Abstract
Purpose
Glucagon-like peptide-1 (GLP-1) receptor agonists (GLP-1RAs) have become increasingly popular as both diabetic and weight loss therapies. One effect of this class of medication is delayed gastric emptying, which may impact the risk of aspiration during anesthesia delivery.
Methods
In this prospective study, we used gastric ultrasound to evaluate the presence of solid gastric contents in both supine and lateral positions after an eight-hour fast in those taking GLP-1RA compared with controls. Participants underwent a second ultrasound evaluation two hours later after drinking 12 fluid ounces of water (approximately 350 mL).
Results
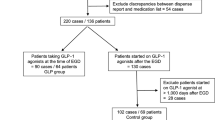
Twenty adults voluntarily enrolled, giving a total of ten participants in each group. In the supine position, 70% of semaglutide participants and 10% of control participants had solids present on gastric ultrasound (risk ratio [RR], 3.50; 95% confidence interval [CI], 1.26 to 9.65; P = 0.02.) In the lateral position, 90% of semaglutide participants and 20% of control participants had solids identified on gastric ultrasound (RR, 7.36; 95% CI, 1.13 to 47.7; P = 0.005). Two hours after drinking clear liquids, the two groups did not differ in the lateral position, but in the supine position, 90% of control group participants were rated as empty compared with only 30% of semaglutide group participants (P = 0.02).
Conclusions
This study provides preliminary evidence that GLP-1RAs may affect gastric emptying and residual gastric contents following an overnight fast and two hours after clear liquids, which may have implications for aspiration risk during anesthetic care.
Résumé
Objectif
Les agonistes des récepteurs du peptide-1 de type glucagon (GLP-1) (AR-GLP-1) sont de plus en plus populaires comme traitements du diabète et pour la perte de poids. L’un des effets de cette classe de médicaments est le retard de la vidange gastrique, ce qui peut avoir un impact sur le risque d’aspiration pendant l’administration de l’anesthésie.
Méthode
Dans cette étude prospective, nous avons utilisé l’échographie gastrique pour évaluer la présence de contenu gastrique solide en décubitus dorsal et latéral après un jeûne de huit heures chez les personnes prenant des AR-GLP-1 par rapport à un groupe témoin. Les participant·es ont passé une deuxième échographie deux heures plus tard, après avoir bu 12 onces liquides d’eau (environ 350 mL).
Résultats
Vingt personnes adultes se sont inscrites volontairement, soit un total de dix participant·es dans chaque groupe. En décubitus dorsal, 70 % des personnes du groupe sémaglutide et 10 % des personnes du groupe témoin présentaient des solides à l’échographie gastrique (risque relatif [RR], 3,50; intervalle de confiance [IC] à 95 %, 1,26 à 9,65; P = 0,02). En décubitus latéral, 90 % des personnes du groupe sémaglutide et 20 % des personnes du groupe témoin présentaient des solides à l’échographie gastrique (RR, 7,36; IC 95 %, 1,13 à 47,7; P = 0,005). Deux heures après avoir bu des liquides clairs, le contenu gastrique évalué ne différait pas entre les deux groupes en position latérale, mais en décubitus dorsal, l’estomac de 90 % des participant·es du groupe témoin a été jugé vide, comparativement à seulement 30 % chez les participant·es du groupe sémaglutide (P = 0,02).
Conclusion
Cette étude procure des données préliminaires selon lesquelles les AR-GLP-1 pourraient affecter la vidange gastrique et le contenu gastrique résiduel après un jeûne d’une nuit et deux heures après l’ingestion de liquides clairs, ce qui pourrait avoir des implications quant au risque d’aspiration pendant les soins anesthésiques.

