Abstract
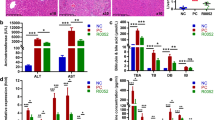
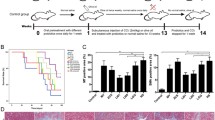
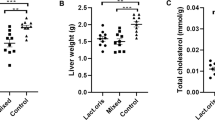
The progression and exacerbation of liver fibrosis are closely related to the gut microbiome. It is hypothesized that some probiotics may slow the progression of liver fibrosis. In human stool analysis [healthy group (n = 44) and cirrhosis group (n = 18)], difference in Lactobacillus genus between healthy group and cirrhosis group was observed. Based on human data, preventive and therapeutic effect of probiotics Lactobacillus lactis and L. rhamnosus was evaluated by using four mice fibrosis models. L. lactis and L. rhamnosus were supplied to 3,5-diethoxycarbonyl-1,4-dihydrocollidine or carbon tetrachloride-induced liver fibrosis C57BL/6 mouse model. Serum biochemical measurements, tissue staining, and mRNA expression in the liver were evaluated. The microbiome was analyzed in mouse cecal contents. In the mouse model, the effects of Lactobacillus in preventing and treating liver fibrosis were different for each microbe species. In case of L. lactis, all models showed preventive and therapeutic effects against liver fibrosis. In microbiome analysis in mouse models administered Lactobacillus, migration and changes in the ratio and composition of the gut microbial community were confirmed. L. lactis and L. rhamnosus showed preventive and therapeutic effects on the progression of liver fibrosis, suggesting that Lactobacillus intake may be a useful strategy for prevention and treatment.





Similar content being viewed by others
Data availability
Data available within the article or its supplementary materials.
References
Acharya, C., & Bajaj, J. S. (2019). Altered microbiome in patients with cirrhosis and complications. Clinical Gastroenterology and Hepatology, 17, 307–321.
Arroyo, V., Moreau, R., Kamath, P. S., Jalan, R., Gines, P., Nevens, F., Fernandez, J., To, U., Garcia-Tsao, G., & Schnabl, B. (2016). Acute-on-chronic liver failure in cirrhosis. Nature Reviews Disease Primers, 2, 16041.
Azad, M. A. K., Sarker, M., Li, T., & Yin, J. (2018). Probiotic species in the modulation of gut microbiota: An overview. BioMed Research International, 2018, 9478630.
Bajaj, J. S., Heuman, D. M., Hylemon, P. B., Sanyal, A. J., Puri, P., Sterling, R. K., Luketic, V., Stravitz, R. T., Siddiqui, M. S., Fuchs, M., Thacker, L. R., Wade, J. B., Daita, K., Sistrun, S., White, M. B., Noble, N. A., Thorpe, C., Kakiyama, G., Pandak, W. M., … Gillevet, P. M. (2014). Randomised clinical trial: Lactobacillus gg modulates gut microbiome, metabolome and endotoxemia in patients with cirrhosis. Alimentary Pharmacology & Therapeutics, 39, 1113–1125.
Bataller, R., & Brenner, D. A. (2005). Liver fibrosis. Journal of Clinical Investigation, 115, 209–218.
Belkaid, Y., & Hand, T. W. (2014). Role of the microbiota in immunity and inflammation. Cell, 157, 121–141.
Carroll, I. M., Andrus, J. M., Bruno-Barcena, J. M., Klaenhammer, T. R., Hassan, H. M., & Threadgill, D. S. (2007). Anti-inflammatory properties of Lactobacillusgasseri expressing manganese superoxide dismutase using the interleukin 10-deficient mouse model of colitis. American Journal of Physiology-Gastrointestinal and Liver Physiology, 293, G729-738.
Dong, S., Chen, Q. L., Song, Y. N., Sun, Y., Wei, B., Li, X. Y., Hu, Y. Y., Liu, P., & Su, S. B. (2016). Mechanisms of ccl4-induced liver fibrosis with combined transcriptomic and proteomic analysis. The Journal of Toxicological Sciences, 41, 561–572.
Edgar, R. C. (2010). Search and clustering orders of magnitude faster than blast. Bioinformatics, 26, 2460–2461.
Famouri, F., Shariat, Z., Hashemipour, M., Keikha, M., & Kelishadi, R. (2017). Effects of probiotics on nonalcoholic fatty liver disease in obese children and adolescents. Journal of Pediatric Gastroenterology and Nutrition, 64, 413–417.
Fukui, H. (2019). Role of gut dysbiosis in liver diseases: What have we learned so far? Diseases, 7, 58.
Ghiassi-Nejad, Z., & Friedman, S. L. (2008). Advances in antifibrotic therapy. Expert Review of Gastroenterology & Hepatology, 2, 803–816.
