Abstract
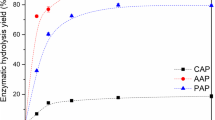
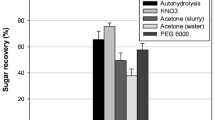
This a two-part study was conducted to improve the enzymatic saccharification of apple pomace (AP) by pretreatment and fed-batch enzymatic hydrolysis. The impact on the enzymatic hydrolysis and structural characterization in AP of four pretreatment processes, sodium hydroxide (SH), ethanol (ETH), liquid hot water (LHW), and Phanerochaete chrysosporium (PC), were evaluated. In addition, the enzymatic hydrolysis efficiency of AP at high solids content was improved by optimizing the enzyme hydrolysis conditions and using fed-batch strategy. The results demonstrated that both ETH 160 °C and PC pretreatments effectively enhanced the efficiency of enzymatic saccharification in AP while disrupting its compact structure. Notably, PC solid-state fermentation for 7 d proved to be a mild yet effective pretreatment method for AP. By utilizing PC pretreated residue at a substrate content of 20% (w/v) with an addition of 5 mg/g pectinase and 50 mg/g Tween 80, along with employing fed-batch enzymatic hydrolysis, the total sugar concentration increased by 30.3% after 72 h. At this point, the highest total sugar concentration reached 77.5 g/L with an enzymatic hydrolysis yield reaching 73% (glucose). This study presents not only the first application of PC as a pretreatment method for AP but also establishes a new enzymatic process with high solids content to provide a new strategy for AP resource utilization.






Similar content being viewed by others
Data Availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
Change history
02 February 2024
The original version of this paper was updated: The number of Equation (12) was overlapping the entry of Table 1
Abbreviations
- AP:
-
Apple pomace
- SH:
-
Sodium hydroxide
- ETH:
-
Ethanol
- LHW:
-
Liquid hot water
- PC:
-
Phanerochaete chrysosporium
- SEM:
-
Scanning electron microscopy
- FTIR:
-
Fourier-transform infrared spectroscopy
- XRD:
-
X-ray diffractometry
- TGA:
-
Thermogravimetric analysis
- FPU:
-
Filter paper units
- HMF:
-
5-hydroxymethylfurfural
- H+ :
-
Hydrogen ion
- OH− :
-
Hydroxyl ion
- CO2 :
-
Carbon dioxide
References
Jing Y, Guo Y, Xia Q, Liu X, Wang Y (2019) Catalytic production of value-added chemicals and liquid fuels from lignocellulosic biomass. Chem 5(10):2520–2546. https://doi.org/10.1016/j.chempr.2019.05.022
Kawaguchi H, Hasunuma T, Ogino C, Kondo A (2016) Bioprocessing of bio-based chemicals produced from lignocellulosic feedstocks. Curr Opin Biotechnol 42:30–39. https://doi.org/10.1016/j.copbio.2016.02.031
FAO U (2019) The state of food and agriculture: Moving forward on food loss and waste reduction. In: FAO, Global Rome, Italy
Narasimmalu A, Ramasamy R (2020) Food processing industry waste and circular economy. IOP Conf Ser Mater Sci Eng 955(1):012089. https://doi.org/10.1088/1757-899X/955/1/012089
Zhang F, Wang T, Wang X, Lu X (2021) Apple pomace as a potential valuable resource for full-components utilization: A review. J Clean Prod 329:129676. https://doi.org/10.1016/j.jclepro.2021.129676
Borujeni NE, Karimi K, Denayer JFM, Kumar R (2022) Apple pomace biorefinery for ethanol, mycoprotein, and value-added biochemicals production by Mucor indicus. Energy 240:122469. https://doi.org/10.1016/j.energy.2021.122469
Borujeni NE, Alavijeh MK, Denayer JFM, Karimi K (2023) A novel integrated biorefinery approach for apple pomace valorization with significant socioeconomic benefits. Renew Energy 208:275–286. https://doi.org/10.1016/j.renene.2023.03.056
