Abstract
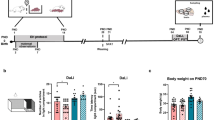
Major Depressive Disorder (MDD) is a severe and multifactorial psychiatric condition. Evidence has shown that environmental factors, such as stress, significantly explain MDD pathophysiology. Studies have hypothesized that changes in histone methylation patterns are involved in impaired glutamatergic signaling. Based on this scenario, this study aims to investigate histone 3 involvement in depression susceptibility or resilience in MDD pathophysiology by investigating cellular and molecular parameters related to i) glutamatergic neurotransmission, ii) astrocytic functioning, and iii) neurogenesis. For this, we subjected male Wistar rats to the Chronic Unpredictable Mild Stress (CUMS) model of depression. We propose that by evaluating the sucrose consumption, open field, and object recognition test performance from animals submitted to CUMS, it is possible to predict with high specificity rats with susceptibility to depressive-like phenotype and resilient to the depressive-like phenotype. We also demonstrated, for the first time, that patterns of H3K4me3, H3K9me3, H3K27me3, and H3K36me3 trimethylation are strictly associated with the resilient or susceptible to depressive-like phenotype in a brain-region-specific manner. Additionally, susceptible animals have reduced DCx and GFAP and resilient animals present increase of AQP-4 immunoreactivity. Together, these results provide evidence that H3 trimethylations are related to the development of the resilient or susceptible to depressive-like phenotype, contributing to further advances in the pathophysiology of MDD and the discovery of mechanisms behind resilience.















Similar content being viewed by others
Data Availability
The datasets generated during and/or analysed during the current study are not publicly available but are available from the corresponding author on reasonable request.
References
Ferrari AJ, Charlson FJ, Norman RE et al (2013) Burden of depressive disorders by country, sex, age, and year: findings from the global burden of disease study 2010. PLoS Med 10:. https://doi.org/10.1371/journal.pmed.1001547
Yang L, Zhao Y, Wang Y et al (2015) The effects of psychological stress on depression. Curr Neuropharmacol 13:494–504
Kessler RC, Berglund P, Demler O et al (2003) The epidemiology of major depressive disorder. Evid Based Eye Care 4:186–187. https://doi.org/10.1097/00132578-200310000-00002
Malhi GS, Mann JJ (2018) Depression. Lancet 392:2299–2312. https://doi.org/10.1016/S0140-6736(18)31948-2
Nestler EJ, Barrot M, Dileone RJ et al (2002) Neurology of depression. Br Med J 3:263–264. https://doi.org/10.1136/bmj.3.5613.263
Tafet GE, Nemeroff CB (2016) The links between stress and depression: psychoneuroendocrinological, genetic, and environmental interactions. 77–88. https://doi.org/10.1176/appi.neuropsych.15030053
Bangasser DA, Valentino RJ (2014) Sex differences in stress-related psychiatric disorders: neurobiological perspectives. Front Neuroendocrinol 35:303–319. https://doi.org/10.1016/j.yfrne.2014.03.008
Hill MN, Hellemans KGC, Verma P et al (2012) Neurobiology of chronic mild stress: parallels to major depression. Neurosci Biobehav Rev 36:2085–2117. https://doi.org/10.1016/j.neubiorev.2012.07.001
Gold PW (2015) The organization of the stress system and its dysregulation in depressive illness. Mol Psychiatry 20:32–47. https://doi.org/10.1038/mp.2014.163