Hemarajata, P., & Versalovic, J. (2013). Effects of probiotics on gut microbiota: Mechanisms of intestinal immunomodulation and neuromodulation. Therapeutic Advances in Gastroenterology, 6, 39–51.
Huang, C. H., Li, S. W., Huang, L., & Watanabe, K. (2018). Identification and classification for the Lactobacilluscasei group. Frontiers in Microbiology, 9, 1974.
Kanmani, P., & Kim, H. (2022). Probiotics counteract the expression of hepatic profibrotic genes via the attenuation of tgf-beta/smad signaling and autophagy in hepatic stellate cells. PLoS ONE, 17, e0262767.
Kim, S. K., Guevarra, R. B., Kim, Y. T., Kwon, J., Kim, H., Cho, J. H., Kim, H. B., & Lee, J. H. (2019). Role of probiotics in human gut microbiome-associated diseases. Journal of Microbiology and Biotechnology, 29, 1335–1340.
Lee, N. Y., Joung, H. C., Kim, B. K., Kim, B. Y., Park, T. S., & Suk, K. T. (2020a). Lactobacillus lactis ckdb001 ameliorate progression of nonalcoholic fatty liver disease through of gut microbiome: Addendum. Gut Microbes, 12, 1829449.
Lee, N. Y., Yoon, S. J., Han, D. H., Gupta, H., Youn, G. S., Shin, M. J., Ham, Y. L., Kwak, M. J., Kim, B. Y., Yu, J. S., Lee, D. Y., Park, T. S., Park, S. H., Kim, B. K., Joung, H. C., Choi, I. S., Hong, J. T., Kim, D. J., Han, S. H., & Suk, K. T. (2020b). Lactobacillus and pediococcus ameliorate progression of non-alcoholic fatty liver disease through modulation of the gut microbiome. Gut Microbes, 11, 882–899.
Liu, Y., Chen, K., Li, F., Gu, Z., Liu, Q., He, L., Shao, T., Song, Q., Zhu, F., Zhang, L., Jiang, M., Zhou, Y., Barve, S., Zhang, X., McClain, C. J., & Feng, W. (2020). Probiotic Lactobacillusrhamnosus gg prevents liver fibrosis through inhibiting hepatic bile acid synthesis and enhancing bile acid excretion in mice. Hepatology, 71, 2050–2066.
Markowiak, P., & Slizewska, K. (2017). Effects of probiotics, prebiotics, and synbiotics on human health. Nutrients, 9, 1–30.
Marotta, F., Barreto, R., Wu, C. C., Naito, Y., Gelosa, F., Lorenzetti, A., Yoshioka, M., & Fesce, E. (2005). Experimental acute alcohol pancreatitis-related liver damage and endotoxemia: Synbiotics but not metronidazole have a protective effect. Chinese Journal of Digestive Diseases, 6, 193–197.
Mulaw, G., SisayTessema, T., Muleta, D., & Tesfaye, A. (2019). In vitro evaluation of probiotic properties of lactic acid bacteria isolated from some traditionally fermented Ethiopian food products. International Journal of Microbiology, 2019, 7179514.
Ortega, M. A., Fraile-Martinez, O., Naya, I., Garcia-Honduvilla, N., Alvarez-Mon, M., Bujan, J., Asunsolo, A., & de la Torre, B. (2020). Type 2 diabetes mellitus associated with obesity (diabesity). The central role of gut microbiota and its translational applications. Nutrients, 12, 2749.
Pan, F., Zhang, L., Li, M., Hu, Y., Zeng, B., Yuan, H., Zhao, L., & Zhang, C. (2018). Predominant gut lactobacillus murinus strain mediates anti-inflammaging effects in calorie-restricted mice. Microbiome., 6, 54.
Pose, E., Sancho-Bru, P., & Coll, M. (2019). 3,5-diethoxycarbonyl-1,4-dihydrocollidine diet: A rodent model in cholestasis research. Methods in Molecular Biology, 1981, 249–257.
Ray, K. (2017). Alcoholic liver disease: Gut-liver axis: Ppis, enterococcus and promotion of alcoholic liver disease. Nature Reviews Gastroenterology & Hepatology, 14, 689.
Schnabl, B., & Brenner, D. A. (2014). Interactions between the intestinal microbiome and liver diseases. Gastroenterology, 146, 1513–1524.
Schuppan, D., & Afdhal, N. H. (2008). Liver cirrhosis. The Lancet, 371, 838–851.
Segawa, S., Wakita, Y., Hirata, H., & Watari, J. (2008). Oral administration of heat-killed lactobacillus brevis sbc8803 ameliorates alcoholic liver disease in ethanol-containing diet-fed c57bl/6n mice. International Journal of Food Microbiology, 128, 371–377.
Sharma, V., Garg, S., & Aggarwal, S. (2013). Probiotics and liver disease. The Permanente Journal, 17, 62–67.
