Abstract
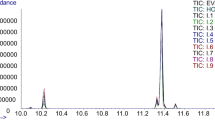
The composition of walnut oils is affected by multiple factors including their variety, their geographical location, and the environmental factors. In this study, the fatty acid composition of walnut oils from Greece and Bulgaria was assessed by gas chromatography coupled with flame ionization detection (GC-FID). The derivatization process involved the use of 14% BF3 after alkaline hydrolysis, and was optimized after investigating the effect of temperature (50–100℃) and methylation time (5–30 min). Twenty walnut oils originating from Greece and Bulgaria were analyzed, and the methyl esters of palmitic acid, stearic acid, oleic acid, linoleic acid, and linolenic acid were tentatively identified on the basis of their commercially available standards, and quantitatively determined. The method demonstrated good linearity (r2 > 0.999) and adequate recoveries (RE = 86.2–92.5%, n = 6). Precision was expressed as repeatability (intra-day precision, n = 3), and as reproducibility (inter-day precision (n = 9) and the results ranged between 2.7 and 4.6%RSD for intra-day experiments, and 4.1–5.8%RSD for inter-day experiments. The limits of detection (LODs) and limits of quantification (LOQs) ranged between 0.21–0.32 mg/kg and 0.63–0.97 mg/kg, respectively. The determined fatty acid methyl esters were quantified and the results were analyzed by Student’s t-test, principal component analysis (PCA), and hierarchical clustering to investigate the differences of the lipid fraction of the walnut oils according to their geographical origin.







Similar content being viewed by others
References
Aldai N, Murray BE, Nájera AI et al (2005) Derivatization of fatty acids and its application for conjugated linoleic acid studies in ruminant meat lipids. J. Sci. Food Agric.
Arena E, Campisi S, Fallico B, Maccarone E (2007) Distribution of fatty acids and phytosterols as a criterion to discriminate geographic origin of pistachio seeds. Food Chem 104:403–408. https://doi.org/10.1016/j.foodchem.2006.09.029
Bada JC, León-Camacho M, Prieto M et al (2010) Characterization of walnut oils (Juglans regia L.) from Asturias, Spain. JAOCS, J Am Oil Chem Soc 87:1469–1474. https://doi.org/10.1007/s11746-010-1629-3
Bertrand B, Villarreal D, Laffargue A et al (2008) Comparison of the effectiveness of fatty acids, chlorogenic acids, and elements for the chemometric discrimination of coffee (Coffea arabica L.) varieties and growing origins. J Agric Food Chem 56:2273–2280. https://doi.org/10.1021/jf073314f
Chiu HH, Kuo CH (2020) Gas chromatography-mass spectrometry-based analytical strategies for fatty acid analysis in biological samples. J Food Drug Anal 28:60–73. https://doi.org/10.1016/j.jfda.2019.10.003
Cittadini MC, Martín D, Gallo S et al (2020) Evaluation of hazelnut and walnut oil chemical traits from conventional cultivars and native genetic resources in a non-traditional crop environment from Argentina. Eur Food Res Technol 246:833–843. https://doi.org/10.1007/s00217-020-03453-8
Cossignani L, Blasi F, Simonetti MS, Montesano D (2018) Fatty acids and phytosterols to discriminate geographic origin of Lycium barbarum berry. Food Anal Methods 11:1180–1188. https://doi.org/10.1007/s12161-017-1098-5
Crews C, Hough P, Godward J et al (2005) Study of the main constituents of some authentic walnut oils. J Agric Food Chem 53:4853–4860. https://doi.org/10.1021/jf0478354
Cuadros-Rodríguez L, Ruiz-Samblás C, Valverde-Som L et al (2016) Chromatographic fingerprinting: an innovative approach for food “identitation” and food authentication - a tutorial. Anal Chim Acta 909:9–23. https://doi.org/10.1016/j.aca.2015.12.042