Maria HV, Ana PGI, Rebeca DA (2017) Biobutanol production from apple pomace: the importance of pretreatment methods on the fermentability of lignocellulosic agro-food wastes. Appl Microbiol Biotechnol 101(21):8041–8052. https://doi.org/10.1007/s00253-017-8522-z
Kamil P, Edyta L, Elzbieta HS (2016) Possibility of using apple pomaces in the process of propionic-acetic fermentation. Electron J Biotechnol 23:1–6. https://doi.org/10.1016/j.ejbt.2016.07.004
Vashisht A, Thakur K, Kauldhar BS, Kumar V, Yadav SK (2019) Waste valorization: Identification of an ethanol tolerant bacterium Acetobacter pasteurianus SKYAA25 for acetic acid production from apple pomace. Sci Total Environ 690:956–964. https://doi.org/10.1016/j.scitotenv.2019.07.070
Parmar I, Rupasinghe HPV (2013) Bio-conversion of apple pomace into ethanol and acetic acid: Enzymatic hydrolysis and fermentation. Bioresour Technol 130:613–620. https://doi.org/10.1016/j.biortech.2012.12.084
Wagle A, Angove MJ, Mahara A, Wagle A, Mainali B, Martins M, Goldbeck R, Paudel SR (2022) Multi-stage pre-treatment of lignocellulosic biomass for multi-product biorefinery: A review. Sustain Energ Technol Assessm 49:101702. https://doi.org/10.1016/j.seta.2021.101702
Mankar AR, Pandey A, Modak A, Pant KK (2021) Pretreatment of lignocellulosic biomass: A review on recent advances. Bioresour Technol 334:125235. https://doi.org/10.1016/j.biortech.2021.125235
Chen WH, Nizetic S, Sirohi R, Huang Z, Luque R, Papadopoulos AM, Sakthivel R, Nguyen XP, Hoang AT (2022) Liquid hot water as sustainable biomass pretreatment technique for bioenergy production: A review. Bioresour Technol 344:126207. https://doi.org/10.1016/j.biortech.2021.126207
Sun Z, Mao Y, Liu S, Zhang H, Xu Y, Geng R, Dong Q (2022) Effect of pretreatment with Phanerochaete chrysosporium on physicochemical properties and pyrolysis behaviors of corn stover. Bioresour Technol 361:127687. https://doi.org/10.1016/j.biortech.2022.127687
Hu M, Cai Z, Zhang J, Yuan L, Fu Q, Ji D (2022) Ammonia fiber expansion (AFEX) combined with white-rot fungi pretreatment to improve enzymatic hydrolysis of lignocellulose. Biomass Convers Biorefin 2190–6823. https://doi.org/10.1007/s13399-022-03075-y
Rouches E, Herpoel-Gimbert I, Steyer JP, Carrere H (2016) Improvement of anaerobic degradation by white-rot fungi pretreatment of lignocellulosic biomass: A review. Renew Sustain Energy Rev 59:179–198. https://doi.org/10.1016/j.rser.2015.12.317
Wittner N, Broos W, Bauwelinck J, Slezsák J, Vlaeminck SE, Cornet I (2021) Enhanced fungal delignification and enzymatic digestibility of poplar wood by combined CuSO4 and MnSO4 supplementation. Process Biochem 108:129–137. https://doi.org/10.1016/j.procbio.2021.06.002
Zhang H, Wang Y, Yang R, Yan Z, Zhao Z, Du C, Chang C (2023) Comparative study on the mild pretreatment processes for enzymatic hydrolysis of wheat straw with high-solid loading. Biomass Convers Biorefin 2190–6823. https://doi.org/10.1007/s13399-023-04319-1
da Silva AS, Espinheira RP, Teixeira RSS, de Souza MF, Ferreira-Leitao V, Bon EPS (2020) Constraints and advances in high-solids enzymatic hydrolysis of lignocellulosic biomass: a critical review. Biotechnol Biofuels 13:58. https://doi.org/10.1186/s13068-020-01697-w
Modenbach AA, Nokes SE (2013) Enzymatic hydrolysis of biomass at high-solids loadings – A review. Biom Bioenergy 56:526–544. https://doi.org/10.1016/j.biombioe.2013.05.031