Similar content being viewed by others
References
Kumar N, D’Alessio DA. Slow and steady wins the race: 25 years developing the GLP-1 receptor as an effective target for weight loss. J Clin Endocrinol Metab 2022; 107: 2148–53. https://doi.org/10.1210/clinem/dgac276
Novograd J, Mullally J, Frishman WH. Semaglutide for weight loss: was it worth the weight? Cardiol Rev 2022; 30: 324–9. https://doi.org/10.1097/crd.0000000000000430
Ard J, Fitch A, Fruh S, Herman L. Weight loss and maintenance related to the mechanism of action of glucagon-like peptide 1 receptor agonists. Adv Ther 2021; 38: 2821–39. https://doi.org/10.1007/s12325-021-01710-0
Friedrichsen M, Breitschaft A, Tadayon S, Wizert A, Skovgaard D. The effect of semaglutide 2.4 mg once weekly on energy intake, appetite, control of eating, and gastric emptying in adults with obesity. Diabetes Obes Metab 2020; 23: 754–62. https://doi.org/10.1111/dom.14280
Drucker DJ. Mechanisms of action and therapeutic application of glucagon-like peptide-1. Cell Metab 2018; 27: 740–56. https://doi.org/10.1016/j.cmet.2018.03.001
Aroda VR, Rosenstock J, Terauchi Y, et al. PIONEER 1: randomized clinical trial of the efficacy and safety of oral semaglutide monotherapy in comparison with placebo in patients with type 2 diabetes. Diabetes Care 2019; 42: 1724–32. https://doi.org/10.2337/dc19-0749
Andreadis P, Karagiannis T, Malandris K, et al. Semaglutide for type 2 diabetes mellitus: a systematic review and meta-analysis. Diabetes Obes Metab 2018; 20: 2255–63. https://doi.org/10.1111/dom.13361
Coveleskie K, Kilpatrick LA, Gupta A, et al. The effect of the GLP‐1 analogue exenatide on functional connectivity within an NTS‐based network in women with and without obesity. Obes Sci Pract 2017; 3: 434–45. https://doi.org/10.1002/osp4.124
Secher A, Jelsing J, Baquero AF, et al. The arcuate nucleus mediates GLP-1 receptor agonist liraglutide-dependent weight loss. J Clin Invest 2014; 124: 4473–88. https://doi.org/10.1172/jci75276
Friedrichsen M, Breitschaft A, Tadayon S, et al. The effect of semaglutide 2.4 mg once weekly on energy intake, appetite, control of eating, and gastric emptying in adults with obesity. Diabetes Obes Metab 2021; 23: 754–62. https://doi.org/10.1111/dom.14280
Van De Putte P, Perlas A. Ultrasound assessment of gastric content and volume. Br J Anaesth 2014; 113: 12–22. https://doi.org/10.1093/bja/aeu151
Dupont G, Gavory J, Lambert P, et al. Ultrasonographic gastric volume before unplanned surgery. Anaesthesia 2017; 72: 1112–6. https://doi.org/10.1111/anae.13963
El-Boghdadly K, Wojcikiewicz T, Perlas A. Perioperative point-of-care gastric ultrasound. BJA Educ 2019; 19: 219–26. https://doi.org/10.1016/j.bjae.2019.03.003
Kruisselbrink R, Gharapetian A, Chaparro LE, et al. Diagnostic accuracy of point-of-care gastric ultrasound. Anesth Analg 2019; 128: 89–94. https://doi.org/10.1213/ane.0000000000003372
Alakkad H, Kruisselbrink R, Chin KJ, et al. Point-of-care ultrasound defines gastric content and changes the anesthetic management of elective surgical patients who have not followed fasting instructions: a prospective case series. Can J Anesth 2015; 62: 1188–95. https://doi.org/10.1007/s12630-015-0449-1
Silveira SQ, da Silva LM, de Campos Vieira Abib A, et al. Relationship between perioperative semaglutide use and residual gastric content: a retrospective analysis of patients undergoing elective upper endoscopy. J Clin Anesth 2023; 87: 111091. https://doi.org/10.1016/j.jclinane.2023.111091
Klein SR, Hobai IA. Semaglutide, delayed gastric emptying, and intraoperative pulmonary aspiration: a case report. Can J Anesth 2023; https://doi.org/10.1007/s12630-023-02440-3
Perlas A, Van De Putte P, Van Houwe P, Chan VW. I-AIM framework for point-of-care gastric ultrasound. Br J Anaesth 2016; 116: 7–11. https://doi.org/10.1093/bja/aev113
Perlas A, Chan VW, Lupu CM, Mitsakakis N, Hanbidge A. Ultrasound assessment of gastric content and volume. Anesthesiology 2009; 111: 82–9. https://doi.org/10.1097/aln.0b013e3181a97250