de Koning S, Janssen HG, van Deursen M, Brinkman UAT (2004) Automated on-line comprehensive two-dimensional LC x GC and LC x GC - ToF MS: instrument design and application to edible oil and fat analysis. J Sep Sci 27:397–409. https://doi.org/10.1002/jssc.200301676
Dogan M, Akgul A (2005) Fatty acid composition of some walnut (Juglans regia L.) cultivars from east Anatolia. Grasas Aceites 56:328–331. https://doi.org/10.3989/gya.2005.v56.i4.101
Dron J, Linke R, Rosenberg E, Schreiner M (2004) Trimethylsulfonium hydroxide as derivatization reagent for the chemical investigation of drying oils in works of art by gas chromatography. J Chromatogr A 1047:111–116. https://doi.org/10.1016/j.chroma.2004.06.013
Gallina-Toschi T, Cerretani L, Bendini A et al (2005) Oxidative stability and phenolic content of virgin olive oil: an analytical approach by traditional and high resolution techniques. J Sep Sci 28:859–870. https://doi.org/10.1002/jssc.200500044
Gandev SI, Arnaudov V, Serbezova D (2015) Selection and cultivation of a local wild walnut type in Bulgaria. Acta Hortic. https://doi.org/10.17660/ACTAHORTIC.2015.1074.20
Granato D, Santos JS, Escher GB et al (2018) Use of principal component analysis (PCA) and hierarchical cluster analysis (HCA) for multivariate association between bioactive compounds and functional properties in foods: a critical perspective. Trends Food Sci Technol 72:83–90. https://doi.org/10.1016/j.tifs.2017.12.006
Jaćimović V, Adakalić M, Ercisli S et al (2020) Fruit quality properties of walnut (Juglans regia l.) genetic resources in Montenegro. Sustain 12:1–19. https://doi.org/10.3390/su12239963
Jarukas L, Kuraite G, Baranauskaite J et al (2021) Optimization and validation of the GC/FID method for the quantification of fatty acids in bee products. Appl Sci 11:1–10. https://doi.org/10.3390/app11010083
Jollife IT, Cadima J (2016) Principal component analysis: a review and recent developments. Philos Trans R Soc A Math Phys Eng Sci 374. https://doi.org/10.1098/rsta.2015.0202
Kalogiouri NP, Samanidou VF (2020) Liquid chromatographic methods coupled to chemometrics: a short review to present the key workflow for the investigation of wine phenolic composition as it is affected by environmental factors. Environ Sci Pollut Res. https://doi.org/10.1007/s11356-020-09681-5
Kalogiouri NP, Alygizakis NA, Aalizadeh R, Thomaidis NS (2016) Olive oil authenticity studies by target and nontarget LC–QTOF-MS combined with advanced chemometric techniques. Anal Bioanal Chem 408:7955–7970. https://doi.org/10.1007/s00216-016-9891-3
Kalogiouri NP, Manousi N, Rosenberg E et al (2020) Advances in the chromatographic separation and determination of bioactive compounds for assessing the nutrient profile of nuts. Curr Anal Chem 16:1–17. https://doi.org/10.2174/1573411016999200729111951
Lavedrine F, Zmirou D, Ravel A et al (1999) Blood cholesterol and walnut consumption: a cross-sectional survey in France. Prev Med (baltim) 28:333–339. https://doi.org/10.1006/pmed.1999.0460
Mikrou T, Pantelidou E, Parasyri N et al (2020) Varietal and geographical discrimination of Greek monovarietal extra virgin olive oils based on squalene, tocopherol, and fatty acid composition. Molecules 25. https://doi.org/10.3390/molecules25173818
Mota MFS, Waktola HD, Nolvachai Y, Marriott PJ (2021) Gas chromatography - mass spectrometry for characterisation, assessment of quality and authentication of seed and vegetable oils. TrAC - Trends Anal Chem 138:116238. https://doi.org/10.1016/j.trac.2021.116238
Pang Z, Chong J, Zhou G et al (2021) MetaboAnalyst 5.0: narrowing the gap between raw spectra and functional insights. Nucleic Acids Res 49:388–396. https://doi.org/10.1093/nar/gkab382
Park YW, Chang PS, Lee JH (2010) Application of triacylglycerol and fatty acid analyses to discriminate blended sesame oil with soybean oil. Food Chem 123:377–383. https://doi.org/10.1016/j.foodchem.2010.04.049
