Abstract
Purpose
Abrupt hemodynamic changes or life-threatening arrhythmias are frequently observed in individuals receiving anesthesia for cardiac arrhythmia ablation. Remimazolam is a novel ultra-short-acting benzodiazepine that has been associated with better hemodynamic stability than conventional anesthetic agents do. This study aimed to investigate whether remimazolam reduces vasoactive agent use compared with desflurane in individuals undergoing ablation for atrial fibrillation under general anesthesia.
Methods
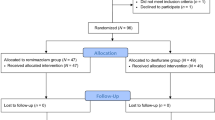
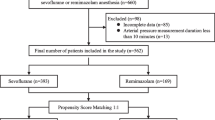
In a retrospective cohort study, we reviewed electronic medical records of adult patients who underwent ablation for atrial fibrillation under general anesthesia between July 2021 and July 2022. We divided patients into remimazolam and desflurane groups according to the principal agent used for anesthesia. The primary endpoint was the overall incidence of vasoactive agent use. We compared the groups using propensity-score matching (PSM) analysis.
Results
A total of 177 patients were included—78 in the remimazolam group and 99 in the desflurane group. After PSM, 78 final patients were included in each group. The overall incidence of vasoactive agent use was significantly lower in the remimazolam group than in the desflurane group (41% vs 74% before PSM; 41% vs 73% after PSM; both, P < 0.001). The incidence rate, duration, and maximum dose of continuous vasopressor infusion were also significantly lower in the remimazolam group (P < 0.001). Use of remimazolam was not associated with increased complications after the ablation procedures.
Conclusions
General anesthesia using remimazolam vs desflurane was associated with significantly reduced vasoactive agent requirement and better hemodynamic stability without increased postoperative complications in patients undergoing ablation for atrial fibrillation.
Résumé
Objectif
Des changements hémodynamiques brusques ou des arythmies potentiellement mortelles sont fréquemment observés chez les personnes recevant une anesthésie pour une ablation d’arythmie cardiaque. Le remimazolam est une nouvelle benzodiazépine à action ultra-courte qui a été associé à une meilleure stabilité hémodynamique que les agents anesthésiques conventionnels. Cette étude visait à déterminer si le remimazolam réduit l’utilisation d’agents vasoactifs par rapport au desflurane chez les personnes bénéficiant d’une ablation pour fibrillation auriculaire sous anesthésie générale.
Méthode
Dans une étude de cohorte rétrospective, nous avons examiné les dossiers médicaux électroniques de patient·es adultes ayant bénéficié d’une ablation pour fibrillation auriculaire sous anesthésie générale entre juillet 2021 et juillet 2022. Nous avons divisé les patient·es en groupes de remimazolam et de desflurane en fonction de l’agent principal utilisé pour l’anesthésie. Le critère d’évaluation principal était l’incidence globale de l’utilisation d’agents vasoactifs. Nous avons comparé les groupes à l’aide d’une analyse d’appariement par score de propension (ASP).
Résultats
Au total, 177 patient·es ont été inclus·es, dont 78 dans le groupe remimazolam et 99 dans le groupe desflurane. Après l’ASP, 78 patient·es ont été inclus·es au final dans chaque groupe. L’incidence globale d’utilisation d’agents vasoactifs était significativement plus faible dans le groupe remimazolam que dans le groupe desflurane (41 % vs 74 % avant l’ASP; 41 % vs 73 % après l’ASP; les deux P < 0,001). Le taux d’incidence, la durée et la dose maximale de perfusion continue de vasopresseurs étaient également significativement plus faibles dans le groupe remimazolam (P < 0,001). L’utilisation de remimazolam n’a pas été associée à une augmentation des complications après les interventions d’ablation.
Conclusion
L’anesthésie générale à base de remimazolam vs desflurane a été associée à une réduction significative des besoins en agents vasoactifs et à une meilleure stabilité hémodynamique sans augmentation des complications postopératoires chez les patient·es bénéficiant d’une ablation pour fibrillation auriculaire.


