Abstract
This work aims to exploit a novel graph neural network to predict the sex of the brain topological network, and to find the sex differences in the cerebrum and cerebellum. A two-branch multi-scale graph convolutional network (TMGCN) is designed to analyze the sex differences of the brain. Two complementary templates are used to construct cerebrum and cerebellum networks, respectively, followed by a two-branch sub-network with multi-scale filters and a trainable weighted fusion strategy for the final prediction. Finally, a trainable graph topk-pooling layer is utilized in our model to visualize key brain regions relevant to the prediction. The proposed TMGCN achieves a prediction accuracy of 84.48%. In the cerebellum, the bilateral Crus I–II, lobule VI and VIIb, and the posterior vermis (VI–X) are discriminative for this task. As for the cerebrum, the discriminative brain regions consist of the bilateral inferior temporal gyrus, the bilateral fusiform gyrus, the bilateral parahippocampal gyrus, the bilateral cingulate gyrus, the bilateral medial ventral occipital cortex, the bilateral lateral occipital cortex, the bilateral amygdala, and the bilateral hippocampus. This study tackles the sex prediction problem from a more comprehensive view, and may provide the resting-state fMRI evidence for further study of sex differences in the cerebellum and cerebrum.
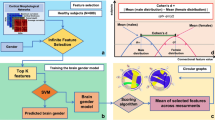
Graphical Abstract






Similar content being viewed by others
Data Availability
We used the rs-fMRI of the Southwest University Longitudinal Imaging Multimodal (SLIM) dataset in the experiment. This dataset is provided by the International Data-sharing Initiative (INDI, http://fcon_1000.projects.nitrc.org/).
Code Availability
Our work was finished by the custom code.
References
Ruigrok AN et al (2014) A meta-analysis of sex differences in human brain structure. Neurosci Biobehav Rev 39:34–50. https://doi.org/10.1016/j.neubiorev.2013.12.004
Xin J, Zhang Y, Tang Y, Yang Y (2019) Brain differences between men and women: evidence from deep learning. Front Neurosci 13:185. https://doi.org/10.3389/fnins.2019.00185
Bluhm RL et al (2008) Default mode network connectivity: effects of age sex, and analytic approach. NeuroReport 19(8):887–891. https://doi.org/10.1097/WNR.0b013e328300ebbf (in English)
Allen EA et al (2011) A baseline for the multivariate comparison of resting-state networks. Front Syst Neurosci 5:2. https://doi.org/10.3389/fnsys.2011.00002
Miller DI, Halpern DF (2014) The new science of cognitive sex differences. Trends Cogn Sci 18(1):37–45. https://doi.org/10.1016/j.tics.2013.10.011
Malpetti M et al (2017) Gender differences in healthy aging and Alzheimer’s Dementia: A (18) F-FDG-PET study of brain and cognitive reserve. Hum Brain Mapp 38(8):4212–4227. https://doi.org/10.1002/hbm.23659
Alaerts K, Swinnen SP, Wenderoth N (2016) Sex differences in autism: a resting-state fMRI investigation of functional brain connectivity in males and females. Soc Cogn Affect Neurosci 11(6):1002–1016. https://doi.org/10.1093/scan/nsw027
Orgo L, Bachmann M, Kalev K, Hinrikus H, Jarvelaid M (2016) Brain functional connectivity in depression: gender differences in EEG. In 2016 Ieee Embs Conference on Biomedical Engineering and Sciences (Iecbes), 2016, pp 270–273. https://doi.org/10.1109/IECBES.2016.7843456
Bassett DS, Sporns O (2017) Network neuroscience. Nat Neurosci 20(3):353–364. https://doi.org/10.1038/nn.4502
Tang Y et al (2016) Aberrant functional brain connectome in people with antisocial personality disorder. Sci Rep 6(1):1–12. https://doi.org/10.1038/srep26209
Tian L, Wang J, Yan C, He Y (2011) Hemisphere-and gender-related differences in small-world brain networks: a resting-state functional MRI study. Neuroimage 54(1):191–202. https://doi.org/10.1016/j.neuroimage.2010.07.066
Zhao Y et al (2017) Automatic recognition of fMRI-derived functional networks using 3-D convolutional neural networks. IEEE Trans Biomed Eng 65(9):1975–1984. https://doi.org/10.1109/TBME.2017.2715281