Shi, D., Lv, L., Fang, D., Wu, W., Hu, C., Xu, L., Chen, Y., Guo, J., Hu, X., Li, A., Guo, F., Ye, J., Li, Y., Andayani, D., & Li, L. (2017). Administration of Lactobacillus salivarius li01 or Pediococcus pentosaceus li05 prevents ccl4-induced liver cirrhosis by protecting the intestinal barrier in rats. Scientific Reports, 7, 6927.
Trautwein, C., Friedman, S. L., Schuppan, D., & Pinzani, M. (2015). Hepatic fibrosis: Concept to treatment. Journal of Hepatology, 62, S15-24.
Turnbaugh, P. J., & Stintzi, A. (2011). Human health and disease in a microbial world. Frontiers in Microbiology, 2, 190.
Walker, A. W., & Lawley, T. D. (2013). Therapeutic modulation of intestinal dysbiosis. Pharmacological Research, 69, 75–86.
Wang, H., Zhou, C., Huang, J., Kuai, X., & Shao, X. (2020). The potential therapeutic role of Lactobacillusreuteri for treatment of inflammatory bowel disease. American Journal of Translational Research, 12, 1569–1583.
Wiegand, J., & Berg, T. (2013). The etiology, diagnosis and prevention of liver cirrhosis: Part 1 of a series on liver cirrhosis. Deutsches Ärzteblatt International, 110, 85–91.
Wynn, T. A. (2008). Cellular and molecular mechanisms of fibrosis. Journal of Pathology, 214, 199–210.
Wynn, T. A., & Ramalingam, T. R. (2012). Mechanisms of fibrosis: Therapeutic translation for fibrotic disease. Nature Medicine, 18, 1028–1040.
Xie, J. T., Shao, Z. H., Vanden Hoek, T. L., Chang, W. T., Li, J., Mehendale, S., Wang, C. Z., Hsu, C. W., Becker, L. B., Yin, J. J., & Yuan, C. S. (2006). Antioxidant effects of ginsenoside re in cardiomyocytes. European Journal of Pharmacology, 532, 201–207.
Yanguas, S. C., Cogliati, B., Willebrords, J., Maes, M., Colle, I., van den Bossche, B., de Oliveira, C., Andraus, W., Alves, V. A. F., Leclercq, I., & Vinken, M. (2016). Experimental models of liver fibrosis. Archives of Toxicology, 90, 1025–1048.
Yoon, S. H., Ha, S. M., Kwon, S., Lim, J., Kim, Y., Seo, H., & Chun, J. (2017). Introducing ezbiocloud: A taxonomically united database of 16s rrna gene sequences and whole-genome assemblies. International Journal of Systematic and Evolutionary Microbiology, 67, 1613–1617.
Zhang, Y. J., Li, S., Gan, R. Y., Zhou, T., Xu, D. P., & Li, H. B. (2015). Impacts of gut bacteria on human health and diseases. International Journal of Molecular Sciences, 16, 7493–7519.
Acknowledgements
This research was supported by Hallym University Research Fund, Korea National Research Foundation (2020R1A6A1A03043026, and 2021M3A9I4021433), Bio Industrial Technology Development Program (20018494) funded by the Ministry of Trade, Industry and Energy (MOTIE, Korea), and the Promotion of Innovative Businesses for Regulation-Free Special Zones funded by the Ministry of SMEs and Startups (MSS, Korea) (P0020622).
Author information
Authors and Affiliations
Contributions
SMW, NYL, and SKK: analysis and interpretation of the data, collection and assembly of data, drafting of the article. SMW and KTS: conception and design, critical revision of the article for important intellectual content, final approval of the article. KKO, HG, SPS, KHK, BKK, HCJ, JJJ, RG, SHH, SJY, and DJK: critical revision of the article for important intellectual content and provision of study materials.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interest, including relevant financial interests, activities, relationships, affiliations, and any other conflict of interest as explicitly and implicitly expressed in the Editorial Policies for Authors.
Ethical approval
This study was controlled in accordance with ethical guidelines from the 1975 Helsinki Declaration as reflected by prior approval by the institutional review notice for human research in the hospital participating in the trial (2016-134). Basic information has been registered on ClinicalTrials.gov for registration in the public trial registry (NCT04339725). Informed consent for enrollment was received from each participant. This experiment was designed by selecting a candidate for the treatment of liver disease among the probiotics that approved by Ministry of Food and Drug Safety, Korean. All the procedures were licensed by the Institutional Animal Care and Use Committee of the College of Medicine, Hallym University R1(2019-04).
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Won, S.M., Lee, N.Y., Oh, KK. et al. Gut Lactobacillus and Probiotics Lactobacillus lactis/rhamnosis Ameliorate Liver Fibrosis in Prevention and Treatment. J Microbiol. 61, 245–257 (2023). https://doi.org/10.1007/s12275-023-00014-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12275-023-00014-y