Radley J, Morilak D, Viau V, Campeau S (2016) Chronic stress and brain plasticity: mechanisms underlying adaptive and maladaptive changes and implications for stress-related CNS disorders. Physiol Behav 176:139–148. https://doi.org/10.1016/j.neubiorev.2015.06.018.Chronic
Arnau-Soler A, Macdonald-Dunlop E, Adams MJ et al (2019) Genome-wide by environment interaction studies of depressive symptoms and psychosocial stress in UK Biobank and Generation Scotland. Transl Psychiatry 9:. https://doi.org/10.1038/s41398-018-0360-y
Caspi A, Sugden K, Moffitt TE et al (2003) Influence of life stress on depression: moderation by a polymorphism in the 5-HTT gene. Science (80- ) 301:386–389. https://doi.org/10.1126/science.1083968
Kim JH, Lee JH, Lee IS et al (2017) Histone lysine methylation and neurodevelopmental disorders. Int J Mol Sci 18:1–20. https://doi.org/10.3390/ijms18071404
Wang ZZ, Yang WX, Zhang Y et al (2015) Phosphodiesterase-4D knock-down in the prefrontal cortex alleviates chronic unpredictable stress-induced depressive-like behaviors and memory deficits in mice. Sci Rep 5:1–15. https://doi.org/10.1038/srep11332
Xu Y, Pan J, Sun J et al (2015) Inhibition of phosphodiesterase 2 reverses impaired cognition and neuronal remodeling caused by chronic stress. Neurobiol Aging 36:955–970. https://doi.org/10.1016/j.neurobiolaging.2014.08.028
Kim YK, Na KS, Myint AM, Leonard BE (2016) The role of pro-inflammatory cytokines in neuroinflammation, neurogenesis and the neuroendocrine system in major depression. Prog Neuro-Psychopharmacol Biol Psychiatry 64:277–284. https://doi.org/10.1016/j.pnpbp.2015.06.008
McEwen BS (2005) Glucocorticoids, depression, and mood disorders: Structural remodeling in the brain. Metabolism 54:20–23. https://doi.org/10.1016/j.metabol.2005.01.008
Herbert J, Goodyer IM, Grossman AB et al (2006) Do corticosteroids damage the brain? J Neuroendocrinol 18:393–411. https://doi.org/10.1111/j.1365-2826.2006.01429.x
Kraushaar DC, Jin W, Maunakea A et al (2013) Genome-wide incorporation dynamics reveal distinct categories of turnover for the histone variant H3.3. Genome Biol 14:1. https://doi.org/10.1186/gb-2013-14-10-r121
Dudek KA, Dion-Albert L, Kaufmann FN et al (2021) Neurobiology of resilience in depression: immune and vascular insights from human and animal studies. Eur J Neurosci 53:183–221. https://doi.org/10.1111/ejn.14547
Lu Y, Yang J, Sun J et al (2020) mRNA and miRNA profiles in the nucleus accumbens are associated with psychological stress-induced susceptible and resilient mice. Pharmacol Biochem Behav 199:173062. https://doi.org/10.1016/j.pbb.2020.173062
Vogel Ciernia A, Laufer BI, Dunaway KW et al (2018) Experience-dependent neuroplasticity of the developing hypothalamus: integrative epigenomic approaches. Epigenetics 13:318–330. https://doi.org/10.1080/15592294.2018.1451720
Basile F, Capaccia C, Zampini D et al (2021) Omics insights into animal resilience and stress factors. Animals 11:1–24. https://doi.org/10.3390/ani11010047
Park J, Lee J, Choi K, Kang HJ (2021) Regulation of behavioral response to stress by microRNA-690. Mol Brain 14:3–6. https://doi.org/10.1186/s13041-021-00728-3
Xu Q, Jiang M, Gu S et al (2020) Early life stress induced DNA methylation of monoamine oxidases leads to depressive-like behavior. Front Cell Dev Biol 8:1–9. https://doi.org/10.3389/fcell.2020.582247
Cattaneo A, Suderman M, Cattane N et al (2020) Long-term effects of stress early in life on microRNA-30a and its network: Preventive effects of lurasidone and potential implications for depression vulnerability. Neurobiol Stress 13:100271. https://doi.org/10.1016/j.ynstr.2020.100271
