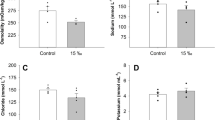
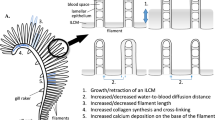
Abstract
Bombay duck Harpodon nehereus has undergone a dramatic shift from a by-catch fish species to the dominant species in the southeast coast of China. This shift has led to economic growth in the fishery and has had a significant impact on community structure, biodiversity, and resource competition in the ecosystem. Strong hypoxia tolerance may be one of the contributing factors for its population growth. This study compared the transcriptional regulation and alternative splicing in H. nehereus from the normoxic and hypoxic zone near the Yangtze Estuary. Compared with normoxia, the number of alternative splicing events increased in the gill and heart but decreased in the muscle after hypoxia, among which exon skipping was the most prevalent type in different tissues. Enrichment analysis showed that differentially expressed genes and genes with differentially alternative exons were mainly enriched in transmembrane transport and glycerophospholipid metabolic process in gill, cell cycle, and FoxO signaling pathway in heart, as well as cell cycle and reduced metabolic rate in muscle. Key candidate genes may be associated with gill remodeling, elevated heart rate, and muscle angiogenesis function. This study provides valuable genetic clues for evaluating molecular regulation strategies related to hypoxia tolerance in marine fishes.





Similar content being viewed by others
References
Alleboina S, Ayalew D, Peravali R, Chen LD, Wong T, Dokun AO (2019) Dual specificity phosphatase 5 regulates perfusion recovery in experimental peripheral artery disease. Vasc Med 24(5):395–404
Anders S, Reyes A, Huber W (2012) Detecting differential usage of exons from RNA-seq data. Genome Res 22(10):2008–2017
Bapat SV (1970) The Bombay duck, Harpodon nehereus (Ham.). Bull Cent Mar Fish Res Inst 21:1–87
Bendix PM, Koenderink GJ, Cuvelier D, Dogic Z, Koeleman BM, Brieher WM, Field CM, Mahadevan L, Weitz DA (2008) A quantitative analysis of contractility in active cytoskeletal protein networks. Biophys J 94(8):3126–3136
Breitburg D, Levin LA, Oschlies A, Gregoire M, Chavez FP, Conley DJ, Garcon V, Gilbert D, Gutierrez D, Isensee K, Jacinto GS, Limburg KE, Montes I, Naqvi SWA, Pitcher GC, Rabalais NN, Roman MR, Rose KA, Seibel BA, Telszewski M, Yasuhara M, Zhang J (2018) Declining oxygen in the global ocean and coastal waters. Science 359(6371): eaam7240. https://doi.org/10.1126/science.aam7240
Burleson ML, Silva PE (2011) Cross tolerance to environmental stressors: effects of hypoxic acclimation on cardiovascular responses of channel catfish (Ictalurus punctatus) to a thermal challenge. J Therm Biol 36(4):250–254
Castillo-Briceno P, Bihan D, Nilges M, Hamaia S, Meseguer J, Garcia-Ayala A, Farndale RW, Mulero V (2011) A role for specific collagen motifs during wound healing and inflammatory response of fibroblasts in the teleost fish gilthead seabream. Mol Immunol 48(6–7):826–834
Chen CC, Gong GC, Shiah FK (2007) Hypoxia in the East China Sea: One of the largest coastal low-oxygen areas in the world. Mar Environ Res 64(4):399–408
Chen BX, Yi SK, Wang WF, He Y, Huang Y, Gao ZX, Liu H, Wang WM, Wang HL (2017) Transcriptome comparison reveals insights into muscle response to hypoxia in blunt snout bream (Megalobrama amblycephala). Gene 624:6–13
Chen SF, Zhou YQ, Chen YR, Gu J (2018) fastp: an ultra-fast all-in-one FASTQ preprocessor. Bioinformatics 34(17):884–890
Chen FJ, Ling XD, Zhao YT, Fu SY (2022) Hypoxia-induced oxidative stress and apoptosis in gills of scaleless carp (Gymnocypris przewalskii). Fish Physiol Biochem 48(4):911–924