Van De Putte P. Bedside gastric ultrasonography to guide anesthetic management in a nonfasted emergency patient. J Clin Anesth 2013 25: 165–6. https://doi.org/10.1016/j.jclinane.2012.10.005
Joshi GP, Abdelmalak BB, Weigel WA, et al. 2023 American Society of Anesthesiologists practice guidelines for preoperative fasting: carbohydrate-containing clear liquids with or without protein, chewing gum, and pediatric fasting duration—a modular update of the 2017 American Society of Anesthesiologists practice guidelines for preoperative fasting. Anesthesiology 2023; 138: 132–51. https://doi.org/10.1097/aln.0000000000004381
Van De Putte P, Vernieuwe L, Jerjir A, Verschueren L, Tacken M, Perlas A. When fasted is not empty: a retrospective cohort study of gastric content in fasted surgical patients†. Br J Anaesth 2017; 118: 363–71. https://doi.org/10.1093/bja/aew435
Sherwin M, Katz D. Using gastric ultrasound to assess gastric content in the pregnant patient. BJA Educ 2021; 21: 404–7. https://doi.org/10.1016/j.bjae.2021.08.001
Kruisselbrink R, Arzola C, Jackson T, Okrainec A, Chan V, Perlas A. Ultrasound assessment of gastric volume in severely obese individuals: a validation study. Br J Anaesth 2017; 118: 77–82. https://doi.org/10.1093/bja/aew400
Spencer AO, Walker AM, Yeung AK, et al. Ultrasound assessment of gastric volume in the fasted pediatric patient undergoing upper gastrointestinal endoscopy: development of a predictive model using endoscopically suctioned volumes. Paediatr Anaesth 2015; 25: 301–8. https://doi.org/10.1111/pan.12581
Van De Putte P, Perlas A. Gastric sonography in the severely obese surgical patient: a feasibility study. Anesth Analg 2014; 119: 1105–10. https://doi.org/10.1213/ane.0000000000000373
Khalil AM, Ragab SG, Botros JM, Abd-Aal HA, Boules ML. gastric residual volume assessment by gastric ultrasound in fasting obese patients: a comparative study. Anesth Pain Med 2021; 11: e109732. https://doi.org/10.5812/aapm.109732
Mahajan V, Hashmi J, Singh R, Samra T, Aneja S. Comparative evaluation of gastric pH and volume in morbidly obese and lean patients undergoing elective surgery and effect of aspiration prophylaxis. J Clin Anesth 2015; 27: 396–400. https://doi.org/10.1016/j.jclinane.2015.03.004
Wynn-Hebden A, Bouch DC. Anaesthesia for the obese patient. BJA Educ 2020; 20: 388–95. https://doi.org/10.1016/j.bjae.2020.07.003
Deng Y, Park A, Zhu L, Xie W, Pan cQ. Effect of semaglutide and liraglutide in individuals with obesity or overweight without diabetes: a systematic review. Ther Adv Chronic Dis 2022; 13: https://doi.org/10.1177/20406223221108064
Author contributions
Marc Sherwin contributed to study design and data analysis, patient recruitment, data collection, and article drafts and finalization. Joshua Hamburger contributed to study design, patient recruitment, data collection, and article drafts and finalization. Daniel Katz and Samuel DeMaria, Jr contributed to study design and data analysis, data collection and interpretation, and article drafts and finalization.
Disclosures
None of the authors report relevant conflicts.
Funding statement
No external funding was received to conduct this study.
Editorial responsibility
This submission was handled by Dr. Philip M. Jones, Deputy Editor-in-Chief, Canadian Journal of Anesthesia/Journal canadien d’anesthésie.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is accompanied by an Editorial. Please see Can J Anesth 2023; https://doi.org/10.1007/s12630-023-02550-y.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sherwin, M., Hamburger, J., Katz, D. et al. Influence of semaglutide use on the presence of residual gastric solids on gastric ultrasound: a prospective observational study in volunteers without obesity recently started on semaglutide. Can J Anesth/J Can Anesth 70, 1300–1306 (2023). https://doi.org/10.1007/s12630-023-02549-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12630-023-02549-5