Abstract
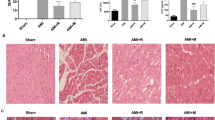
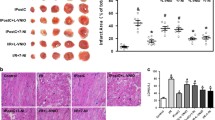
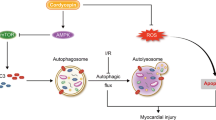
Remote ischemic conditioning (RIC) can be effectively applied for cardio-protection. Here, to clarify whether RIC exerts myocardial protection via aldehyde dehydrogenase 2 (ALDH2), we established a myocardial ischemia/reperfusion (I/R) model in C57BL/6 and ALDH2 knockout (ALDH2-KO) mice and treated them with RIC. Echocardiography and single-cell contraction experiments showed that RIC significantly improved myocardial function and alleviated I/R injury in C57BL/6 mice but did not exhibit its cardioprotective effects in ALDH2-KO mice. TUNEL, Evan’s blue/triphenyl tetrazolium chloride, and reactive oxygen species (ROS) assays showed that RIC’s effect on reducing myocardial cell apoptosis, myocardial infarction area, and ROS levels was insignificant in ALDH2-KO mice. Our results showed that RIC could increase ALDH2 protein levels, activate sirtuin 3 (SIRT3)/hypoxia-inducible factor 1-alpha (HIF1α), inhibit autophagy, and exert myocardial protection. This study revealed that RIC could exert myocardial protection via the ALDH2/SIRT3/HIF1α signaling pathway by reducing 4-HNE secretion.






Similar content being viewed by others
Data Availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Code Availability
Not applicable.
Abbreviations
- RIC:
-
Remote ischemic conditioning
- ALDH2:
-
Aldehyde dehydrogenase 2
- I/R:
-
Ischemia/reperfusion
- ROS:
-
Reactive oxygen species
- SIRT3:
-
Sirtuin 3
- HIF1α:
-
Hypoxia-inducible factor 1-alpha
- 4-HNE:
-
4-hydroxynonenal
- ALDH2-KO:
-
ALDH2 knockout
- TTC:
-
1% 2,3,5-triphenyl tetrazolium chloride
- PS:
-
Peak shortening
- -dL/dt:
-
Maximal velocity of shortening
- DHE:
-
Dihydroethidium
- LVFS:
-
Left ventricular fractional shortening
- LVEF:
-
Left ventricular ejection fraction
- AAR:
-
Area at risk
- WT:
-
Wild-type
References
Heusch G. Myocardial ischaemia-reperfusion injury and cardioprotection in perspective. Nat Rev Cardiol. 2020;17:773–89.
Davidson SM, Ferdinandy P, Andreadou I, et al. Multitarget strategies to reduce myocardial ischemia/reperfusion injury: JACC review topic of the week. J Am Coll Cardiol. 2019;73:89–99.
Hirschhäuser C, Lissoni A, Görge PM, et al. Connexin 43 phosphorylation by casein kinase 1 is essential for the cardioprotection by ischemic preconditioning. Basic Res Cardiol. 2021;116:21.
Hong DM, Lee EH, Kim HJ, et al. Does remote ischaemic preconditioning with postconditioning improve clinical outcomes of patients undergoing cardiac surgery? Remote Ischaemic Preconditioning with Postconditioning Outcome Trial. Eur Heart J. 2014;35:176–83.
Wei X, Wu B, Zhao J, et al. Myocardial hypertrophic preconditioning attenuates cardiomyocyte hypertrophy and slows progression to heart failure through upregulation of S100A8/A9. Circulation. 2015;131:1506–17.
Skyschally A, Gent S, Amanakis G, et al. Across-species transfer of protection by remote ischemic preconditioning with species-specific myocardial signal transduction by reperfusion injury salvage kinase and survival activating factor enhancement pathways. Circ Res. 2015;117:279–88.
Amanakis G, Kleinbongard P, Heusch G, et al. Attenuation of ST-segment elevation after ischemic conditioning maneuvers reflects cardioprotection online. Basic Res Cardiol. 2019;114:22.
Zarbock A, Schmidt C, Van Aken H, et al. Effect of remote ischemic preconditioning on kidney injury among high-risk patients undergoing cardiac surgery: a randomized clinical trial. JAMA. 2015;313:2133–41.