Maitra S, Khandelwal N, Kootar S et al (2020) Histone Lysine Demethylase JMJD2D/KDM4D and Family Members Mediate Effects of Chronic Social Defeat Stress on Mouse Hippocampal Neurogenesis and Mood Disorders. Brain Sci 10(11):833. https://doi.org/10.3390/brainsci10110833
Sun H, Zhang X, Kong Y et al (2021) Maternal separation-induced histone acetylation correlates with BDNF-programmed synaptic changes in an animal model of PTSD with sex differences. Mol Neurobiol 58:1738–1754. https://doi.org/10.1007/s12035-020-02224-6
Machado-Vieira R, Salvadore G, Luckenbaugh DA et al (2008) Rapid onset of antidepressant action: a new paradigm in the research and treatment of major depressive disorder. J Clin Psychiatry 69:946–958. https://doi.org/10.4088/JCP.v69n0610
Ramaker MJ, Dulawa SC (2017) Identifying fast-onset antidepressants using rodent models. Mol Psychiatry 22:656–665. https://doi.org/10.1038/mp.2017.36
Berger SL (2007) The complex language of chromatin regulation during transcription. Nature 447:407–412. https://doi.org/10.1038/nature05915
Bale TL, Baram TZ, Brown AS et al (2010) Early life programming and neurodevelopmental disorders. Biol Psychiatry 68:314–319. https://doi.org/10.1016/j.biopsych.2010.05.028
Millan MJ (2013) An epigenetic framework for neurodevelopmental disorders: From pathogenesis to potential therapy. Neuropharmacology 68:2–82. https://doi.org/10.1016/j.neuropharm.2012.11.015
Khavari B, Cairns MJ (2020) Epigenomic dysregulation in schizophrenia: in search of disease etiology and biomarkers. Cells 9:1–27. https://doi.org/10.3390/cells9081837
Kuehner JN, Bruggeman EC, Wen Z, Yao B (2019) Epigenetic regulations in neuropsychiatric disorders. Front Genet 10:1–30. https://doi.org/10.3389/fgene.2019.00268
Penner-Goeke S, Binder EB (2019) Epigenetics and depression. Dialogues Clin Neurosci 21:397–405. https://doi.org/10.31887/DCNS.2019.21.4/ebinder
Blokhin IO, Khorkova O, Saveanu RV, Wahlestedt C (2020) Molecular mechanisms of psychiatric diseases. Neurobiol Dis 146:105136. https://doi.org/10.1016/j.nbd.2020.105136
Varga Z, Csabai D, Miseta A et al (2017) Chronic stress affects the number of GABAergic neurons in the orbitofrontal cortex of rats. Behav Brain Res 316:104–114. https://doi.org/10.1016/j.bbr.2016.08.030
Palmfeldt J, Henningsen K, Aistrup S et al (2016) Molecular and cellular neuroscience protein biomarkers of susceptibility and resilience to stress in a rat model of depression. Mol Cell Neurosci 74:87–95. https://doi.org/10.1016/j.mcn.2016.04.001
Czéh B, Fuchs E, Wiborg O, Simon M (2016) Animal models of major depression and their clinical implications. Prog Neuro-Psychopharmacology Biol Psychiatry 64:293–310. https://doi.org/10.1016/j.pnpbp.2015.04.004
Willner P (2005) Chronic Mild Stress (CMS) revisited : consistency and behavioural-neurobiological concordance in the effects of CMS. Neuropsychobiology 52:90–110. https://doi.org/10.1159/000087097
Willner P (1997) Validity, reliability and utility of the chronic mild stress model of depression: a 10-year review and evaluation. Psychopharmacology 134:319–329
Bergström A, Jayatissa MN, Mørk A, Wiborg O (2008) Stress sensitivity and resilience in the chronic mild stress rat model of depression; an in situ hybridization study. Brain Res 1196:41–52. https://doi.org/10.1016/j.brainres.2007.12.025
Almeida RF, Nonose Y, Ganzella M et al (2021) Antidepressant-like effects of chronic guanosine in the olfactory bulbectomy mouse model. Front Psychiatry 12:1–14. https://doi.org/10.3389/fpsyt.2021.701408