Zanuso E, Ruiz HA, Domingues L, Teixeira JA (2022) Oscillatory flow bioreactor operating at high solids loading for enzymatic hydrolysis of lignocellulosic biomass. Biochem Eng J 187:108632. https://doi.org/10.1016/j.bej.2022.108632
Pino MS, Rodríguez-Jasso RM, Michelin M, Flores-Gallegos AC, Morales-Rodriguez R, Teixeira JA, Ruiz HA (2018) Bioreactor design for enzymatic hydrolysis of biomass under the biorefinery concept. Chem Eng J 347:119–136. https://doi.org/10.1016/j.cej.2018.04.057
Xu C, Zhang J, Zhang Y, Guo Y, Xu H, Xu J, Wang Z (2019) Enhancement of high-solids enzymatic hydrolysis efficiency of alkali pretreated sugarcane bagasse at low cellulase dosage by fed-batch strategy based on optimized accessory enzymes and additives. Bioresour Technol 292:121993. https://doi.org/10.1016/j.biortech.2019.121993
Xu C, Xiong Y, Zhang J, Li K, Zhong S, Huang S, Peng Y (2023) Liquid hot water pretreatment combined with high-solids enzymatic hydrolysis and fed-batch fermentation for succinic acid sustainable processed from sugarcane bagasse. Bioresour Technol 369:128389. https://doi.org/10.1016/j.biortech.2022.128389
Zhang J, Li K, Liu S, Huang S, Xu C (2022) Alkaline hydrogen peroxide pretreatment combined with bio-additives to boost high-solids enzymatic hydrolysis of sugarcane bagasse for succinic acid processing. Bioresour Technol 345:126550. https://doi.org/10.1016/j.biortech.2021.126550
Hu J, Chandra R, Arantes V, Gourlay K, Dyk JSV, Saddler JN (2015) The addition of accessory enzymes enhances the hydrolytic performance of cellulase enzymes at high solid loadings. Bioresour Technol 186:149–153. https://doi.org/10.1016/j.biortech.2015.03.055
Mukasekuru MR, Kaneza P, Sun H, Sun FF, He J, Zheng P (2020) Fed-batch high-solids enzymatic saccharification of lignocellulosic substrates with a combination of additives and accessory enzymes. Ind Crops Prod 146:112156. https://doi.org/10.1016/j.indcrop.2020.112156
Cheng L, Hu X, Hong Y, Li Z, Li C, Gu Z (2023) Reduction of inhibitory effects on cellulose degradation with pectinase treatment in potato residue. Ind Crops Prod 192:116010. https://doi.org/10.1016/j.indcrop.2022.116010
Gong Z, Yang G, Song J, Zheng P, Liu J, Zhu W, Shuai L (2021) Understanding the promoting effect of non-catalytic protein on enzymatic hydrolysis efficiency of lignocelluloses. Bioresour Bioprocess 8(1):9. https://doi.org/10.1186/s40643-021-00363-9
Jiang F, Qian C, Esker AR, Roman M (2017) Effect of nonionic surfactants on dispersion and polar interactions in the adsorption of cellulases onto lignin. J Phys Chem B 121(41):9607–9620. https://doi.org/10.1021/acs.jpcb.7b07716
Chen J, Yang S, Alam MA, Wang Z, Zhang J, Huang S, Xu J (2021) Novel biorefining method for succinic acid processed from sugarcane bagasse. Bioresour Technol 324:124615. https://doi.org/10.1016/j.biortech.2020.124615
Zheng A, Zhao K, Li L, Zhao Z, Jiang L, Huang Z, Li H (2018) Quantitative comparison of different chemical pretreatment methods on chemical structure and pyrolysis characteristics of corncobs. J Energy Inst 91(5):676–682. https://doi.org/10.1016/j.joei.2017.06.002
Segal L, Creely JJ, Martin AE, Conrad CM (1959) An empirical method for estimating the degree of crystallinity of native cellulose using the x-ray diffractometer. Text Res J 29(10):786–794. https://doi.org/10.1177/004051755902901003
Sluiter A, Hames B, Ruiz R, Scarlata C, Sluiter J, Templeton D, Crocker D (2012) Determination of structural carbohydrates and lignin in biomass. National Renewable Energy Laboratory, USA