Chen G, Pang MX, Yu XM, Wang JR, Tong JO (2021) Transcriptome sequencing provides insights into the mechanism of hypoxia adaption in bighead carp (Hypophthalmichthys nobilis). Comp Biochem Phys D 40:100891. https://doi.org/10.1016/j.cbd.2021.100891
Dhillon RS, Yao LL, Matey V, Chen BJ, Zhang AJ, Cao ZD, Fu SJ, Brauner CJ, Wang YXS, Richards JG (2013) Interspecific differences in hypoxia-induced gill remodeling in carp. Physiol Biochem Zool 86(6):727–739
Diaz RJ, Rosenberg R (2008) Spreading dead zones and consequences for marine ecosystems. Science 321(5891):926–929
Downie AT, Lefevre S, Illing B, Harris J, Jarrold MD, McCormick MI, Nilsson GE, Rummer JL (2023) Rapid physiological and transcriptomic changes associated with oxygen delivery in larval anemonefish suggest a role in adaptation to life on hypoxic coral reefs. Plos Biol 21(5):e3002102. https://doi.org/10.1371/journal.pbio.3002102
Dutertre M, Sanchez G, De Cian MC, Barbier J, Dardenne E, Gratadou L, Dujardin G, Le Jossic-Corcos C, Corcos L, Auboeuf D (2010) Cotranscriptional exon skipping in the genotoxic stress response. Nat Struct Mol Biol 17(11):1358-U1218
Feng C, Li XH, Sha H, Luo XZ, Zou GW, Liang HW (2022) Comparative transcriptome analysis provides novel insights into the molecular mechanism of the silver carp (Hypophthalmichthys molitrix) brain in response to hypoxia stress. Comp Biochem Phys D 41:100951. https://doi.org/10.1016/j.cbd.2021.100951
Firdaus M, Lelono TD, Bintoro G, Saleh R, Salim G (2018) The expression of the body shape in fish species Harpadon nehereus (Hamilton, 1822) in the waters of Juata Laut, Tarakan city, North Kalimantan. AACL Bioflux 11(3):613–624
Foissac S, Sammeth M (2007) ASTALAVISTA: dynamic and flexible analysis of alternative splicing events in custom gene datasets. Nucleic Acids Res 35:W297–W299
Gilkes DM, Semenza GL, Wirtz D (2014) Hypoxia and the extracellular matrix: drivers of tumour metastasis. Nat Rev Cancer 14(6):430–439
Grau-Bove X, Ruiz-Trillo I, Irimia M (2018) Origin of exon skipping-rich transcriptomes in animals driven by evolution of gene architecture. Genome Biol 19(1):135. https://doi.org/10.1186/s13059-018-1499-9
Hammond EM, Giaccia AJ (2005) The role of p53 in hypoxia-induced apoptosis. Biochem Bioph Res Co 331(3):718–725
Hashimoto T, Yamasaki S, Taguchi S (2003) Alterations in the expression of myosin heavy chain isoforms in hypoxia-induced hypertrophied ventricles in rats. Comp Biochem Phys B 136(1):139–145
Hedrick SM (2009) The cunning little vixen: Foxo and the cycle of life and death. Nat Immunol 10(10):1057–1063
Heidebrecht HJ, Buck F, Steinmann J, Sprenger R, Wacker HH, Parwaresch R (1997) p100: A novel proliferation-associated nuclear protein specifically restricted to cell cycle phases S, G(2), and M. Blood 90(1):226–233
Huang H, Bogstie JN, Vogel HJ (2012) Biophysical and structural studies of the human calcium- and integrin-binding protein family: understanding their functional similarities and differences. Biochem Cell Biol 90(5):646–656
Jiang RJ, Sun HQ, Li XF, Zhou YD, Chen F, Xu KD, Li PF, Zhang HL (2022) Habitat suitability evaluation of Harpadon nehereus in nearshore of Zhejiang province, China. Front Mar Sci 9:961735
Joyce W, Ozolina K, Mauduit F, Ollivier H, Claireaux G, Shiels HA (2016) Individual variation in whole-animal hypoxia tolerance is associated with cardiac hypoxia tolerance in a marine teleost. Biol Letters 12(1):20150708. https://doi.org/10.1098/rsbl.2015.0708
Kanehisa M, Goto S, Furumichi M, Tanabe M, Hirakawa M (2000) KEGG for representation and analysis of molecular networks involving diseases and drugs. Nucleic Acids Res 38:D335–D360
Kang B, Bakun A, Lin LS, Pauly D (2022) Increase of a hypoxia-tolerant fish, Harpadon nehereus (Synodontidae), as a result of ocean deoxygenation off southwestern China. Environ Biol Fish 105(10):1399–1403
