Abstract
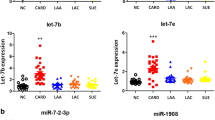
Inflammation and immunity play important roles in the pathogenesis of ischemic stroke. This study aimed to explore key regulatory genes in acute ischemic stroke (AIS) and their underlying mechanisms to provide new research targets for the diagnosis and treatment of ischemic stroke. We searched for differentially expressed mRNAs and miRNAs in patients with AIS and healthy populations in GEO databases, constructed a miRNA–mRNA network, and screened key miRNAs using least absolute shrinkage and selection operator regression and the support vector machine-recursive feature elimination model. Correlations between key miRNAs and infiltrating immune cells and inflammatory factors were analyzed using CIBERSORT and immunoassays and verified using clinical experiments. Bioinformatics analysis identified hsa-miR-877-5p as a key regulatory miRNA in AIS that can modulate immune and inflammatory responses. In clinical studies, it was verified by quantitative PCR analysis that the expression of hsa-miR-877-5p in the blood of AIS patients was higher than that of the healthy group. Then, enzyme-linked immunosorbent assay revealed that the expression of IL-23 and TNF-α related to inflammation in AIS patients was higher than that of the healthy. Quantitative PCR further found that the relative mRNA expression of IL-23, CXCR3, and TNF-α in AIS group was higher than that of the healthy group. This study may provide a basis for a more comprehensive understanding of the potential mechanism of the occurrence and development of AIS, and hsa-miR-877-5p and its downstream effectors IL-23, CXCR3, and TNF-α may be potential intervention targets in AIS.











Similar content being viewed by others
Data Availability
The datasets generated during and/or analyzed during the current study are not publicly available due to protect patient privacy but are available from the corresponding author on reasonable request.
References
Sommer CJ (2017) Ischemic stroke: experimental models and reality. Acta Neuropathol 133(2):245–261. https://doi.org/10.1007/s00401-017-1667-0
Puig B, Brenna S, Magnus T (2018) Molecular communication of a dying neuron in stroke. Int J Mol Sci 19(9):2834. https://doi.org/10.3390/ijms19092834
Meng H, Zhao H, Cao X, Hao J, Zhang H, Liu Y et al (2019) Double-negative T cells remarkably promote neuroinflammation after ischemic stroke. Proc Natl Acad Sci 116(12):5558–5563. https://doi.org/10.1073/pnas.1814394116
Shichita T, Sugiyama Y, Ooboshi H, Sugimori H, Nakagawa R, Takada I et al (2009) Pivotal role of cerebral interleukin-17-producing gammadeltaT cells in the delayed phase of ischemic brain injury. Nat Med 15(8):946–950. https://doi.org/10.1038/nm.1999
Gelderblom M, Gallizioli M, Ludewig P, Thom V, Arunachalam P, Rissiek B et al (2018) IL-23 (interleukin-23)-producing conventional dendritic cells control the detrimental IL-17 (interleukin-17) response in stroke. Stroke 49(1):155–164. https://doi.org/10.1161/STROKEAHA.117.019101
Zaremba J, Losy J (2001) Early TNF-alpha levels correlate with ischaemic stroke severity. Acta Neurol Scand 104(5):288–295. https://doi.org/10.1034/j.1600-0404.2001.00053.x
Iadecola C, Anrather J (2011) The immunology of stroke: from mechanisms to translation. Nat Med 17(7):796–808. https://doi.org/10.1038/nm.2399
de Oliveira LRA, de Oliveira Albuquerque A, Silva CISM, Silva JM, de Figueiredo Cavalcante Casadevall MQ, de Azevedo OGR, de Souza Pires Albuquerque VL et al (2020) Preconditioning with L-Ala-Gln reduces the expression of inflammatory markers (TNF-α, NF-κB, IL-6 and HO-1) in an injury animal model of cerebrovascular ischemia in Meriones unguiculatus (gerbils). Acta Cir Bras 35(6):e202000601. https://doi.org/10.1590/s0102-865020200060000001
Iadecola C, Anrather J (2011) The immunology of stroke: from mechanisms to translation. Nat Med 17(7):796–808. https://doi.org/10.1038/nm.2399
Cai W, Shi L, Zhao J, Xu F, Dufort C, Ye Q, Yang T, Dai X et al (2022) Neuroprotection against ischemic stroke requires a specific class of early responder T cells in mice. J Clin Invest 132(15):e157678. https://doi.org/10.1172/JCI157678