Chen Y, Yang H, Zou H, Sun T, Li M, Zhai J, Tang WZ (2020) Effects of acid/alkali pretreatments on lignocellulosic biomass mono-digestion and its co-digestion with waste activated sludge. J Clean Prod 277:123998. https://doi.org/10.1016/j.jclepro.2020.123998
Chen JH, Xu JK, Huang PL, Sun RC (2016) Effect of alkaline pretreatment on the preparation of regenerated lignocellulose fibers from bamboo stem. Cellulose 23(4):2727–2739. https://doi.org/10.1007/s10570-016-0983-1
Shi J, Lu Y, Zhang Y, Cai L, Shi SQ (2018) Effect of thermal treatment with water, H2SO4 and NaOH aqueous solution on color, cell wall and chemical structure of poplar wood. Sci Rep 8(1):17735. https://doi.org/10.1038/s41598-018-36086-9
Xie X, Chen M, Tong W, Song K, Wang J, Wu S, Chu Q (2023) Comparative study of acid- and alkali-catalyzed 1,4-butanediol pretreatment for co-production of fermentable sugars and value-added lignin compounds. Biotechnol Biofuels Bioprod 16(1):52. https://doi.org/10.1186/s13068-023-02303-5
Ferreira JA, Taherzadeh MJ (2020) Improving the economy of lignocellulose-based biorefineries with organosolv pretreatment. Bioresour Technol 299:122695. https://doi.org/10.1016/j.biortech.2019.122695
Choi JH, Jang SK, Kim JH, Park SY, Kim JC, Jeong H, Choi IG (2019) Simultaneous production of glucose, furfural, and ethanol organosolv lignin for total utilization of high recalcitrant biomass by organosolv pretreatment. Renew Energy 130:952–960. https://doi.org/10.1016/j.renene.2018.05.052
Shang G, Zhang C, Wang F, Qiu L, Guo X, Xu F (2019) Liquid hot water pretreatment to enhance the anaerobic digestion of wheat straw—effects of temperature and retention time. Environ Sci Pollut Res Int 26(28):29424–29434. https://doi.org/10.1007/s11356-019-06111-z
Li HQ, Li CL, Sang T, Xu J (2013) Pretreatment on Miscanthus lutarioriparious by liquid hot water for efficient ethanol production. Biotechnol Biofuels 6(1):76. https://doi.org/10.1186/1754-6834-6-76
Sabanci K, Buyukkileci AO (2018) Comparison of liquid hot water, very dilute acid and alkali treatments for enhancing enzymatic digestibility of hazelnut tree pruning residues. Bioresour Technol 261:158–165. https://doi.org/10.1016/j.biortech.2018.03.136
Elisashvili V, Kachlishvili E, Penninckx, (2008) Effect of growth substrate, method of fermentation, and nitrogen source on lignocellulose-degrading enzymes production by white-rot basidiomycetes. J Ind Microbiol Biotechnol 35(11):1531–1538. https://doi.org/10.1007/s10295-008-0454-2
Martins JR, Schmatz AA, Salazar-Bryan AM, Brienzo M (2022) Effect of dilute acid pretreatment on the sugarcane leaf for fermentable sugars production. Sugar Tech 24(5):1540–1550. https://doi.org/10.1007/s12355-021-01106-y
Liu G, Han D, Yang S (2022) Combinations of mild chemical and bacterial pretreatment for improving enzymatic saccharification of corn stover. Biotechnol Biotechnol Equip 36(1):598–608. https://doi.org/10.1080/13102818.2022.2112910
Karimi K, Taherzadeh MJ (2016) A critical review of analytical methods in pretreatment of lignocelluloses: Composition, imaging, and crystallinity. Bioresour Technol 200:1008–1018. https://doi.org/10.1016/j.biortech.2015.11.022
Peng H, Gao L, Li M, Shen Y, Qian Q, Li X (2014) Steam explosion-ionic liquid pretreatments on wetland lignocellulosic biomasses of Phragmites (sp) and Thalia dealbata for Bio H2 conversion. RSC Adv 4(69):36603–36614. https://doi.org/10.1039/C4RA06739H
