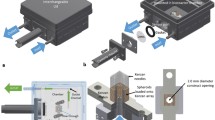
Abstract
Various culture devices have been developed as fundamental technologies for facilitating bioengineering studies. Culture devices are designed to prepare specific culture environments. Thus, both macrostructures and surface micromorphology should be considered in the device design. Although fabricating devices with elaborate designs incurs high production costs, disposable materials are typically used for culture devices. However, some metallic materials are strong, stable, and biocompatible. Bioengineers have not applied these materials to culture devices because of the difficulty of processing. An emerging technology using three-dimensional (3D) printing has been developed, which can produce complex designs using metal. We demonstrate the applicability and potential of metal 3D printing for fabricating culture devices toward the development of the bioengineering discipline. As a specific example, we fabricated metallic culture devices where the environment of cultured tissues can be improved. One of the biggest factors determining the culture environment is active media supply. To attain active media supply to the tissue, devices having culture surfaces with mesh structures having holes far larger than cells were proposed. Cell sheets were cultured as tissue models, realizing tissue culture with such structures. The cultured tissue showed increased metabolism, indicating enhanced media supply owing to mesh surfaces. The biocompatibility of the 3D printed metal device was confirmed by viability assays on cultured cells, and reusability of the device was confirmed by mechanical and biochemical evaluations. We believe this study serves as a reference for using metallic 3D printing as an option for fabricating culture devices, which will promote bioengineering research.
Similar content being viewed by others
References
Matsumoto, E., N. Koide, H. Hanzawa, M. Kiyama, M. Ohta, J. Kuwabara, S. Takeda, and M. Takahashi (2019) Fabricating retinal pigment epithelial cell sheets derived from human induced pluripotent stem cells in an automated closed culture system for regenerative medicine. PLoS One 14: e0212369.
Broeckx, S., M. Zimmerman, S. Crocetti, M. Suls, T. Mariën, S. J. Ferguson, K. Chiers, L. Duchateau, A. Franco-Obregón, K. Wuertz, and J. H. Spaas (2014) Regenerative therapies for equine degenerative joint disease: a preliminary study. PLoS One 9: e85917.
Traister, A., R. Patel, A. Huang, S. Patel, J. Plakhotnik, J. E. Lee, M. G. Medina, C. Welsh, P. Ruparel, L. Zhang, M. Friedberg, J. Maynes, and J. Coles (2018) Cardiac regenerative capacity is age- and disease-dependent in childhood heart disease. PLoS One 13: e0200342. (Erratum published 2021, PLoS One 16: e0245808)
Nakao, M., C. Imashiro, T. Kuribara, Y. Kurashina, K. Totani, and K. Takemura (2019) Formation of large scaffold-free 3-D aggregates in a cell culture dish by ultrasound standing wave trapping. Ultrasound Med. Biol. 45: 1306–1315.
Ackermann, M., H. Kempf, M. Hetzel, C. Hesse, A. R. Hashtchin, K. Brinkert, J. W. Schott, K. Haake, M. P. Kühnel, S. Glage, C. Figueiredo, D. Jonigk, K. Sewald, A. Schambach, S. Wronski, T. Moritz, U. Martin, R. Zweigerdt, A. Munder, and N. Lachmann (2018) Bioreactor-based mass production of human iPSC-derived macrophages enables immunotherapies against bacterial airway infections. Nat. Commun. 9: 5088.
Watanabe, M., K. Yano, K. Okawa, T. Yamashita, K. Tajima, K. Sawada, H. Yagi, Y. Kitagawa, K. Tanishita, and R. Sudo (2019) Construction of sinusoid-scale microvessels in perfusion culture of a decellularized liver. Acta Biomater. 95: 307–318.
Sakaguchi, K., Y. Tobe, J. Yang, R. Tanaka, K. Yamanaka, J. Ono, and T. Shimizu (2021) Bioengineering of a scaffold-less three-dimensional tissue using net mould. Biofabrication 13: https://doi.org/10.1088/1758-5090/ac23e3.
Esch, E. W., A. Bahinski, and D. Huh (2015) Organs-on-chips at the frontiers of drug discovery. Nat. Rev. Drug Discov. 14: 248–260.
Oyama, T., C. Imashiro, T. Kuriyama, H. Usui, K. Ando, T. Azuma, A. Morikawa, K. Kodeki, O. Takahara, and K. Takemura (2021) Acoustic streaming induced by MHz-frequency ultrasound extends the volume limit of cell suspension culture. J. Acoust. Soc. Am. 149: 4180.
van Duinen, V., D. Zhu, C. Ramakers, A. J. van Zonneveld, P. Vulto, and T. Hankemeier (2019) Perfused 3D angiogenic sprouting in a high-throughput in vitro platform. Angiogenesis 22: 157–165.