Popoli M, Yan Z, Mcewen BS, Sanacora G (2011) The stressed synapse : the impact of stress and glucocorticoids on glutamate transmission. 13:. https://doi.org/10.1038/nrn3138
Horta RN, Kahl VFS, Sarmento MDS et al (2016) Protective effects of acerola juice on genotoxicity induced by iron in vivo. Genet Mol Biol 39:122–128. https://doi.org/10.1590/1678-4685-GMB-2015-0157
Collins AR (2004) The comet assay for DNA damage and repair: Principles, applications, and limitations. Appl Biochem Biotechnol - Part B Mol Biotechnol 26:249–261. https://doi.org/10.1385/MB:26:3:249
Da Silva ALG, da Rosa HT, Karnopp TE et al (2013) Evaluation of DNA damage in COPD patients and its correlation with polymorphisms in repair genes. BMC Med Genet 14:. https://doi.org/10.1186/1471-2350-14-93
Shechter D, Dormann HL, Allis CD, Hake SB (2007) Extraction, purification and analysis of histones. Nat Protoc 2:1445–1457. https://doi.org/10.1038/nprot.2007.202
Beldjoud H, Messanvi F, NadifKasri N, Roozendaal B (2016) Extraction, identification, and quantification of histones from small quantities of specific brain tissue. Curr Protoc Neurosci 2016:4.38.1-4.38.20. https://doi.org/10.1002/cpns.11
De Oliveira J, Moreira ELG, Dos Santos DB et al (2014) Increased susceptibility to amyloid-β-induced neurotoxicity in mice lacking the low-density lipoprotein receptor. J Alzheimer’s Dis 41:43–60. https://doi.org/10.3233/JAD-132228
Sales AJ, Guimarães FS, Joca SRL (2021) DNA methylation in stress and depression: from biomarker to therapeutics. Acta Neuropsychiatr. https://doi.org/10.1017/neu.2021.18
Antoniuk S, Bijata M, Ponimaskin E, Wlodarczyk J (2019) Chronic unpredictable mild stress for modeling depression in rodents: Meta-analysis of model reliability. Neurosci Biobehav Rev 99:101–116. https://doi.org/10.1016/j.neubiorev.2018.12.002
Hao Y, Ge H, Sun M, Gao Y (2019) Selecting an appropriate animal model of depression. Int J Mol Sci 20:1–16. https://doi.org/10.3390/ijms20194827
Leite-Almeida H, Pinto-Ribeiro F, Almeida A (2015) Animal models for the study of comorbid pain and psychiatric disorders. Mod Trends Pharmacopsychiatry 30:1–21. https://doi.org/10.1159/000435929
Liu YY, Zhou XY, Yang LN et al (2017) Social defeat stress causes depression-like behavior with metabolite changes in the prefrontal cortex of rats. PLoS One. https://doi.org/10.1371/journal.pone.0176725
Couch Y, Trofimov A, Markova N et al (2016) Low-dose lipopolysaccharide (LPS) inhibits aggressive and augments depressive behaviours in a chronic mild stress model in mice. J Neuroinflammation 13:1–17. https://doi.org/10.1186/s12974-016-0572-0
Jianguo L, Xueyang J, Cui W et al (2019) Altered gut metabolome contributes to depression-like behaviors in rats exposed to chronic unpredictable mild stress. Transl Psychiatry 9:. https://doi.org/10.1038/s41398-019-0391-z
Song AQ, Gao B, Fan JJ et al (2020) NLRP1 inflammasome contributes to chronic stress-induced depressive-like behaviors in mice. J Neuroinflammation 17:1–13. https://doi.org/10.1186/s12974-020-01848-8
Xu S, Liu Y, Pu J et al (2020) Chronic stress in a rat model of depression disturbs the glutamine–glutamate–GABA cycle in the striatum, hippocampus, and cerebellum. Neuropsychiatr Dis Treat 16:557–570. https://doi.org/10.2147/NDT.S245282
Furman D, Campisi J, Verdin E et al (2019) Chronic inflammation in the etiology of disease across the life span. Nat Med 25:1822–1832. https://doi.org/10.1038/s41591-019-0675-0