Ktena SI et al (2018) Metric learning with spectral graph convolutions on brain connectivity networks. Neuroimage 169:431–442. https://doi.org/10.1016/j.neuroimage.2017.12.052
Yang H et al (2019) Interpretable multimodality embedding of cerebral cortex using attention graph network for identifying bipolar disorder. In: Medical Image Computing and Computer Assisted Intervention – MICCAI 2019. https://doi.org/10.1007/978-3-030-32248-9_89
Ma G et al. (2019) Deep graph similarity learning for brain data analysis. In: Proceedings of the 28th ACM International Conference on Information and Knowledge Management. https://doi.org/10.1145/3357384.3357815
Liu J, Ma G, Jiang F, Lu C, Yu PS, Ragin AB (2019) Community-preserving graph convolutions for structural and functional joint embedding of brain networks. In 2019 IEEE International Conference on Big Data (Big Data), pp 1163–1168. https://doi.org/10.1109/BigData47090.2019.9005586
Parisot S et al (2018) Disease prediction using graph convolutional networks: application to autism spectrum disorder and alzheimer’s disease. Med Image Anal 48:117–130. https://doi.org/10.1016/j.media.2018.06.001
Li X et al (2021) Braingnn: Interpretable brain graph neural network for fmri analysis. Med Image Anal 74:102233. https://doi.org/10.1016/j.media.2021.102233
Yao D et al (2021) A mutual multi-scale triplet graph convolutional network for classification of brain disorders using functional or structural connectivity. IEEE Trans Med Imaging 40(4):1279–1289. https://doi.org/10.1109/TMI.2021.3051604
Kim BH, Ye JC (2020) Understanding graph isomorphism network for rs-fMRI functional connectivity analysis. Front Neurosci 14:630. https://doi.org/10.3389/fnins.2020.00630
Arslan S, Ktena SI, Glocker B, Rueckert D (2018) Graph saliency maps through spectral convolutional networks: application to sex classification with brain connectivity. In Biomedical Image Analysis and Integrating Medical Imaging and Non-Imaging Modalities https://doi.org/10.1007/978-3-030-00689-1_1
Filippi M, Valsasina P, Misci P, Falini A, Comi G, Rocca MA (2013) The organization of intrinsic brain activity differs between genders: a resting-state fMRI study in a large cohort of young healthy subjects. Hum Brain Mapp 34(6):1330–1343. https://doi.org/10.1002/hbm.21514
Fan L et al (2010) Sexual dimorphism and asymmetry in human cerebellum: an MRI-based morphometric study. Brain Res 1353:60–73. https://doi.org/10.1016/j.brainres.2010.07.031
Andersen BB, Gundersen HJG, Pakkenberg B (2003) Aging of the human cerebellum: a stereological study. J Comp Neurol 466(3):356–365. https://doi.org/10.1002/cne.10884
Tiemeier H, Lenroot RK, Greenstein DK, Tran L, Pierson R, Giedd JN (2010) Cerebellum development during childhood and adolescence: a longitudinal morphometric MRI study. Neuroimage 49(1):63–70. https://doi.org/10.1016/j.neuroimage.2009.08.016
Gur RC et al (1995) Sex differences in regional cerebral glucose metabolism during a resting state. Science 267(5197):528–531. https://doi.org/10.1126/science.7824953
Jiang T (2013) Brainnetome: a new -ome to understand the brain and its disorders. Neuroimage 80:263–272. https://doi.org/10.1016/j.neuroimage.2013.04.002
Fan L et al (2016) The human brainnetome atlas: a new brain atlas based on connectional architecture. Cereb Cortex 26(8):3508–3526. https://doi.org/10.1093/cercor/bhw157
Tzourio-Mazoyer N et al (2002) Automated anatomical labeling of activations in SPM using a macroscopic anatomical parcellation of the MNI MRI single-subject brain. Neuroimage 15(1):273–289. https://doi.org/10.1006/nimg.2001.0978
Li Y et al (2010) Cerebellum abnormalities in idiopathic generalized epilepsy with generalized tonic-clonic seizures revealed by diffusion tensor imaging. PLoS ONE 5(12):e15219. https://doi.org/10.1371/journal.pone.0015219
Guo W et al (2013) Is there a cerebellar compensatory effort in first-episode, treatment-naive major depressive disorder at rest? Prog Neuro-Psychopharmacol Biol Psychiatry 46:13–18. https://doi.org/10.1016/j.pnpbp.2013.06.009