Karar J, Maity A (2011) PI3K/AKT/mTOR pathway in angiogenesis. Front Mol Neurosci 4:1–8. https://doi.org/10.3389/fnmol.2011.00051
Ki SM, Kim JH, Won SY, Oh SJ, Lee IY, Bae YK, Chung KW, Choi BO, Park B, Choi EJ, Lee JE (2020) CEP41-mediated ciliary tubulin glutamylation drives angiogenesis through AURKA-dependent deciliation. Embo Rep 21(2):e48290. https://doi.org/10.15252/embr.201948290
Kim D, Paggi JM, Park C, Bennett C, Salzberg SL (2019) Graph-based genome alignment and genotyping with HISAT2 and HISAT-genotype. Nat Biotechnol 37(8):907–915. https://doi.org/10.1038/s41587-019-0201-4
LaGory EL, Giaccia AJ (2016) The ever-expanding role of HIF in tumour and stromal biology. Nat Cell Biol 18(4):356–365
Lee KE, Simon MC (2015) SnapShot: hypoxia-inducible factors. Cell 163(5):1288–1288
Lee CY, Hu SY, Gong HY, Chen MHC, Lu JK, Wu JL (2009) Suppression of myostatin with vector-based RNA interference causes a double-muscle effect in transgenic zebrafish. Biochem Bioph Res Co 387(4):766–771
Li MH, Wang DS (2017) Gene editing nuclease and its application in tilapia. Sci Bull 62(3):165–173
Li DJ, Zhang J, Huang DJ, Wu Y, Liang J (2002) Oxygen depletion off the Changjiang (Yangtze River) estuary. Sci China Ser D 45(12):1137–1146
Li J, Zhang GS, Yin DQ, Li Y, Zhang YR, Cheng JH, Zhang K, Ji J, Wang T, Jia YY, Yin SW (2022) Integrated application of multiomics strategies provides insights into the environmental hypoxia response in pelteobagrus vachelli muscle. Mol Cell Proteomics 21(3):100196. https://doi.org/10.1016/j.mcpro.2022.100196
Liao Y, Smyth GK, Shi W (2014) featureCounts: an efficient general purpose program for assigning sequence reads to genomic features. Bioinformatics 30(7):923–930
Liu CL, Shi GP (2019) Calcium-activated chloride channel regulator 1 (CLCA1): More than a regulator of chloride transport and mucus production. World Allergy Organ 12(11):100077. https://doi.org/10.1016/j.waojou.2019.100077
Masoud GN, Li W (2015) HIF-1α pathway: role, regulation and intervention for cancer therapy. Acta Pharm Sin B 5(5):378–389
Mu YN, Li WR, Wei ZY, He LH, Zhang WN, Chen XH (2020) Transcriptome analysis reveals molecular strategies in gills and heart of large yellow croaker (Larimichthys crocea) under hypoxia stress. Fish Shellfish Immun 104:304–313
Nie HT, Wang HM, Jiang KY, Yan XW (2020) Transcriptome analysis reveals differential immune related genes expression in Ruditapes philippinarum under hypoxia stress: potential HIF and NF-kappa B crosstalk in immune responses in clam. Bmc Genomics 21(1):318. https://doi.org/10.1186/s12864-020-6734-6
Nilsen TW, Graveley BR (2010) Expansion of the eukaryotic proteome by alternative splicing. Nature 463(7280):457–463
Nooralabettu KP (2008) Effect of sun drying and artificial drying of fresh, salted Bombay duck (Harpadon nehereus) on the physical characteristics of the product. J Aquat Food Prod T 17(2):99–116
Palazón A, Aragonés J, Morales-Kastresana A, Ortiz M, de Landázuri MO, Melero I (2012) Molecular pathways: hypoxia response in immune cells fighting or promoting cancer. Clin Cancer Res Off J Am Assoc Cancer 18(5):1207–1213. https://doi.org/10.1158/1078-0432.CCR-11-1591
Podrabsky JE, Lopez JP, Fan TWM, Higashi R, Somero GN (2007) Extreme anoxia tolerance in embryos of the annual killifish Austrofundulus limnaeus: insights from a metabolomics analysis. J Exp Biol 210(13):2253–2266
Poss KD, Wilson LG, Keating MT (2002) Heart regeneration in zebrafish. Science 298(5601):2188–2190
Qiang J, Zhu XW, He J, Tao YF, Bao JW, Zhu JH, Xu P (2020) miR-34a regulates the activity of HIF-1a and P53 signaling pathways by promoting GLUT1 in genetically improved farmed tilapia (GIFT, Oreochromis niloticus) under hypoxia stress. Front Physiol 11:670. https://doi.org/10.3389/fphys.2020.00670