Bogdanova Olena V, Kanekar Shami, E D’Anci Kristen, F Renshaw Perry (2017) Factors influencing behavior in the forced swim test. Physiol Behav 176:139–148. https://doi.org/10.1016/j.physbeh.2013.05.012
Beurel E, Toups M, Nemeroff CB (2016) The bidirectional relationship of depression and inflammation: double trouble. Physiol Behav 176:139–148. https://doi.org/10.1016/j.neuron.2020.06.002.The
Raza MU, Tufan T, Wang Y et al (2016) DNA damage in major psychiatric diseases. Neurotox Res 30:251–267. https://doi.org/10.1007/s12640-016-9621-9
Czarny P, Bialek K, Ziolkowska S et al (2020) DNA damage and repair in neuropsychiatric disorders. What do we know and what are the future perspectives? Mutagenesis 35:79–106. https://doi.org/10.1093/mutage/gez035
Jorgensen A, Breitenstein K, Kalliokoski O et al (2019) Markers of HPA-axis activity and nucleic acid damage from oxidation after electroconvulsive stimulations in rats Correspondence : Anders Jorgensen , M . D ., Ph . D , Psychiatric Centre Copenhagen ( Rigshospitalet ) Edel Sauntes Allé 10 , DK-2100 Copenhag. 1–29
Czarny P, Kwiatkowski D, Kacperska D et al (2015) Elevated level of DNA damage and impaired repair of oxidative DNA damage in patients with recurrent depressive disorder. Med Sci Monit 21:412–418. https://doi.org/10.12659/MSM.892317
Ahmadimanesh M, Abbaszadegan MR, Morshedi Rad D et al (2019) Effects of selective serotonin reuptake inhibitors on DNA damage in patients with depression. J Psychopharmacol 33:1364–1376. https://doi.org/10.1177/0269881119874461
Skucha A, Ebner J, Grebien F (2019) Roles of setd2 in leukemia—transcription, dna-damage, and beyond. Int J Mol Sci 20:. https://doi.org/10.3390/ijms20051029
Pathak SS, Maitra S, Chakravarty S, Kumar A (2016) Histone lysine demethylases of JMJD2 or KDM4 family are important epigenetic regulators in reward circuitry in the etiopathology of depression. Neuropsychopharmacology 42:854–863. https://doi.org/10.1038/npp.2016.231
Bannister AJ, Kouzarides T (2011) Regulation of chromatin by histone modifications. Cell Res 21:381–395. https://doi.org/10.1038/cr.2011.22
Jakovcevski M, Ruan H, Shen EY et al (2015) Neuronal Kmt2a/Mll1 histone methyltransferase is essential for prefrontal synaptic plasticity and working memory. J Neurosci 35:5097–5108. https://doi.org/10.1523/JNEUROSCI.3004-14.2015
Kerimoglu C, Agis-Balboa RC, Kranz A et al (2013) Histone-methyltransferase mll2 (kmt2B) is required for memory formation in mice. J Neurosci 33:3452–3464. https://doi.org/10.1523/JNEUROSCI.3356-12.2013
Guenther MG, Levine SS, Boyer LA et al (2007) A chromatin landmark and transcription initiation at most promoters in human cells. Cell 130:77–88. https://doi.org/10.1016/j.cell.2007.05.042
Roidl D, Hacker C (2014) Histone methylation during neural development. Cell Tissue Res 356:539–552. https://doi.org/10.1007/s00441-014-1842-8
Fontebasso AM, Schwartzentruber J, Khuong-Quang DA et al (2013) Mutations in SETD2 and genes affecting histone H3K36 methylation target hemispheric high-grade gliomas. Acta Neuropathol 125:659–669. https://doi.org/10.1007/s00401-013-1095-8
Cascante A, Klum S, Biswas M et al (2014) Gene-specific methylation control of H3K9 and H3K36 on neurotrophic BDNF versus astroglial GFAP genes by KDM4A/C regulates neural stem cell differentiation. J Mol Biol 426:3467–3477. https://doi.org/10.1016/j.jmb.2014.04.008
Di Benedetto B, Malik VA, Begum S et al (2016) Fluoxetine requires the endfeet protein aquaporin-4 to enhance plasticity of astrocyte processes. Front Cell Neurosci 10:1–12. https://doi.org/10.3389/fncel.2016.00008