Collin G, Hulshoff Pol HE, Haijma SV, Cahn W, Kahn RS, van den Heuvel MP (2011) Impaired cerebellar functional connectivity in schizophrenia patients and their healthy siblings. Front Psych 2:73. https://doi.org/10.3389/fpsyt.2011.00073
Chung MK, Luo Z, Adluru N, Alexander AL, Davidson RJ, Goldsmith HH (2018) Heritability of nested hierarchical structural brain network. In 2018 40th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), IEEE, pp 554–557 https://doi.org/10.1109/EMBC.2018.8512359
Yu S, Yue G, Elazab A, Song X, Wang T, Lei B (2019) Multi-scale graph convolutional network for mild cognitive impairment detection. In Graph Learning in Medical Imaging https://doi.org/10.1007/978-3-030-35817-4_10
Kazi A et al (2019) InceptionGCN: receptive field aware graph convolutional network for disease prediction. In Information Processing in Medical Imaging https://doi.org/10.1007/978-3-030-20351-1_6
Liu W et al (2017) Longitudinal test-retest neuroimaging data from healthy young adults in southwest China. Scientific Data 4(1):1–9. https://doi.org/10.1038/sdata.2017.17
Satterthwaite TD et al (2013) An improved framework for confound regression and filtering for control of motion artifact in the preprocessing of resting-state functional connectivity data. Neuroimage 64:240–256. https://doi.org/10.1016/j.neuroimage.2012.08.052
Jenkinson M, Bannister P, Brady M, Smith S (2002) Improved optimization for the robust and accurate linear registration and motion correction of brain images. Neuroimage 17(2):825–841. https://doi.org/10.1006/nimg.2002.1132
Wei D, Yang J, Li W, Wang K, Zhang Q, Qiu J (2014) Increased resting functional connectivity of the medial prefrontal cortex in creativity by means of cognitive stimulation. Cortex 51:92–102. https://doi.org/10.1016/j.cortex.2013.09.004
Tian X et al (2016) Assessment of trait anxiety and prediction of changes in state anxiety using functional brain imaging: a test-retest study. Neuroimage 133:408–416. https://doi.org/10.1016/j.neuroimage.2016.03.024
Yan C-G et al (2013) A comprehensive assessment of regional variation in the impact of head micromovements on functional connectomics. Neuroimage 76:183–201. https://doi.org/10.1016/j.neuroimage.2013.03.004
Wu Z, Pan S, Chen F, Long G, Zhang C, Yu PS (2021) A comprehensive survey on graph neural networks. IEEE Trans Neural Netw Learn Syst 32(1):4–24. https://doi.org/10.1109/TNNLS.2020.2978386
Xie Y, Yao C, Gong M, Chen C, Qin AK (2020) Graph convolutional networks with multi-level coarsening for graph classification. Knowledge-Based Syst 194:105578. https://doi.org/10.1016/j.knosys.2020.105578
Guo F, Li Z, Xin Z, Zhu X, Wang L, Zhang J (2021) Dual Graph U-Nets for Hyperspectral Image Classification. IEEE J Sel Top Appl Earth Observations Remote Sensing 14:8160–8170. https://doi.org/10.1109/JSTARS.2021.3103744
Li X et al (2020) Pooling regularized graph neural network for fMRI biomarker analysis. In Med Image Comput Comput Assist Interv https://doi.org/10.1007/978-3-030-59728-3_61
Xia M, Wang J, He Y (2013) BrainNet Viewer: a network visualization tool for human brain connectomics. PLoS ONE 8(7):e68910. https://doi.org/10.1371/journal.pone.0068910
Shine JM, Aburn MJ, Breakspear M, Poldrack RA (2018) The modulation of neural gain facilitates a transition between functional segregation and integration in the brain. Elife 7:e31130. https://doi.org/10.7554/eLife.31130.001
Sanz-Arigita EJ et al (2010) Loss of ‘small-world’ networks in Alzheimer’s disease: graph analysis of FMRI resting-state functional connectivity. PLoS ONE 5(11):e13788. https://doi.org/10.1371/journal.pone.0013788
Stoodley CJ, Schmahmann JD (2009) Functional topography in the human cerebellum: a meta-analysis of neuroimaging studies. Neuroimage 44(2):489–501. https://doi.org/10.1016/j.neuroimage.2008.08.039
Bernard JA et al (2012) Resting state cortico-cerebellar functional connectivity networks: a comparison of anatomical and self-organizing map approaches. Front Neuroanatomy 6:31. https://doi.org/10.3389/fnana.2012.00031
Steele CJ, Chakravarty MM (2018) Gray-matter structural variability in the human cerebellum: lobule-specific differences across sex and hemisphere. Neuroimage 170:164–173. https://doi.org/10.1016/j.neuroimage.2017.04.066
