Abstract
Purpose
Nowadays, the oxidative phosphorylation (OXPHOS) correlated with leukemogenesis and treatment response is extensive. Thus, exploration of novel approaches in disrupting OXPHOS in AML is urgently needed.
Materials and methods
Bioinformatical analysis of TCGA AML dataset was performed to identify the molecular signaling of OXPHOS. The OXPHOS level was measured through a Seahorse XFe96 cell metabolic analyzer. Flow cytometry was applied to measure mitochondrial status. Real-time qPCR and western blot were used to analyze the expression of mitochondrial or inflammatory factors. MLL-AF9-induced leukemic mice were conducted to measure the anti-leukemia effect of chidamide.
Results
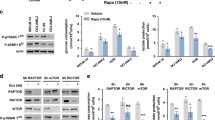
Here, we reported that AML patients with high OXPHOS level were in a poor prognosis, which was associated with high expression of HDAC1/3 (TCGA). Inhibition of HDAC1/3 by chidamide inhibited cell proliferation and induced apoptotic cell death in AML cells. Intriguingly, chidamide could disrupt mitochondrial OXPHOS as assessed by inducing mitochondrial superoxide and reducing oxygen consumption rate, as well as decreasing mitochondrial ATP production. We also observed that chidamide augmented HK1 expression, while glycolysis inhibitor 2-DG could reduce the elevation of HK1 and improve the sensitivity of AML cells exposed to chidamide. Furthermore, HDAC3 was correlated with hyperinflammatory status, while chidamide could downregulate the inflammatory signaling in AML. Notably, chidamide eradicated leukemic cells in vivo and prolonged the survival time of MLL-AF9-induced AML mice.
Conclusion
Chidamide disrupted mitochondrial OXPHOS, promoted cell apoptosis and reduced inflammation in AML cells. These findings exhibited a novel mechanism that targeting OXPHOS would be a novel strategy for AML treatment.









Similar content being viewed by others
Data availability
The data presented in this study are available on request from the corresponding author.
References
de Beauchamp L, Himonas E, Helgason GV. Mitochondrial metabolism as a potential therapeutic target in myeloid leukaemia. Leukemia. 2022;36(1):1–12.
Amaya ML, Inguva A, Pei S, Jones C, Krug A, Ye H, et al. The STAT3-MYC axis promotes survival of leukemia stem cells by regulating SLC1A5 and oxidative phosphorylation. Blood. 2022;139(4):584–96.
Lagadinou ED, Sach A, Callahan K, Rossi RM, Neering SJ, Minhajuddin M, et al. BCL-2 inhibition targets oxidative phosphorylation and selectively eradicates quiescent human leukemia stem cells. Cell Stem Cell. 2013;12(3):329–41.
Pei S, Minhajuddin M, Adane B, Khan N, Stevens BM, Mack SC, et al. AMPK/FIS1-mediated mitophagy Is required for self-renewal of human AML stem cells. Cell Stem Cell. 2018;23(1):86-100.e6.
Khan DH, Mullokandov M, Wu Y, Voisin V, Gronda M, Hurren R, et al. Mitochondrial carrier homolog 2 is necessary for AML survival. Blood. 2020;136(1):81–92.
Ma J, Liu B, Yu D, Zuo Y, Cai R, Yang J, et al. SIRT3 deacetylase activity confers chemoresistance in AML via regulation of mitochondrial oxidative phosphorylation. Br J Haematol. 2019;187(1):49–64.
Cheng Y, He C, Wang M, Ma X, Mo F, Yang S, et al. Targeting epigenetic regulators for cancer therapy: mechanisms and advances in clinical trials. Signal Transduct Target Ther. 2019;4:62.
Sun Y, Hong JH, Ning Z, Pan D, Fu X, Lu X, et al. Therapeutic potential of tucidinostat, a subtype-selective HDAC inhibitor, in cancer treatment. Front Pharmacol. 2022;13: 932914.