Tian F, Yuan C, Hu L, Shan S (2017) MicroRNA-93 inhibits inflammatory responses and cell apoptosis after cerebral ischemia reperfusion by targeting interleukin-1 receptor-associated kinase 4. Exp Ther Med 14(4):2903–2910. https://doi.org/10.3892/etm.2017.4874
Lee SJ, Kim JM, Lee ES, Park KY, Kim HR (2023) Relationship between MicroRNA signature and arterial stiffness in patients with ischemic stroke. J Clin Neurol 19(1):28–35. https://doi.org/10.3988/jcn.2023.19.1.28
Iadecola C, Buckwalter MS, Anrather J (2020) Immune responses to stroke: mechanisms, modulation, and therapeutic potential. J Clin Invest 130(6):2777–2788. https://doi.org/10.1172/JCI135530
Chinese Medical Association Branch of Neurology, Group of Cerebrovascular diseases, Branch of Neurology, Chinese Medical Association (2019) Diagnostic points of various major cerebrovascular diseases in China 2019. Chin J Neurol 52(9):710–715. https://doi.org/10.3760/cma.j.issn.1006-7876.2019.09.003
Xiaoyang Y, Petersen F, Yaqing S, Brigitte K, Junie D, Ahmadi Junping Y, Jacqueline W, Xiaoqing W et al (2021) Transfer of PBMC from SSc patients induces autoantibodies and systemic inflammation in Rag2-/-/IL2rg-/- mice. Front Immunol 12:677970. https://doi.org/10.3389/fimmu.2021.677970
Kölliker-Frers R, Udovin L, Otero-Losada M, Kobiec T, Herrera MI, Palacios J, Razzitte G, Capani F (2021) Neuroinflammation: an integrating overview of reactive-neuroimmune cell interactions in health and disease. Mediators Inflamm 2021:9999146. https://doi.org/10.1155/2021/9999146
Sun C, Zhu X, Tao T, Zhang D, Wang Y, Xu H, Ren Y, Wang Y (2017) The β4GalT1 affects the fibroblast-like synoviocytes invasion in rheumatoid arthritis by modifying N-linked glycosylation of CXCR3.Eur. J Cell Biol 96(2):172–181. https://doi.org/10.1016/j.ejcb.2017.02.001
Boyé K, Pujol N, Alves I, Chen YP, Daubon T, Lee YZ, Dedieu S, Constantin M et al (2017) The role of CXCR3/LRP1 cross-talk in the invasion of primary brain tumors. Nat Commun 8(1):1571. https://doi.org/10.1038/s41467-017-01686-y
Li HL, Huang Y, Zhou YL, Teng RH, Zhou SZ, Lin JP, Yang Y, Zhu SM et al (2020) Motif chemokine 10 contributes to the development of neuropathic pain by increasing the permeability of the blood-spinal cord barrie. Front Immunol 11:477. https://doi.org/10.3389/fimmu.2020.00477
Krauthausen M, Kummer MP, Zimmermann J, Reyes-Irisarri E, Terwel D, Bulic B, Heneka MT, Müller M (2015) CXCR3 promotes plaque formation and behavioral deficits in an Alzheimer's disease model. J Clin Invest 125(1):365–378. https://doi.org/10.1172/JCI66771
Goldberg EL, Letian A, Dlugos T, Leveau C, Dixit VD (2023) Innate immune cell-intrinsic ketogenesis is dispensable for organismal metabolism and age-related inflammation. J Biol Chem 299(3):103005. https://doi.org/10.1016/j.jbc.2023.103005
Goldberg SH, van der Meer P, Hesselgesser J, Jaffer S, Kolson DL, Albright AV, González-Scarano F, Lavi E (2001) CXCR3 expression in human central nervous systemdiseases. Neuropathol Appl Neurobiol 27(2):127–138. https://doi.org/10.1046/j.1365-2990.2001.00312.x
Zeng J, Bao T, Yang K, Zhu X, Wang S, Xiang W, Ge A, Zeng L et al (2023) The mechanism of microglia-mediated immune inflammation in ischemic stroke and the role of natural botanical components in regulating microglia: a review. Front Immunol 13:1047550. https://doi.org/10.3389/fimmu.2022.1047550
Liang M, Zhang L, Geng Z (2021) Advances in the development of biomarkers for poststroke epilepsy. Biomed Res Int 2021:5567046. https://doi.org/10.1155/2021/5567046
Chen AQ, Fang Z, Chen XL, Yang S, Zhou YF, Mao L, Xia YP, Jin HJ et al (2019) Microglia-derived TNF-α mediatesendothelial necroptosis aggravating blood brain-barrier disruption after ischemic stroke. Cell Death Dis 10(7):487. https://doi.org/10.1038/s41419-019-1716-9
Stone MJ, Hayward JA, Huang C, Huma Z, Sanchez J (2017) Mechanisms of regulation of the chemokine-receptor network. Int J Mol Sci 18(2):342. https://doi.org/10.3390/ijms18020342
Spronk E, Sykes G, Falcione S, Munsterman D, Joy T, Kamtchum-TatueneJ JGC (2021) Hemorrhagic transformation in ischemic stroke and the role of inflammation. Front Neurol 12:661955. https://doi.org/10.3389/fneur.2021.661955
Pluta R, Januszewski S, Czuczwar SJ (2021) Neuroinflammation in post-ischemic neurodegeneration of the brain: friend, foe, or both? Int J Mol Sci 22(9):4405. https://doi.org/10.3390/ijms22094405