Negrón-Oyarzo I, Lara-Vásquez A, Palacios-García I et al (2016) Schizophrenia and reelin: a model based on prenatal stress to study epigenetics, brain development and behavior. Biol Res 49:1–10. https://doi.org/10.1186/s40659-016-0076-5
Teixeira CM, Martín ED, Sahún I et al (2011) Overexpression of reelin prevents the manifestation of behavioral phenotypes related to schizophrenia and bipolar disorder. Neuropsychopharmacology 36:2395–2405. https://doi.org/10.1038/npp.2011.153
Golden SA, Christoffel DJ, Hodes GE et al (2013) Epigenetic regulation of synaptic remodeling in stress disorders. Nat Med 19:337–344. https://doi.org/10.1038/nm.3090.Epigenetic
McEwen BS, Bowles NP, Gray JD et al (2015) Mechanisms of stress in the brain. Nat Neurosci 18:1353–1363. https://doi.org/10.1038/nn.4086.Mechanisms
Rao P, Yallapu MM, Sari Y et al (2015) Designing novel nanoformulations targeting glutamate transporter excitatory amino acid transporter 2: Implications in treating drug addiction. J Pers Nanomed 1(1):3–9
McEwen BS, Nasca C, Gray JD (2016) Stress effects on neuronal structure: hippocampus, amygdala, and prefrontal cortex. Neuropsychopharmacology 41:3–23. https://doi.org/10.1038/npp.2015.171
Yang F, Wang H, Chen H et al (2020) RAGE Signaling pathway in hippocampus dentate gyrus involved in GLT-1 decrease induced by chronic unpredictable stress in rats. Brain Res Bull 163:49–56. https://doi.org/10.1016/j.brainresbull.2020.06.020
Skupio U, Tertil M, Sikora M et al (2015) Behavioral and molecular alterations in mice resulting from chronic treatment with dexamethasone: relevance to depression. Neuroscience 286:141–150. https://doi.org/10.1016/j.neuroscience.2014.11.035
Ivens S, Çalışkan G, Papageorgiou I et al (2019) Persistent increase in ventral hippocampal long-term potentiation by juvenile stress: a role for astrocytic glutamine synthetase. Glia 67:2279–2293. https://doi.org/10.1002/glia.23683
Shirayama Y, Takahashi M, Osone F et al (2017) Myo-inositol, glutamate, and glutamine in the prefrontal cortex, hippocampus, and amygdala in major depression. Biol Psychiatry Cogn Neurosci Neuroimaging 2:196–204. https://doi.org/10.1016/j.bpsc.2016.11.006
Rajkowska G, Stockmeier C (2013) Astrocyte pathology in major depressive disorder: insights from human postmortem brain tissue. Curr Drug Targets 14:1225–1236. https://doi.org/10.2174/13894501113149990156
Kantrowitz JT, Dong Z, Milak MS et al (2021) Ventromedial prefrontal cortex/anterior cingulate cortex Glx, glutamate, and GABA levels in medication-free major depressive disorder. Transl Psychiatry 11:1–6. https://doi.org/10.1038/s41398-021-01541-1
Cotter DR, Pariante CM, Everall IP (2001) Glial cell abnormalities in major psychiatric disorders: the evidence and implications. Brain Res Bull 55:585–595. https://doi.org/10.1016/S0361-9230(01)00527-5
Czéh B, Simon M, Schmelting B et al (2006) Astroglial plasticity in the hippocampus is affected by chronic psychosocial stress and concomitant fluoxetine treatment. Neuropsychopharmacology 31:1616–1626. https://doi.org/10.1038/sj.npp.1300982
Li K, Yan L, Zhang Y et al (2020) Seahorse treatment improves depression-like behavior in mice exposed to CUMS through reducing inflammation/oxidants and restoring neurotransmitter and neurotrophin function. J Ethnopharmacol 250:. https://doi.org/10.1016/j.jep.2019.112487