Richards JG (2009) Chapter 10 Metabolic and molecular responses of fish to hypoxia. Fish Physiol 27:443–485
Robinson MD, McCarthy DJ, Smyth GK (2010) edgeR: a Bioconductor package for differential expression analysis of digital gene expression data. Bioinformatics 26(1):139–140
Rossi GS, Wright PA (2020) Hypoxia-seeking behavior, metabolic depression and skeletal muscle function in an amphibious fish out of water. J Exp Biol 223(2):jeb213355. https://doi.org/10.1242/jeb.213355
Schito L, Semenza GL (2016) Hypoxia-inducible factors: master regulators of cancer progression. Trends Cancer 2(12):758–770
Semenza GL, Jiang BH, Leung SW, Passantino R, Concordet JP, Maire P, Giallongo A (1996) Hypoxia response elements in the aldolase A, enolase 1, and lactate dehydrogenase A gene promoters contain essential binding sites for hypoxia-inducible factor 1. J Biol Chem 271(51):32529–32537
Song PQ, Zhang HS, Zheng XQ, Li YY, Lin LS, Li YA (2021) Trophic plasticity of Bombay Duck (Harpadon nehereus) in the South-Central East China Sea based on stable isotope evidence. Front Mar Sci 8:728773. https://doi.org/10.3389/fmars.2021.728773
Sun P, Dai F, Chen Y, Shan X, Jin X (2015) Seasonal Variations in Structure of Fishery Resource in the Yangtze River Estuary and Its Adjacent Waters (in Chinese). Prog Fish Sci 36(6):8–16
Sun HQ (2020) A study on the biological characteristics, temporal and spatial distribution, and the relationship with environmental factors of Harpadon nehereus. In: Zhejiang Province coastal waters (in Chinese). Master’s thesis, Zhejiang Ocean University
Taqwa A, Burhanuddin AI, Niartiningsih A, Nessa MN (2020) Nomei fish (Harpadon nehereus, Ham. 1822) reproduction biology in Tarakan waters. IOP Conf Ser Earth Environ Sci 473:012012
Tian CX, Lin XH, Saetan W, Huang Y, Shi HJ, Jiang DN, Chen HP, Deng SP, Wu TL, Zhang YL, Li GL, Zhu CH (2020) Transcriptome analysis of liver provides insight into metabolic and translation changes under hypoxia and reoxygenation stress in silver sillago (Sillago sihama). Comp Biochem Phys D 36:100715. https://doi.org/10.1016/j.cbd.2020.100715
Turko AJ, Cisternino B, Wright PA (2020) Calcified gill filaments increase respiratory function in fishes. P Roy Soc B-Biol Sci 287(1920):20192796. https://doi.org/10.1098/rspb.2019.2796
Vase VK, Kumar R, Nakhwa AD, Rahangdale S, Jayasankar J, Koya KM, Divu D, Rohit P (2020) Fishery and estimation of potential yield of Bombay duck Harpadon nehereus (Hamilton, 1822) along Gujarat coast. India Indian J Fish 67(1):1–7
Vu TD, Oshima K, Matsumura K, Iwasaki Y, Chiu MT, Nikaido M, Okada N (2022) Alternative splicing plays key roles in response to stress across different stages of fighting in the fish Betta splendens. Bmc Genomics 22(5):920. https://doi.org/10.1186/s12864-022-08677-4
Wang LL, Li JT, Kang B (2022) Fish growth evaluation and fishery dynamic assessment of Harpadon nehereus providing insights for its population expansion in the Northern East China Sea. Hydrobiologia 849(21):4725–4739
Wanida S, Ye MH, Lin XH, Lin XZ, Zhang YL, Huang Y, Du T, Li GL, Tian CX (2021) Comparative Transcriptome Analysis of Heart Tissue in Response to Hypoxia in Silver Sillago (Sillago sihama). J Ocean U China 20(4):949–958
Wei QS, Yu ZG, Xia CS, Zang JY, Zhang XL (2011) A preliminary analysis on the dynamic characteristics of the hypoxic zone adjacent to the Changjiang Estuary in summer. Acta Oceanol Sin 33:100–109
Wei QS, Wang BD, Chen JF, Xia CS, Qu DP, Xie LP (2015) Recognition on the forming-vanishing process and underlying mechanisms of the hypoxia off the Yangtze River estuary. Sci China Earth Sci 58(4):628–648
Wei QS, Yuan YQ, Song SQ, Zhao YH, Sun JC, Li CW, Wang BD (2022) Spatial variability of hypoxia and coupled physical-biogeochemical controls off the Changjiang (Yangtze River) Estuary in summer. Front Mar Sci 9:987368. https://doi.org/10.3389/fmars.2022.987368