Park, H. J., Z. Fan, Y. Bai, Q. Ren, Y. Rbaibi, K. R. Long, M. L. Gliozzi, N. Rittenhouse, J. D. Locker, A. C. Poholek, and O. A. Weisz (2020) Transcriptional programs driving shear stress-induced differentiation of kidney proximal tubule cells in culture. Front. Physiol. 11: 587358.
Imashiro, C., B. Kang, Y. Lee, Y. H. Hwang, S. Im, D. E. Kim, K. Takemura, and H. Lee (2021) Propagating acoustic waves on a culture substrate regulate the directional collective cell migration. Microsyst. Nanoeng. 7: 90.
Anggayasti, W. L., C. Imashiro, T. Kuribara, K. Totani, and K. Takemura (2020) Low-frequency mechanical vibration induces apoptosis of A431 epidermoid carcinoma cells. Eng. Life Sci. 20: 232–238.
Lam, R. H. W., Y. Sun, W. Chen, and J. Fu (2012) Elastomeric microposts integrated into microfluidics for flow-mediated endothelial mechanotransduction analysis. Lab. Chip. 12: 1865–1873.
Talò, G., C. Turrisi, C. Arrigoni, C. Recordati, I. Gerges, M. Tamplenizza, A. Cappelluti, S. A. Riboldi, and M. Moretti (2018) Industrialization of a perfusion bioreactor: prime example of a non-straightforward process. J. Tissue Eng. Regen. Med. 12: 405–415.
Imashiro, C., Y. Tokuoka, K. Kikuhara, T. G. Yamada, K. Takemura, and A. Funahashi (2020) Direct cell counting using macro-scale smartphone images of cell aggregates. IEEE Access 8: 170033–170043.
Kato, R., D. Iejima, H. Agata, I. Asahina, K. Okada, M. Ueda, H. Honda, and H. Kagami (2010) A compact, automated cell culture system for clinical scale cell expansion from primary tissues. Tissue Eng. Part C Methods 16: 947–956.
Olmer, R., A. Lange, S. Selzer, C. Kasper, A. Haverich, U. Martin, and R. Zweigerdt (2012) Suspension culture of human pluripotent stem cells in controlled, stirred bioreactors. Tissue Eng. Part C Methods 18: 772–784.
Tauchi, H., C. Imashiro, T. Kuribara, G. Fujii, Y. Kurashina, K. Totani, and K. Takemura (2019) Effective and intact cell detachment from a clinically ubiquitous culture flask by combining ultrasonic wave exposure and diluted trypsin. Biotechnol. Bioprocess Eng. 24: 536–543.
Fukuma, Y., T. Inui, C. Imashiro, Y. Kurashina, and K. Takemura (2020) Homogenization of initial cell distribution by secondary flow of medium improves cell culture efficiency. PLoS One 15:e0235827.
Hung, P. J., P. J. Lee, P. Sabounchi, N. Aghdam, R. Lin, and L. P. Lee (2005) A novel high aspect ratio microfluidic design to provide a stable and uniform microenvironment for cell growth in a high throughput mammalian cell culture array. Lab. Chip. 5: 44–48.
Yamashita, T., P. Kollmannsberger, K. Mawatari, T. Kitamori, and V. Vogel (2016) Cell sheet mechanics: how geometrical constraints induce the detachment of cell sheets from concave surfaces. Acta Biomater. 45: 85–97.
Sakaguchi, K., T. Shimizu, S. Horaguchi, H. Sekine, M. Yamato, M. Umezu, and T. Okano (2013) In vitro engineering of vascularized tissue surrogates. Sci. Rep. 3: 1316.
Carter, S. S. D., L. Barbe, M. Tenje, and G. Mestres (2020) Exploring microfluidics as a tool to evaluate the biological properties of a titanium alloy under dynamic conditions. Biomater. Sci. 8: 6309–6321.
Time to revise the Sustainable Development Goals. (2020) Nature 583: 331–332.
Sachs, J., G. Schmidt-Traub, and G. Lafortune (2020) Speaking truth to power about the SDGs. Nature 584: 344.
Wu, X., S. Liu, K. Chen, F. Wang, C. Feng, L. Xu, and D. Zhang (2021) 3D printed chitosan-gelatine hydrogel coating on titanium alloy surface as biological fixation interface of artificial joint prosthesis. Int. J. Biol. Macromol. 182: 669–679.