Gui L, Cao J, Ji D, Zhang H, Fan Q, Zhu J, et al. Chidamide combined with cyclophosphamide, doxorubicin, vincristine and prednisone in previously untreated patients with peripheral T-cell lymphoma. Chin J Cancer Res. 2021;33(5):616–26.
Zhong M, Lin F, Jiang Y, Pan G, Tan J, Zhou H, et al. Therapeutic interaction of Apatinib and Chidamide in T-Cell acute lymphoblastic Leukemia through interference with mitochondria associated biogenesis and intrinsic apoptosis. J Pers Med. 2021;11(10):977.
Chen K, Yang Q, Zha J, Deng M, Zhou Y, Fu G, et al. Preclinical evaluation of a regimen combining chidamide and ABT-199 in acute myeloid leukemia. Cell Death Dis. 2020;11(9):778.
Wang H, Liu YC, Zhu CY, Yan F, Wang MZ, Chen XS, et al. Chidamide increases the sensitivity of refractory or relapsed acute myeloid leukemia cells to anthracyclines via regulation of the HDAC3 -AKT-P21-CDK2 signaling pathway. J Exp Clin Cancer Res. 2020;39(1):278.
Li G, Li D, Yuan F, Cheng C, Chen L, Wei X. Synergistic effect of chidamide and venetoclax on apoptosis in acute myeloid leukemia cells and its mechanism. Ann Transl Med. 2021;9(20):1575.
Yu G, Wang LG, Han Y, He QY. clusterProfiler: an R package for comparing biological themes among gene clusters. OMICS. 2012;16(5):284–7.
Aran D, Hu Z, Butte AJ. xCell: digitally portraying the tissue cellular heterogeneity landscape. Genome Biol. 2017;18(1):220.
Mills EL, Kelly B, O’Neill LAJ. Mitochondria are the powerhouses of immunity. Nat Immunol. 2017;18(5):488–98.
Molina JR, Sun Y, Protopopova M, Gera S, Bandi M, Bristow C, et al. An inhibitor of oxidative phosphorylation exploits cancer vulnerability. Nat Med. 2018;24(7):1036–46.
Minucci S, Pelicci PG. Histone deacetylase inhibitors and the promise of epigenetic (and more) treatments for cancer. Nat Rev Cancer. 2006;6(1):38–51.
Bhaskara S, Knutson SK, Jiang G, Chandrasekharan MB, Wilson AJ, Zheng S, et al. Hdac3 is essential for the maintenance of chromatin structure and genome stability. Cancer Cell. 2010;18(5):436–47.
Long J, Fang WY, Chang L, Gao WH, Shen Y, Jia MY, et al. Targeting HDAC3, a new partner protein of AKT in the reversal of chemoresistance in acute myeloid leukemia via DNA damage response. Leukemia. 2017;31(12):2761–70.
Guo C, Li J, Steinauer N, Wong M, Wu B, Dickson A, et al. Histone deacetylase 3 preferentially binds and collaborates with the transcription factor RUNX1 to repress AML1-ETO-dependent transcription in t(8;21) AML. J Biol Chem. 2020;295(13):4212–23.
Beyer M, Romanski A, Mustafa AM, Pons M, Buchler I, Vogel A, et al. HDAC3 Activity is essential for human leukemic cell growth and the expression of beta-catenin, MYC, and WT1. Cancers (Basel). 2019;11(10):1436.
Chi Z, Chen S, Xu T, Zhen W, Yu W, Jiang D, et al. Histone deacetylase 3 couples mitochondria to drive IL-1beta-dependent inflammation by configuring fatty acid oxidation. Mol Cell. 2020;80(1):43-58.e7.
Weinberg SE, Chandel NS. Targeting mitochondria metabolism for cancer therapy. Nat Chem Biol. 2015;11(1):9–15.
Sriskanthadevan S, Jeyaraju DV, Chung TE, Prabha S, Xu W, Skrtic M, et al. AML cells have low spare reserve capacity in their respiratory chain that renders them susceptible to oxidative metabolic stress. Blood. 2015;125(13):2120–30.