Chen CH, Ferreira JC, Gross ER, et al. Targeting aldehyde dehydrogenase 2: new therapeutic opportunities. Physiol Rev. 2014;94:1–34.
Gross ER, Zambelli VO, Small BA, et al. A personalized medicine approach for Asian Americans with the aldehyde dehydrogenase 2*2 variant. Annu Rev Pharmacol Toxicol. 2015;55:107–27.
Ma LL, Ding ZW, Yin PP, et al. Hypertrophic preconditioning cardioprotection after myocardial ischaemia/reperfusion injury involves ALDH2-dependent metabolism modulation. Redox Biol. 2021;43:101960.
Chen CH, Budas GR, Churchill EN, et al. Activation of aldehyde dehydrogenase-2 reduces ischemic damage to the heart. Science. 2008;321:1493–5.
Ueta CB, Campos JC, Albuquerque RPE, et al. Cardioprotection induced by a brief exposure to acetaldehyde: role of aldehyde dehydrogenase 2. Cardiovasc Res. 2018;114:1006–15.
Zhang ZX, Li H, He JS, et al. Remote ischemic postconditioning alleviates myocardial ischemia/reperfusion injury by up-regulating ALDH2. Eur Rev Med Pharmacol Sci. 2018;22:6475–84.
Yu Y, Jia XJ, Zong QF, et al. Remote ischemic postconditioning protects the heart by upregulating ALDH2 expression levels through the PI3K/Akt signaling pathway. Mol Med Rep. 2014;10:536–42.
Li CY, Ma W, Liu KP, et al. Advances in intervention methods and brain protection mechanisms of in situ and remote ischemic postconditioning. Metab Brain Dis. 2021;36:53–65.
Joseph B, Pandit V, Zangbar B, et al. Secondary brain injury in trauma patients: the effects of remote ischemic conditioning. J Trauma Acute Care Surg. 2015;78:698–703.
Kitagawa K, Kawamoto T, Kunugita N, et al. Aldehyde dehydrogenase (ALDH) 2 associates with oxidation of methoxyacetaldehyde; in vitro analysis with liver subcellular fraction derived from human and Aldh2 gene targeting mouse. FEBS Lett. 2000;476:306–11.
Sun X, Gao R, Li W, et al. Alda-1 treatment promotes the therapeutic effect of mitochondrial transplantation for myocardial ischemia-reperfusion injury. Bioact Mater. 2021;6:2058–69.
Gao RF, Li X, Xiang HY, et al. The covalent NLRP3-inflammasome inhibitor Oridonin relieves myocardial infarction induced myocardial fibrosis and cardiac remodeling in mice. Int Immunopharmacol. 2021;90:107133.
Ackers-Johnson M, Li PY, Holmes AP, et al. A simplified, Langendorff-free method for concomitant isolation of viable cardiac myocytes and nonmyocytes from the adult mouse heart. Circ Res. 2016;119:909–20.
Fan Q, Tao R, Zhang H, et al. Dectin-1 contributes to myocardial ischemia/reperfusion injury by regulating macrophage polarization and neutrophil infiltration. Circulation. 2019;139:663–78.
Si Y, Bao H, Han L, et al. Dexmedetomidine attenuation of renal ischaemia-reperfusion injury requires sirtuin 3 activation. Br J Anaesth. 2018;121:1260–71.
Wang Z, Sun R, Wang G, et al. SIRT3-mediated deacetylation of PRDX3 alleviates mitochondrial oxidative damage and apoptosis induced by intestinal ischemia/reperfusion injury. Redox Biol. 2020;28:101343.
Lu Z, Bourdi M, Li JH, et al. SIRT3-dependent deacetylation exacerbates acetaminophen hepatotoxicity. EMBO Rep. 2011;12:840–6.
Stiermaier T, Jensen JO, Rommel KP, et al. Combined intrahospital remote ischemic perconditioning and postconditioning improves clinical outcome in ST-elevation myocardial infarction. Circ Res. 2019;124:1482–91.
Vaibhav K, Braun M, Khan MB, et al. Remote ischemic post-conditioning promotes hematoma resolution via AMPK-dependent immune regulation. J Exp Med. 2018;215:2636–54.