Zhang, X., Y. Zhang, and Z. Jin (2022) A review of the biotribology of medical devices. Friction 10: 4–30.
Enomoto, J., N. Mochizuki, K. Ebisawa, T. Osaki, T. Kageyama, D. Myasnikova, T. Nittami, and J. Fukuda (2016) Engineering thick cell sheets by electrochemical desorption of oligopeptides on membrane substrates. Regen. Ther. 3: 24–31.
Imashiro, C., H. Takeshita, T. Morikura, S. Miyata, K. Takemura, and J. Komotori (2021) Development of accurate temperature regulation culture system with metallic culture vessel demonstrates different thermal cytotoxicity in cancer and normal cells. Sci. Rep. 11: 21466.
Imashiro, C., Y. Kurashina, and K. Takemura (2016) Cell patterning method using resonance vibration of a metallic cell cultivation substrate. Adv. Biomed. Eng. 5: 142–148.
Kurashina, Y., M. Hirano, C. Imashiro, K. Totani, J. Komotori, and K. Takemura (2017) Enzyme-free cell detachment mediated by resonance vibration with temperature modulation. Biotechnol. Bioeng. 114: 2279–2288.
Imashiro, C., Y. Ida, S. Miyata, and J. Komotori (2022) Titanium culture vessel capable of controlling culture temperature for evaluation of cell thermotolerance. Mater. Trans. 63: 373–378.
Buchanan, C. and L. Gardner (2019) Metal 3D printing in construction: a review of methods, research, applications, opportunities and challenges. Eng. Struct. 180: 332–348.
Bou-Ghannam, S., K. Kim, D. W. Grainger, and T. Okano (2021) 3D cell sheet structure augments mesenchymal stem cell cytokine production. Sci. Rep. 11: 8170.
Imashiro, C. and T. Shimizu (2021) Fundamental technologies and recent advances of cell-sheet-based tissue engineering. Int. J. Mol. Sci. 22: 425.
Choi, Y. S., S. E. Noh, S. M. Lim, and D. Kim (2010) Optimization of ex vivo hematopoietic stem cell expansion in intermittent dynamic cultures. Biotechnol. Lett. 32: 1969–1975.
Haraguchi, Y., T. Shimizu, T. Sasagawa, H. Sekine, K. Sakaguchi, T. Kikuchi, W. Sekine, S. Sekiya, M. Yamato, M. Umezu, and T. Okano (2012) Fabrication of functional three-dimensional tissues by stacking cell sheets in vitro. Nat. Protoc. 7: 850–858.
Heidemann, R., D. Lütkemeyer, H. Büntemeyer, and J. Lehmann (1998) Effects of dissolved oxygen levels and the role of extra- and intracellular amino acid concentrations upon the metabolism of mammalian cell lines during batch and continuous cultures. Cytotechnology 26: 185–197.
Bowler, E., S. Porazinski, S. Uzor, P. Thibault, M. Durand, E. Lapointe, K. M. A. Rouschop, J. Hancock, I. Wilson, and M. Ladomery (2018) Hypoxia leads to significant changes in alternative splicing and elevated expression of CLK splice factor kinases in PC3 prostate cancer cells. BMC Cancer 18: 355.
Tristan, C., N. Shahani, T. W. Sedlak, and A. Sawa (2011) The diverse functions of GAPDH: views from different subcellular compartments. Cell. Signal. 23: 317–323.
Colell, A., D. R. Green, and J. E. Ricci (2009) Novel roles for GAPDH in cell death and carcinogenesis. Cell Death Differ. 16: 1573–1581.
Corrêa-Giannella, M. L. and U. F. Machado (2013) SLC2A4gene: a promising target for pharmacogenomics of insulin resistance. Pharmacogenomics 14: 847–850.
Cairns, R. A., I. S. Harris, and T. W. Mak (2011) Regulation of cancer cell metabolism. Nat. Rev. Cancer 11: 85–95.
Imashiro, C., K. Yamasaki, R. Tanaka, Y. Tobe, K. Sakaguchi, and T. Shimizu (2021) Perfusable system using porous collagen gel scaffold actively provides fresh culture media to a cultured 3D tissue. Int. J. Mol. Sci. 22: 6780.
Coletti, C., M. J. Jaroszeski, A. Pallaoro, A. M. Hoff, S. Iannotta, and S. E. Saddow (2007) Biocompatibility and wettability of crystalline SiC and Si surfaces. Annu. Int. Conf. IEEE Eng. Med. Biol. Soc. 2007: 5850–5853.