Williams KJ, Cassidy AA, Verhille CE, Lamarre SG, MacCormack TJ (2019) Diel cycling hypoxia enhances hypoxia tolerance in rainbow trout (Oncorhynchus mykiss): evidence of physiological and metabolic plasticity. J Exp Biol 222(14) jeb206045. https://doi.org/10.1242/jeb.206045
Wu RSS (2002) Hypoxia: from molecular responses to ecosystem responses. Mar Pollut Bull 45(1–12):35–45
Xiao WH (2015) The hypoxia signaling pathway and hypoxic adaptation in fishes. Sci China Life Sci 58(2):148–155
Xu XN, Chen SL, Jiang ZX, Nissa MU, Zou SM (2022) Gill remodeling increases the respiratory surface area of grass carp (Ctenopharyngodon idella) under hypoxic stress. Comp Biochem Phys A 272:111278
Yang TY, Huang XX, Jiang YL (2022) Reveal the population genetic characteristics of Bombay duck (Harpadon nehereus) in coastal waters of China with genotyping-by-sequencing technique. J Ocean U China 21(5):1373–1380
Yong L, Jiahua C (2015) A preliminary analysis of variation characteristics of structure and average trophic level of the main fishery species caught by paired bottom trawl in the East China Sea and the Yellow Sea during the fall season (in Chinese). J Fisheries China 390(5):691–702
Yu GC, Wang LG, Han YY, He QY (2012) clusterProfiler: an R package for comparing biological themes among gene clusters. OMICS 16(5):284–287
Zhang J, Cowie G, Naqvi SWA (2013) Hypoxia in the changing marine environment. Environ Res Lett 8(1):015025. https://doi.org/10.1088/1748-9326/8/1/015025
Zhang B, Jin XS (2014) Feeding habits and ontogenetic diet shifts of Bombay duck. Harpadon Nehereus Chin J Oceanol Limn 32(3):542–548
Zhao LL, Cui C, Liu Q, Sun JL, He K, Adam AA, Luo J, Li ZQ, Wang Y, Yang S (2020) Combined exposure to hypoxia and ammonia aggravated biological effects on glucose metabolism, oxidative stress, inflammation and apoptosis in largemouth bass (Micropterus salmoides). Aquat Toxicol 224:105514. https://doi.org/10.1016/j.aquatox.2020.105514
Zhao SS, Su XL, Pan RJ, Lu LQ, Zheng GD, Zou SM (2022) The transcriptomic responses of blunt snout bream (Megalobrama amblycephala) to acute hypoxia stress alone, and in combination with bortezomib. Bmc Genomics 23(1):162. https://doi.org/10.1186/s12864-022-08399-7
Zhou F, Huang DJ, Ni XB (2010) Hydrographic analysis on the multi-time scale variability of hypoxia adjacent to the Changjiang River Estuary (in Chinese). Acta Ecol Sin 30:4728–4740
Zhu ZY, Zhang J, Wu Y, Zhang YY, Lin J, Liu SM (2011) Hypoxia off the Changjiang (Yangtze River) Estuary: Oxygen depletion and organic matter decomposition. Mar Chem 125(1–4):108–116
Acknowledgements
The authors thank all members of the laboratory for their valuable discussions.
Funding
This study was supported by the National Natural Science Foundation of China (No.U20A2087 and No.41976091).
Author information
Authors and Affiliations
Contributions
Z.S. was involved in the methodology, software, formal analysis, data curation, writing—original draft, and visualization. L.W. carried out the investigation, resources, writing—original draft, and validation. Y.K. was involved in software use, formal analysis, writing—original draft, aand validation. J.S. performed the formal analysis, data curation, and validation. B.K. carried out the conceptualization, validation, writing—review and editing, supervision, project administration, and funding acquisition.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sun, Z., Wang, L., Kong, Y. et al. Transcriptional regulation and alternative splicing reveal the molecular strategies of Bombay duck Harpadon nehereus to hypoxia. Fish Sci 90, 201–213 (2024). https://doi.org/10.1007/s12562-024-01753-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12562-024-01753-6