Patterson E, Wall R, Fitzgerald GF et al (2012) Health implications of high dietary omega-6 polyunsaturated fatty acids. J Nutr Metab 1–16. https://doi.org/10.1155/2012/539426
Pla M, Hernández P, Ariño B et al (2007) Prediction of fatty acid content in rabbit meat and discrimination between conventional and organic production systems by NIRS methodology. Food Chem 100:165–170. https://doi.org/10.1016/j.foodchem.2005.09.029
Poggetti L, Ferfuia C, Chiabà C, et al (2018) Kernel oil content and oil composition in walnut (Juglans regia L.) accessions from north-eastern Italy. J Sci Food Agric. https://doi.org/10.1002/jsfa.8542
Rivera-Rangel LR, Aguilera-Campos KI, García-Triana A et al (2018) Comparison of oil content and fatty acids profile of Western Schley, Wichita, and native pecan nuts cultured in Chihuahua, Mexico. J Lipids 2018:1–6. https://doi.org/10.1155/2018/4781345
Savage GP, Dutta PC, McNeil DL (1999) Fatty acid and tocopherol contents and oxidative stability of walnut oils. JAOCS, J Am Oil Chem Soc 76:1059–1063. https://doi.org/10.1007/s11746-999-0204-2
Tranchida PQ, Giannino A, Mondello M et al (2008) Elucidation of fatty acid profiles in vegetable oils exploiting group-type patterning and enhanced sensitivity of comprehensive two-dimensional gas chromatography. J Sep Sci 31:1797–1802. https://doi.org/10.1002/jssc.200800002
Valianou L, Wei S, Mubarak MS et al (2011) Identification of organic materials in icons of the Cretan School of iconography. J Archaeol Sci 38:246–254. https://doi.org/10.1016/j.jas.2010.08.025
Verardo V, Bendini A, Cerretani L et al (2009) Capillary gas chromatography analysis of lipid composition and evaluation of phenolic compounds by micellar electrokinetic chromatography in italian walnut (juglans regia l.): irrigation and fertilization influence. J Food Qual 32:262–281. https://doi.org/10.1111/j.1745-4557.2009.00249.x
Wilks DS (2011) Cluster Analysis Int Geophys 100:603–616. https://doi.org/10.1016/B978-0-12-385022-5.00015-4
Zhang M, Yang X, Zhao HT et al (2015) A quick method for routine analysis of C18 trans fatty acids in non-hydrogenated edible vegetable oils by gas chromatography-mass spectrometry. Food Control 57:293–301. https://doi.org/10.1016/j.foodcont.2015.04.027
Zhou S, Xu Z, Liu F (2017) Method for determining the optimal number of clusters based on agglomerative hierarchical clustering. IEEE Trans Neural Networks Learn Syst 28:3007–3017. https://doi.org/10.1109/TNNLS.2016.2608001
Funding
This research was co-financed by Greece and the European Union (European Social Fund, ESF) through the Operational Programme “Human Resources Development, Education and Lifelong Learning” in the context of the project “Reinforcement of Postdoctoral Researchers—2nd Cycle” (MIS- 5033021), implemented by the State Scholarships Foundation (IKY) with grant no. 2019–050-0503–17749.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical Approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Informed Consent
Not applicable.
Conflict of Interest
Natasa P. Kalogiouri declares that there is no conflict of interest. Natalia Manousi declares that there is no conflict of interest. Ioannis Mourtzinos declares that there is no conflict of interest. Erwin Rosenberg declares that there is no conflict of interest. George A. Zachariadis declares that there is no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kalogiouri, N.P., Manousi, N., Mourtzinos, I. et al. A Rapid GC-FID Method for the Determination of Fatty Acids in Walnut Oils and Their Use as Markers in Authenticity Studies. Food Anal. Methods 15, 761–771 (2022). https://doi.org/10.1007/s12161-021-02157-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12161-021-02157-3