Xiong Y, Guan KL. Mechanistic insights into the regulation of metabolic enzymes by acetylation. J Cell Biol. 2012;198(2):155–64.
Wang C, Cui G, Wang D, Wang M, Chen Q, Wang Y, et al. Crosstalk of oxidative phosphorylation-related subtypes, establishment of a prognostic signature and immune infiltration characteristics in colorectal adenocarcinoma. Cancers (Basel). 2022;14(18):4503.
Chen W, Yang Z, Chen Y. A novel oxidative phosphorylation-associated gene signature for prognosis prediction in patients with hepatocellular carcinoma. Dis Markers. 2022;2022:3594901.
Luna-Yolba R, Marmoiton J, Gigo V, Marechal X, Boet E, Sahal A, et al. Disrupting mitochondrial electron transfer chain complex i decreases immune checkpoints in murine and human acute myeloid leukemic cells. Cancers (Basel). 2021;13(14):3499.
Wang J, Fang Y, Ma S, Su N, Zhang Y, Huang H, et al. Comparison of chidamide-contained treatment modalities versus chemotherapy in the second-line treatment for relapsed or refractory peripheral T-cell lymphoma. Leuk Res. 2021;111: 106705.
Li Y, Wang Y, Zhou Y, Li J, Chen K, Zhang L, et al. Cooperative effect of chidamide and chemotherapeutic drugs induce apoptosis by DNA damage accumulation and repair defects in acute myeloid leukemia stem and progenitor cells. Clin Epigenetics. 2017;9:83.
Wang L, Luo J, Chen G, Fang M, Wei X, Li Y, et al. Chidamide, decitabine, cytarabine, aclarubicin, and granulocyte colony-stimulating factor (CDCAG) in patients with relapsed/refractory acute myeloid leukemia: a single-arm, phase 1/2 study. Clin Epigenetics. 2020;12(1):132.
Acknowledgements
We thank Dr. Shou-Jie Wang from Center for Precision Medicine, SYSU Platform of Metabolomics for the assistance with Seahorse XF Cell Mito Stress experiment.
Funding
This study was supported by Talent Training Project of The Third Affiliated Hospital of Sun Yat-sen University.
Author information
Authors and Affiliations
Contributions
JYW and ZJL: conceived and designed the experiments. JDW and JQX: performed the experiments. JDW analyzed the data and wrote the article. All authors revised the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Informed consent
Not applicable.
Ethical approval and Research involving human participants and/or animals
The animal study was conducted with the approval of Institutional Animal Care and Use Committee.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
12094_2023_3079_MOESM1_ESM.tif
Supplementary file1 Effects of different metabolic inhibitors with chidamide. AML cells were treated chidamide (1 μM) or/and FCCP (1 µM/2 µM) (A, E), Oligomycin (1 µM/2 µM) (B, F), Rotenone (0.5 µM/1 µM) (C, G) or Metfoemin (5 mM/10 mM) (D, H) for 24 h and CCK-8 assay was applied to detect cell viability. Data were presented as mean ± SD, *p<0.05, **p<0.01, ***p<0.001 (TIF 9851 KB)
12094_2023_3079_MOESM2_ESM.tif
Supplementary file2 Relation between HDAC3 and immune cell infiltration is performed. (A) Immune cell enrichment was performed in HDAC3 high and low AML patients by ssGSEA. (B) Unsupervised clustering of immune checkpoint genes and infiltrating cells were analyzed in HDAC3 high and low groups. (C) Correlation between each infiltrating cell signature was assessed by spearman analysis. (D) Infiltrating cell infiltration abundance analysis in HDAC3 high and low groups was shown (TIF 32323 KB)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, JD., Xu, JQ., Long, ZJ. et al. Disruption of mitochondrial oxidative phosphorylation by chidamide eradicates leukemic cells in AML. Clin Transl Oncol 25, 1805–1820 (2023). https://doi.org/10.1007/s12094-023-03079-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12094-023-03079-8