Perez-Urrutia N, Mendoza C, Alvarez-Ricartes N et al (2017) Intranasal cotinine improves memory, and reduces depressive-like behavior, and GFAP + cells loss induced by restraint stress in mice. Exp Neurol 295:211–221. https://doi.org/10.1016/j.expneurol.2017.06.016
Araya-Callís C, Hiemke C, Abumaria N, Flugge G (2012) Chronic psychosocial stress and citalopram modulate the expression of the glial proteins GFAP and NDRG2 in the hippocampus. Psychopharmacology 224:209–222. https://doi.org/10.1007/s00213-012-2741-x
Collins BE, Greer CB, Coleman BC, Sweatt JD (2019) Histone H3 lysine K4 methylation and its role in learning and memory 06 Biological Sciences 0604 Genetics 11 Medical and Health Sciences 1109 Neurosciences. Epigenetics Chromatin 12:1–16. https://doi.org/10.1186/s13072-018-0251-8
Dang R, Guo YY, Zhang K et al (2019) Predictable chronic mild stress promotes recovery from LPS-induced depression. Mol Brain 12:1–12. https://doi.org/10.1186/s13041-019-0463-2
Wang H, Huang F, Hu Z, Zhang W (2017) Early-life social isolation-induced depressive-like behavior in rats results in microglial activation and neuronal histone methylation that are mitigated by minocycline. Neurotox Res. https://doi.org/10.1007/s12640-016-9696-3
Acknowledgements
Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq), Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES), Fundação de Amparo à Pesquisa do Estado do Rio Grande do Sul (FAPERGS), Programa Pesquisa para o SUS (PPSUS)/FAPERGS and Pró-reitora de Pesquisa (PROPESQ‐UFRGS) that supported this study. Programa de Pós-Graduação em Biologia Celular e Molecular (PPGBCM).All authors have read and agree with authority.
Funding
This study was supported by: FAPERGS PPSUS (21/2551-0000090-2), FAPERGS PRONEX (16/2551-0000499-4), CNPq, CAPES and PROPESQ/UFRGS.
Author information
Authors and Affiliations
Contributions
Lucas Santos: the central idea of research, animal handling and experimentation, behavioural tests, immunofluorescence, biochemical and molecular analyses, writing and reviewing the manuscript.
Luiza Behrens: animal handling and experimentation, behavioural tests, biochemical and molecular analyses.
Camila Barbosa: animal handling and experimentation and behavioural tests.
Camila Tiefensee-Ribeiro: animal handling and experimentation and euthanasia.
Helen Rosa-Silva: animal management and experimentation and DNA damage and repair techniques.
Nauana Somensi: animal handling and experimentation and biochemical analyses.
Pedro Ozorio Brum: euthanasia and immunofluorescence.
Alexandre Kleber Silveira: animal handling and experimentation and euthanasia.
Matheus Scarapatto Rodrigues: euthanasia and immunofluorescence.
Jade de Oliveira: manuscript review and immunofluorescence.
Daniel Gelain: a manuscript review.
Roberto Almeida: the central idea of the manuscript's research, writing and review.
José Cláudio Fonseca Moreira: the central idea of the manuscript's research, writing and review.
Corresponding author
Ethics declarations
Ethics Approval
All applicable international, national, and institutional guidelines for the care and use of the animals were followed. The Animal Experimentation Ethics Committee (CONCEA) of the UFRGS approved the research protocol under authorization number 35277.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Santos, L., Behrens, L., Barbosa, C. et al. Histone 3 Trimethylation Patterns are Associated with Resilience or Stress Susceptibility in a Rat Model of Major Depression Disorder. Mol Neurobiol (2024). https://doi.org/10.1007/s12035-024-03912-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12035-024-03912-3