Heusch G. Molecular basis of cardioprotection: signal transduction in ischemic pre-, post-, and remote conditioning. Circ Res. 2015;116:674–99.
Tsibulnikov SY, Maslov LN, Gorbunov AS, et al. A review of humoral factors in remote preconditioning of the heart. J Cardiovasc Pharmacol Ther. 2019;24:403–21.
Hummitzsch L, Zitta K, Fritze L, et al. Effects of remote ischemic preconditioning (RIPC) and chronic remote ischemic preconditioning (cRIPC) on levels of plasma cytokines, cell surface characteristics of monocytes and in-vitro angiogenesis: a pilot study. Basic Res Cardiol. 2021;116:60.
Gao J, Min F, Wang S, et al. Effect of Rho-kinase and autophagy on remote ischemic conditioning-induced cardioprotection in rat myocardial ischemia/reperfusion injury model. Cardiovasc Ther. 2022;2022:6806427.
Yan Z, Du L, Liu Q, et al. Remote limb ischaemic conditioning produces cardioprotection in rats with testicular ischaemia-reperfusion injury. Exp Physiol. 2021;106:2223–34.
Zhao Y, Ding M, Yan F, et al. Inhibition of the JAK2/STAT3 pathway and cell cycle re-entry contribute to the protective effect of remote ischemic pre-conditioning of rat hindlimbs on cerebral ischemia/reperfusion injury. CNS Neurosci Ther. 2022. https://doi.org/10.1111/cns.14023.
Mi L, Zhang N, Wan J, et al. Remote ischemic post-conditioning alleviates ischemia/reperfusion-induced intestinal injury via the ERK signaling pathway-mediated RAGE/HMGB axis. Mol Med Rep. 2021:24.
Ajoolabady A, Aslkhodapasandhokmabad H, Aghanejad A, et al. Mitophagy receptors and mediators: therapeutic targets in the management of cardiovascular ageing. Ageing Res Rev. 2020;62:101129.
Santin Y, Fazal L, Sainte-Marie Y, et al. Mitochondrial 4-HNE derived from MAO-A promotes mitoCa(2+) overload in chronic postischemic cardiac remodeling. Cell Death Differ. 2020;27:1907–23.
Beretta M, Wölkart G, Schernthaner M, et al. Vascular bioactivation of nitroglycerin is catalyzed by cytosolic aldehyde dehydrogenase-2. Circ Res. 2012;110:385–93.
Ma H, Guo R, Yu L, et al. Aldehyde dehydrogenase 2 (ALDH2) rescues myocardial ischaemia/reperfusion injury: role of autophagy paradox and toxic aldehyde. Eur Heart J. 2011;32:1025–38.
Zhai M, Li B, Duan W, et al. Melatonin ameliorates myocardial ischemia reperfusion injury through SIRT3-dependent regulation of oxidative stress and apoptosis. J Pineal Res. 2017;63.
Harris PS, Gomez JD, Backos DS, et al. Characterizing sirtuin 3 deacetylase affinity for aldehyde dehydrogenase 2. Chem Res Toxicol. 2017;30:785–93.
Koeppen M, Lee JW, Seo SW, et al. Hypoxia-inducible factor 2-alpha-dependent induction of amphiregulin dampens myocardial ischemia-reperfusion injury. Nat Commun. 2018;9:816.
Cai Z, Luo W, Zhan H, et al. Hypoxia-inducible factor 1 is required for remote ischemic preconditioning of the heart. Proc Natl Acad Sci U S A. 2013;110:17462–7.
Kalakech H, Tamareille S, Pons S, et al. Role of hypoxia inducible factor-1α in remote limb ischemic preconditioning. J Mol Cell Cardiol. 2013;65:98–104.
Zhao Y, Wang B, Zhang J, et al. ALDH2 (aldehyde dehydrogenase 2) protects against hypoxia-induced pulmonary hypertension. Arterioscler Thromb Vasc Biol. 2019;39:2303–19.
Laitakari A, Ollonen T, Kietzmann T, et al. Systemic inactivation of hypoxia-inducible factor prolyl 4-hydroxylase 2 in mice protects from alcohol-induced fatty liver disease. Redox Biol. 2019;22:101145.