Reis CER, Junior NL, Bento HBS, de Carvalho AKF, de Souza Vandenberghe LP, Soccol CR, Chandel AK (2023) Process strategies to reduce cellulase enzyme loading for renewable sugar production in biorefineries. Chem Eng J 451:138690. https://doi.org/10.1016/j.cej.2022.138690
kuo ch, huang cy, shieh cj, wang hmd, tseng cy (2017) hydrolysis of orange Peel with Cellulase and Pectinase to Produce Bacterial Cellulose using Gluconacetobacter xylinus. Waste Biomass Valoriz 10(1):85–93. https://doi.org/10.1007/s12649-017-0034-7
Zhang H, Chen W, Han X, Zeng Y, Zhang J, Gao Z, Xie J (2021) Intensification of sugar production by using Tween 80 to enhance metal-salt catalyzed pretreatment and enzymatic hydrolysis of sugarcane bagasse. Bioresour Technol 339:125522. https://doi.org/10.1016/j.biortech.2021.125522
Hernández-Beltrán JU, Fontalvo J, Hernández-Escoto H (2021) Fed-batch enzymatic hydrolysis of plantain pseudostem to fermentable sugars production and the impact of particle size at high solids loadings. Biomass Convers Biorefin 11(6):2975–2982. https://doi.org/10.1007/s13399-020-00669-2
Gao W, Li Z, Liu T, Wang Y (2021) Production of high-concentration fermentable sugars from lignocellulosic biomass by using high solids fed-batch enzymatic hydrolysis. Biochem Eng J 176:108186. https://doi.org/10.1016/j.bej.2021.108186
Magyar M, Sousa LDC, Jin M, Sarks C, Balan V (2016) Conversion of apple pomace waste to ethanol at industrial relevant conditions. Appl Microbiol Biotechnol 100(16):7349–7358. https://doi.org/10.1007/s00253-016-7665-7
Gama R, Van Dyk JS, Pletschke BI (2015) Optimisation of enzymatic hydrolysis of apple pomace for production of biofuel and biorefinery chemicals using commercial enzymes. 3 Biotech 5(6):1075–1087. https://doi.org/10.1007/s13205-015-0312-7
Shiva FCB, Rosa MRJ, Rajeev KS, Hector AR (2022) High-solids loading processing for an integrated lignocellulosic biorefinery: Effects of transport phenomena and rheology – A review. Bioresour Technol 351:127044. https://doi.org/10.1016/j.biortech.2022.127044
Gomes MG, Gurgel LVA, Baffi MA, Pasquini D (2020) Pretreatment of sugarcane bagasse using citric acid and its use in enzymatic hydrolysis. Renew Energ 157:332–341. https://doi.org/10.1016/j.renene.2020.05.002
Acknowledgements
The authors would like to thank the Henan Province Science and Technology Development Plan Project for financial support, and secondly, the research platform of the College of Life Sciences, Henan Normal University for technical support.
Funding
This work was supported by the Henan Province Science and Technology Development Plan Project in China [grant numbers 212102110359 and 222102110362].
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Jianjun Chen: Conceptualization, Methodology, Funding acquisition, and manuscript draft preparation; Jiameng Zhou: Methodology and manuscript draft preparation; Rongjie Yuan: Software; Xue Shao: Data curation; Yaoyajie Lu: Visualization and Validation; Weidi Sun: Project administration. Xianglin Cao: Conceptualization, Resources, Data curation, Writing-review & editing.
Corresponding author
Ethics declarations
Competing Interests
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chen, J., Zhou, J., Yuan, R. et al. Mild Pretreatment Combined with Fed-Batch Strategy to Improve the Enzymatic Efficiency of Apple Pomace at High-Solids Content. Bioenerg. Res. (2024). https://doi.org/10.1007/s12155-024-10719-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12155-024-10719-6