Huţanu A, Iancu M, Bălaşa R, Maier S, Dobreanu M (2018) Predicting functional outcome of ischemic stroke patients in Romania based on plasma CRP, sTNFR-1, D-Dimers, NGAL and NSE measured using a biochip array. Acta Pharmacol Sin 39(7):1228–1236. https://doi.org/10.1038/aps.2018.26
Sotgiu S, Zanda B, Marchetti B, Fois ML, Arru G, Pes GM, Salaris FS, Arru A et al (2006) Inflammatory biomarkers in blood of patientswith acute brain ischemia. Eur J Neurol 13(5):505–513. https://doi.org/10.1111/j.1468-1331.2006.01280
Peng T, Jiang Y, Farhan M, Lazarovici P, Chen L, Zheng W (2019) Anti-inflammatory effects of traditional Chinese medicines on preclinical in vivo models of brain ischemia-reperfusion-injury: prospects for neuroprotective drug discovery and therapy. Front Pharmacol 10:204. https://doi.org/10.3389/fphar.2019.00204
Lay CS, Bridges A, Goulding J, Briddon SJ, Soloviev Z, Craggs PD, Hill SJ (2022) Probing the binding of interleukin-23 to individual receptor components and the IL-23 heteromeric receptor complex in living cells using NanoBRET. Cell Chem Biol 29(1):19–29. https://doi.org/10.1016/j.chembiol.2021.05.002
Larosa M, Zen M, Gatto M, Jesus D, Zanatta E, Iaccarino L, Inês L, Doria A (2019) IL-12 and IL-23/Th17 axis in systemic lupus erythematosus. Exp Biol Med (Maywood) 244(1):42–51. https://doi.org/10.1177/1535370218824547
Nitsch L, Zimmermann J, Krauthausen M, Hofer MJ, Saggu R, Petzold GC, Heneka MT, Getts DR et al (2019) CNS-specific synthesis of interleukin 23 induces a progressive cerebellar ataxia and the accumulation of both T and B cells in the brain: characterization of a novel transgenic mouse model. Mol Neurobiol 56(12):7977–7993. https://doi.org/10.1007/s12035-019-1640-0
Tahmasebinia F, Pourgholaminejad A (2017) The role of Th17 cells in auto-inflammatory neurological disorders. Prog Neuropsychopharmacol Biol Psychiatry 79(Pt B):408–416. https://doi.org/10.1016/j.pnpbp.2017.07.023
Milovanovic J, Arsenijevic A, Stojanovic B, Kanjevac T, Arsenijevic D, Radosavljevic G, Milovanovic M, Arsenijevic N (2020) Interleukin-17 in chronic inflammatory. Front Immunol 11:947. https://doi.org/10.3389/fimmu.2020.00947
Jiraskova Zakostelska Z, Reiss Z, Tlaskalova-Hogenova H, Rob F (2023) Paradoxical reactions to anti-TNFα and anti-IL-17 treatment in psoriasis patients: are skin and/or gut microbiota involved? Dermatol Ther (Heidelb) 13(4):911–933. https://doi.org/10.1007/s13555-023-00904-4
Acknowledgements
We would like to thank Experimental Center, Shandong University of Traditional Chinese Medicine, Jinan, 250355, PR China.
Funding
This study was funded by the Natural Science Foundation of China (82205064) and the Natural Science Foundation of Shandong Province (ZR2021QH110).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection, and analysis were performed by Si-Shuo Zhang, Ji-Wei Zhang, Kai-Xin Zhang, Wen-Qiang Cui, Hong-Wei Zhi, Hai-Tao Li, and Hong-Yun Wu. The first draft of the manuscript was written by Ya-Han Wang and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no competing interests.
Ethics Approval
Human experimentations in this study was approved by the Ethics Committee of our hospital, and all enrolled subjects signed informed consent (Date: 2023/06/20/No. 2023-066-KY).
This study was performed in line with the principles of the Declaration of Helsinki. Approval was granted by the Ethics Committee of the First Clinical Medical School of Shandong University of Chinese Medicine (Date: 2022/07/12/No. SDUTCM20220712001).
Consent to Participate
Written informed consent was obtained from the clinical participants.
Consent to Publish
The authors affirm that human research participants provided informed consent for publication of the images in Fig. 9.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Si-Shuo Zhang, Ji-Wei Zhang and Kai-Xin Zhang are the co-first authors.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhang, SS., Zhang, JW., Zhang, KX. et al. Hsa-miR-877-5p Expression in Acute Ischemic Stroke Based on Bioinformatics Analysis and Clinical Validation. Mol Neurobiol 61, 1990–2005 (2024). https://doi.org/10.1007/s12035-023-03675-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-023-03675-3