Ji W, Wei S, Hao P, et al. Aldehyde dehydrogenase 2 has cardioprotective effects on myocardial ischaemia/reperfusion injury via suppressing mitophagy. Front Pharmacol. 2016;7:101.
Kornfeld OS, Hwang S, Disatnik MH, et al. Mitochondrial reactive oxygen species at the heart of the matter: new therapeutic approaches for cardiovascular diseases. Circ Res. 2015;116:1783–99.
Guéraud F. 4-Hydroxynonenal metabolites and adducts in pre-carcinogenic conditions and cancer. Free Radic Biol Med. 2017;111:196–208.
Mak S, Lehotay DC, Yazdanpanah M, et al. Unsaturated aldehydes including 4-OH-nonenal are elevated in patients with congestive heart failure. J Card Fail. 2000;6:108–14.
Eng MY, Luczak SE, Wall TL. ALDH2, ADH1B, and ADH1C genotypes in Asians: a literature review. Alcohol Res Health. 2007;30:22–7.
Yukawa Y, Muto M, Hori K, et al. Combination of ADH1B*2/ALDH2*2 polymorphisms alters acetaldehyde-derived DNA damage in the blood of Japanese alcoholics. Cancer Sci. 2012;103:1651–5.
Li H, Borinskaya S, Yoshimura K, et al. Refined geographic distribution of the oriental ALDH2*504Lys (nee 487Lys) variant. Ann Hum Genet. 2009;73:335–45.
Funding
This research was funded by the Jiangxi Provincial Natural Science Foundation Project (20071BBG70067, 20181074), and the National Natural Science Foundation of China (81160019 and 81360031).
Author information
Authors and Affiliations
Contributions
Rifeng Gao, Chunyu Lv, and Yanan Qu designed the research and wrote the paper. Rifeng Gao, Heng Yang, Xiaolei Sun, and Chuangze Hao performed the experiments. Rifeng Gao, Xiaosheng Hu, Yiqing Yang, and Yanhua Tang contributed new reagents/analytic tools and provide critical suggestions. Rifeng Gao, Chunyu Lv, and Yanan Qu analyzed the data. Xiaosheng Hu, Yiqing Yang, and Yanhua Tang edited the paper.
Corresponding authors
Ethics declarations
Consent for Publication
Not applicable.
Consent to Participate
Not applicable.
Animal Studies
All animal experiments were approved by the Animal Care Ethics Committee of Fudan University and performed in accordance with the National Institutes of Health Guide for the Care and Use of Laboratory Animals. The mice were kept under a 12:12-h light/dark cycle at a consistent temperature and humidity and were also given ad libitum access to food and water. Additional dose of analgesics was given if the animals appeared to be experiencing pain (based on criteria such as immobility and failure to eat). At the indicated time points, mice were euthanized by CO2/cervical dislocation, and tissues were subsequently harvested for analyses.
Conflict of Interest
The authors declare no competing interests.
Additional information
Associate Editor Yihua Bei oversaw the review of this article
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information

Supplementary Figure 1
RIC treatment preserves cardiac function after I/R. Representative images of echocardiography tracing in I/R mice treated with RIC in each group after 24 h reperfusion (a). Heart rate (BPM, n>6, b). Left ventricular ejection fraction (LVEF, n>6, c). Left ventricular fractional shortening (LVFS, n>6, D). Peak shortening (% cell lengthening, n>6, e). Maximal shortening velocity (-dl/dt, n>6, f). Data are depicted as the mean ± SEM. Statistical significance was determined by two-way ANOVA with a post-hoc Holm-Sidak test, ns, not significant; *P<0.05; ***P<0.001; compared with the control group. (PNG 1135 kb)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Gao, R., Lv, C., Qu, Y. et al. Remote Ischemic Conditioning Mediates Cardio-protection After Myocardial Ischemia/Reperfusion Injury by Reducing 4-HNE Levels and Regulating Autophagy via the ALDH2/SIRT3/HIF1α Signaling Pathway. J. of Cardiovasc. Trans. Res. 17, 169–182 (2024). https://doi.org/10.1007/s12265-023-10355-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12265-023-10355-z