Womer FY et al (2016) Sexual dimorphism of the cerebellar vermis in schizophrenia. Schizophrenia Res 176(2–3):164–170. https://doi.org/10.1016/j.schres.2016.06.028
Lee K-H et al (2007) Increased cerebellar vermis white-matter volume in men with schizophrenia. J Psychiatric Res 41(8):645–651. https://doi.org/10.1016/j.jpsychires.2006.03.001
Rossi A, Stratta P, Fabrizio M, de Cataldo S, Casacchia M (1993) Cerebellar vermal size in schizophrenia: a male effect. Biol Psychiatry 33(5):354–357. https://doi.org/10.1016/0006-3223(93)90324-7
Okugawa G, Sedvall GC, Agartz I (2003) Smaller cerebellar vermis but not hemisphere volumes in patients with chronic schizophrenia. Am J Psychiatry 160(9):1614–1617. https://doi.org/10.1176/appi.ajp.160.9.1614
Haznedar MM, Buchsbaum MS, Hazlett EA, Shihabuddin L, New A, Siever LJ (2004) Cingulate gyrus volume and metabolism in the schizophrenia spectrum. Schizophrenia Res 71(2–3):249–262. https://doi.org/10.1016/j.schres.2004.02.025
Brun CC et al (2009) Sex differences in brain structure in auditory and cingulate regions. NeuroReport 20(10):930. https://doi.org/10.1097/wnr.0b013e32832c5e65
Chen X, Sachdev PS, Wen W, Anstey KJ (2007) Sex differences in regional gray matter in healthy individuals aged 44–48 years: a voxel-based morphometric study. Neuroimage 36(3):691–699. https://doi.org/10.1016/j.neuroimage.2007.03.063
van Eijk L et al (2020) Region-specific sex differences in the hippocampus. Neuroimage 215:116781. https://doi.org/10.1016/j.neuroimage.2020.116781
Sneider JT, Rogowska J, Sava S, Yurgelun-Todd DA (2011) A preliminary study of sex differences in brain activation during a spatial navigation task in healthy adults. Perceptual Motor Skills 113(2):461–480. https://doi.org/10.2466/04.22.24.27
Marwha D, Halari M, Eliot L (2017) Meta-analysis reveals a lack of sexual dimorphism in human amygdala volume. Neuroimage 147:282–294. https://doi.org/10.1016/j.neuroimage.2016.12.021
Mather M, Lighthall NR, Nga L, Gorlick MA (2010) Sex differences in how stress affects brain activity during face viewing. NeuroReport 21(14):933. https://doi.org/10.1097/WNR.0b013e32833ddd92
Bear D, Schiff D, Saver J, Greenberg M, Freeman R (1986) Quantitative analysis of cerebral asymmetries: fronto-occipital correlation, sexual dimorphism and association with handedness. Arch Neurol 43(6):598–603. https://doi.org/10.1001/archneur.1986.00520060060019
Acknowledgements
This work was supported in part by the High Performance Computing Center of Central South University. The author would like to thank the 2020 Key Project of Research on Postgraduate Education and Teaching Reform of Central South University [grant numbers 2020JGA011], the 2020 Hunan Province Degree and Postgraduate Education Reform Research Project [grant number 2020JGZD014] and the Research Fund of the Guangxi Key Lab of Multi-source Information Mining and Security [grant number MIMS20-08] for their supports.
Funding
This work was supported by the 2020 Key Project of Research on Postgraduate Education and Teaching Reform of Central South University [grant numbers 2020JGA011], the 2020 Hunan Province Degree and Postgraduate Education Reform Research Project [grant number 2020JGZD014] and the Research Fund of the Guangxi Key Lab of Multi-source Information Mining and Security [grant number MIMS20-08].
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Ethical Approval
This study was approved by the Research Ethics Committee of Central South University and The University of Oklahoma.
Consent to Participate
The authors have agreed to participate in this work.
Consent for Publication
The publication of this work was approved by Central South University and The University of Oklahoma.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Gao, Y., Tang, Y., Zhang, H. et al. Sex Differences of Cerebellum and Cerebrum: Evidence from Graph Convolutional Network. Interdiscip Sci Comput Life Sci 14, 532–544 (2022). https://doi.org/10.1007/s12539-021-00498-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12539-021-00498-5