Similar content being viewed by others
References
Kim KM. Remimazolam: pharmacological characteristics and clinical applications in anesthesiology. Anesth Pain Med (Seoul) 2022; 17: 1–11. https://doi.org/10.17085/apm.21115
Goudra BG, Singh PM. Remimazolam: the future of its sedative potential. Saudi J Anaesth 2014; 8: 388–91. https://doi.org/10.4103/1658-354x.136627
Pastis NJ, Yarmus LB, Schippers F, et al. Safety and efficacy of remimazolam compared with placebo and midazolam for moderate sedation during bronchoscopy. Chest 2019; 155: 137–46. https://doi.org/10.1016/j.chest.2018.09.015
Liu T, Lai T, Chen J, et al. Effect of remimazolam induction on hemodynamics in patients undergoing valve replacement surgery: a randomized, double-blind, controlled trial. Pharmacol Res Perspect 2021; 9: e00851. https://doi.org/10.1002/prp2.851
Doi M, Morita K, Takeda J, Sakamoto A, Yamakage M, Suzuki T. Efficacy and safety of remimazolam versus propofol for general anesthesia: a multicenter, single-blind, randomized, parallel-group, phase IIb/III trial. J Anesth 2020; 34: 543–53. https://doi.org/10.1007/s00540-020-02788-6
Dada RS, Hayanga JWA, Woods K, et al. Anesthetic choice for atrial fibrillation ablation: a national anesthesia clinical outcomes registry analysis. J Cardiothorac Vasc Anesth 2021; 35: 2600–6. https://doi.org/10.1053/j.jvca.2020.12.046
Martin CA, Curtain JP, Gajendragadkar PR, et al. Improved outcome and cost effectiveness in ablation of persistent atrial fibrillation under general anaesthetic. Europace 2018; 20: 935–42. https://doi.org/10.1093/europace/eux057
Li KH, Sang T, Chan C, et al. Anaesthesia use in catheter ablation for atrial fibrillation: a systematic review and meta-analysis of observational studies. Heart Asia 2019; 11: e011155. https://doi.org/10.1136/heartasia-2018-011155
Malladi V, Naeini PS, Razavi M, Collard CD, Anton JM, Tolpin DA. Endovascular ablation of atrial fibrillation. Anesthesiology 2014; 120: 1513–9. https://doi.org/10.1097/aln.0000000000000261
Hamid A. Anesthesia for cardiac catheterization procedures. Heart Lung Vessel 2014; 6: 225–31.
von Elm E, Altman DG, Egger M, et al. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: guidelines for reporting observational studies. Lancet 2007; 370: 1453–7. https://doi.org/10.1016/s0140-6736(07)61602-x
Jentzer JC, Vallabhajosyula S, Khanna AK, Chawla LS, Busse LW, Kashani KB. Management of refractory vasodilatory shock. Chest 2018; 154: 416–26. https://doi.org/10.1016/j.chest.2017.12.021
Hansen BB, Klopfer SO. Optimal full matching and related designs via network flows. J Comput Graph Stat 2006; 15: 609–27. https://doi.org/10.1198/106186006X137047
Hansen BB. Full matching in an observational study of coaching for the SAT. J Am Stat Assoc 2004; 99: 609–18. https://doi.org/10.1198/016214504000000647
Schüttler J, Eisenried A, Lerch M, Fechner J, Jeleazcov C, Ihmsen H. Pharmacokinetics and Pharmacodynamics of Remimazolam (CNS 7056) after continuous infusion in healthy male volunteers: part I. Pharmacokinetics and clinical pharmacodynamics. Anesthesiology 2020; 132: 636–51. https://doi.org/10.1097/aln.0000000000003103
Kleiman RB, Darpo B, Thorn M, Stoehr T, Schippers F. Potential strategy for assessing QT/QTc interval for drugs that produce rapid changes in heart rate: electrocardiographic assessment of the effects of intravenous remimazolam on cardiac repolarization. Br J Clin Pharmacol 2020; 86: 1600–9. https://doi.org/10.1111/bcp.14270
Rex DK, Bhandari R, Desta T, et al. A phase III study evaluating the efficacy and safety of remimazolam (CNS 7056) compared with placebo and midazolam in patients undergoing colonoscopy. Gastrointest Endosc 2018; 88: 427–37. https://doi.org/10.1016/j.gie.2018.04.2351
Rex DK, Bhandari R, Lorch DG, Meyers M, Schippers F, Bernstein D. Safety and efficacy of remimazolam in high risk colonoscopy: a randomized trial. Dig Liver Dis 2021; 53: 94–101. https://doi.org/10.1016/j.dld.2020.10.039
Chen SH, Yuan TM, Zhang J, et al. Remimazolam tosilate in upper gastrointestinal endoscopy: a multicenter, randomized, non-inferiority, phase III trial. J Gastroenterol Hepatol 2021; 36: 474–81. https://doi.org/10.1111/jgh.15188
Nakanishi T, Sento Y, Kamimura Y, Tsuji T, Kako E, Sobue K. Remimazolam for induction of anesthesia in elderly patients with severe aortic stenosis: a prospective, observational pilot study. BMC Anesthesiol 2021; 21: 306. https://doi.org/10.1186/s12871-021-01530-3