Bonaventura, G., R. Iemmolo, V. La Cognata, M. Zimbone, F. La Via, M. E. Fragalà, M. L. Barcellona, R. Pellitteri, and S. Cavallaro (2019) Biocompatibility between silicon or silicon carbide surface and neural stem cells. Sci. Rep. 9: 11540.
Takahashi, H. and T. Okano (2015) Cell sheet-based tissue engineering for organizing anisotropic tissue constructs produced using microfabricated thermoresponsive substrates. Adv. Healthc. Mater. 4: 2388–2407.
Plutoni, C., E. Bazellieres, M. Le Borgne-Rochet, F. Comunale, A. Brugues, M. Séveno, D. Planchon, S. Thuault, N. Morin, S. Bodin, X. Trepat, and C. Gauthier-Rouvière (2016) P-cadherin promotes collective cell migration via a Cdc42-mediated increase in mechanical forces. J. Cell Biol. 212: 199–217.
Imashiro, C., M. Hirano, T. Morikura, Y. Fukuma, K. Ohnuma, Y. Kurashina, S. Miyata, and K. Takemura (2020) Detachment of cell sheets from clinically ubiquitous cell culture vessels by ultrasonic vibration. Sci. Rep. 10: 9468.
Kurashina, Y., C. Imashiro, M. Hirano, T. Kuribara, K. Totani, K. Ohnuma, J. Friend, and K. Takemura (2019) Enzyme-free release of adhered cells from standard culture dishes using intermittent ultrasonic traveling waves. Commun. Biol. 2: 393.
Kasai, K., Y. Kimura, and S. Miyata (2017) Improvement of adhesion and proliferation of mouse embryonic stem cells cultured on ozone/UV surface-modified substrates. Mater. Sci. Eng. C Mater. Biol. Appl. 78: 354–361.
Haraguchi, Y., Y. Kagawa, K. Sakaguchi, K. Matsuura, T. Shimizu, and T. Okano (2017) Thicker three-dimensional tissue from a “symbiotic recycling system” combining mammalian cells and algae. Sci. Rep. 7: 41594.
Castro Monsores, K. G., A. O. da Silva, S. de Sant’ Ana Oliveira, R. P. Weber, P. F. Filho, and S. N. Monteiro (2021) Influence of ultraviolet radiation on polystyrene. J. Mater. Res. Technol. 13: 359–365.
Zhao, X., W. Fu, X. Yang, and T. G. Langdon (2008) Microstructure and properties of pure titanium processed by equal-channel angular pressing at room temperature. Scr. Mater. 59: 542–545.
Takada, T., D. Sasaki, K. Matsuura, K. Miura, S. Sakamoto, H. Goto, T. Ohya, T. Iida, J. Homma, T. Shimizu, and N. Hagiwara (2022) Aligned human induced pluripotent stem cell-derived cardiac tissue improves contractile properties through promoting unidirectional and synchronous cardiomyocyte contraction. Biomaterials 281: 121351.
Acknowledgements
This work was supported by JSPS KAKENHI (20J00337). The authors gratefully acknowledge Dr. Takahiro G. Yamada (Department of Biosciences and Informatics, Keio University) for his assistance with the statistical analysis.
Author information
Authors and Affiliations
Contributions
Conceptualization: CI, JK, and KS. Data Curation: CI, SM, AE, and TS. Formal Analysis: CI, TM, and MH. Funding Acquisition: CI. Investigation: CI, TM, MH, and AE. Methodology: CI and KS. Project Administration: KS and TS. Resources: JK, SM, KS, and TS. Supervision: KS and TS. Validation: TS. Visualization: CI, TM, MH, and AE. Writing - Original Draft Preparation: CI, TM, MH, and AE. Writing - Review & Editing: JK, SM, KS, and TS
Corresponding authors
Ethics declarations
T.S. is a stockholder of CellSeed, Inc. Tokyo Women’s Medical University received research funds from CellSeed, Inc.
Neither ethical approval nor informed consent was required for this study.
Additional information
Publisher’s Note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Imashiro, C., Morikura, T., Hayama, M. et al. Metallic Vessel with Mesh Culture Surface Fabricated Using Three-dimensional Printing Engineers Tissue Culture Environment. Biotechnol Bioproc E 28, 181–191 (2023). https://doi.org/10.1007/s12257-022-0227-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12257-022-0227-1