIIMS/IEC/2016/238).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Parent Institute Where Research Was Conducted: All India Institute of Medical Sciences, Jodhpur, Rajasthan, INDIA
Rights and permissions
About this article
Cite this article
Chugh, A., Purohit, P., Vishnoi, J.R. et al. Correlation of hsa miR-101-5p and hsa miR-155-3p Expression With c-Fos in Patients of Oral Submucous Fibrosis (OSMF) and Oral Squamous Cell Carcinoma (OSCC). J. Maxillofac. Oral Surg. 22, 381–387 (2023). https://doi.org/10.1007/s12663-021-01668-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12663-021-01668-0