Landoni G, Lomivorotov VV, Nigro Neto C, et al. Volatile anesthetics versus total intravenous anesthesia for cardiac surgery. N Engl J Med 2019; 380: 1214–25. https://doi.org/10.1056/nejmoa1816476
Kaushal RP, Vatal A, Pathak R. Effect of etomidate and propofol induction on hemodynamic and endocrine response in patients undergoing coronary artery bypass grafting/mitral valve and aortic valve replacement surgery on cardiopulmonary bypass. Ann Card Anaesth 2015; 18: 172–8. https://doi.org/10.4103/0971-9784.154470
Hannam J, Mitchell S, Cumin D, et al. Haemodynamic profiles of etomidate vs propofol for induction of anaesthesia: a randomised controlled trial in patients undergoing cardiac surgery. Br J Anaesth 2019; 122: 198–205. https://doi.org/10.1016/j.bja.2018.09.027
Noseir RK, Ficke DJ, Kundu A, Arain SR, Ebert TJ. Sympathetic and vascular consequences from remifentanil in humans. Anesth Analg 2003; 96: 1645–50. https://doi.org/10.1213/01.ane.0000061587.13631.67
Sajedi P, Rahimian A, Khalili G. Comparative evaluation between two methods of induced hypotension with infusion of Remifentanil and Labetalol during sinus endoscopy. J Res Pharm Pract 2016; 5: 264–71. https://doi.org/10.4103/2279-042x.192463
Schmidt M, Marschang H, Clifford S, et al. Trends in inflammatory biomarkers during atrial fibrillation ablation across different catheter ablation strategies. Int J Cardiol 2012; 158: 33–8. https://doi.org/10.1016/j.ijcard.2010.12.084
Huffman JC, Stern TA. The use of benzodiazepines in the treatment of chest pain: a review of the literature. J Emerg Med 2003; 25: 427–37. https://doi.org/10.1016/j.jemermed.2003.01.001
Dixon R, Edwards IR, Pilcher J. Diazepam in immediate post-myocardial infarct period. A double blind trial. Br Heart J 1980; 43: 535–40. https://doi.org/10.1136/hrt.43.5.535
Wiltshire HR, Kilpatrick GJ, Tilbrook GS, Borkett KM. A Placebo- and midazolam-controlled phase I single ascending-dose study evaluating the safety, pharmacokinetics, and pharmacodynamics of remimazolam (CNS 7056): part II. Population pharmacokinetic and pharmacodynamic modeling and simulation. Anesth Analg 2012; 115: 284–96. https://doi.org/10.1213/ane.0b013e318241f68a
Sheng XY, Liang Y, Yang XY, et al. Safety, pharmacokinetic and pharmacodynamic properties of single ascending dose and continuous infusion of remimazolam besylate in healthy Chinese volunteers. Eur J Clin Pharmacol 2020; 76: 383–91. https://doi.org/10.1007/s00228-019-02800-3
Yamamoto T, Kurabe M, Kamiya Y. Re-sleeping after reversal of remimazolam by flumazenil. J Anesth 2021; 35: 322. https://doi.org/10.1007/s00540-021-02915-x
Penninga EI, Graudal N, Ladekarl MB, Jürgens G. Adverse events associated with flumazenil treatment for the management of suspected benzodiazepine intoxication--a systematic review with meta-analyses of randomised trials. Basic Clin Pharmacol Toxicol 2016; 118: 37–44. https://doi.org/10.1111/bcpt.12434
Author contributions
Sun Woo Nam, In-Ae Song, and Kyoung-Woon Joung contributed to the study design and drafting of the paper. Subin Yim, Chang Ik Choi, Insun Park, Sun Woo Nam, and In-Ae Song contributed to data acquisition. Sun Woo Nam, In-Ae Song, Chang Ik Choi, and Insun Park contributed to data analysis. Sun Woo Nam, In-Ae Song, and Kyoung-Woon Joung contributed to data interpretation and critical revisions of the paper.
Disclosures
The authors claimed no conflicts of interest.
Funding statement
None.
Editorial responsibility
This submission was handled by Dr. Stephan K. W. Schwarz, Editor-in-Chief, Canadian Journal of Anesthesia/Journal canadien d’anesthésie.
Author information
Authors and Affiliations
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Supplementary file1 (PDF 20 KB)
eTableAnesthetic agents used for induction and maintenance
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Nam, S.W., Yim, S., Choi, C.I. et al. Effects of remimazolam on hemodynamic changes during cardiac ablation for atrial fibrillation under general anesthesia: a propensity-score-matched retrospective cohort study. Can J Anesth/J Can Anesth 70, 1495–1503 (2023). https://doi.org/10.1007/s12630-023-02514-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12630-023-02514-2