Abstract
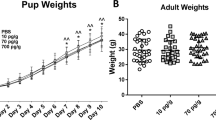
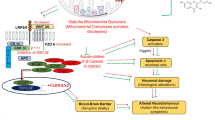
MgSO4 has been used for the past two decades as neuroprotective treatment in a variety of preterm conditions. Despite the putative advantages of MgSO4 as a neuroprotective agent in the preterm brain, the short- and long-term molecular function of MgSO4 as a neuroprotective agent has not been fully elucidated. Neuregulin (NRG1)-ErbB4 signaling plays a critical role in embryonic brain development, in the biology of dopaminergic, GABAergic, and glutamatergic systems. We hypothesize that this pathway may be associated with the neuroprotective role of MgSO4. The current study aims to investigate the ability of MgSO4 to modulate the normal developing expression pattern of selected genes related to the NRG1-ErbB, dopaminergic, GABAergic, and glutamatergic systems. We demonstrate that overall short-term treatment of dam rats with MgSO4 affects the expression of fetal brain NRG1, NRG3, ErbB4, GAD67, tyrosine hydroxylase (TH), dopamine D2 and D1 receptors, GluN1, and GluN2B. More specifically, the administration of MgSO4 alters the expression of NRG-ErbB, GAD67, TH, and D2R at early gestation day 16 (GD16) regardless of the activation of the maternal immune system by lipopolysaccharide (LPS). Our data suggest that MgSO4 treatment may affect the expression of major neuronal systems and pathways mostly at an early gestation day. These changes might be an initial clue (foundation stone) in the molecular mechanism that underlies the beneficial effect of MgSO4 as a neuroprotective agent for the developmental brain.




Similar content being viewed by others
References
Bilbo SD, Schwarz JM (2012) The immune system and developmental programming of brain and behavior. Front Neuroendocrinol 33:267–286. https://doi.org/10.1016/j.yfrne.2012.08.006
Bilbo SD, Block CL, Bolton JL, Hanamsagar R, Tran PK (2018) Beyond infection - maternal immune activation by environmental factors, microglial development, and relevance for autism spectrum disorders. Exp Neurol 299:241–251. https://doi.org/10.1016/j.expneurol.2017.07.002
Brown AS, Conway F (2019) Maternal immune activation and related factors in the risk of offspring psychiatric disorders. Frontiers in Psychiatry 10:1–6. https://doi.org/10.3389/fpsyt.2019.00430
Burd I, Breen K, Friedman A, Chai J, Elovitz MA (2010) Magnesium sulfate reduces inflammation-associated brain injury in fetal mice. Am J Obstet Gynecol 202:292.e1–292.e9. https://doi.org/10.1016/j.ajog.2010.01.022
Chollat C, Sentilhes L, Marret S (2018) Fetal neuroprotection by magnesium sulfate: from translational research to clinical application. Front Neurol 9:1–7. https://doi.org/10.3389/fneur.2018.00247
Dabbah-Assadi F, Alon D, Golani I, Doron R, Kremer I, Beloosesky R, Shamir A (2019) The influence of immune activation at early vs late gestation on fetal NRG1-ErbB4 expression and behavior in juvenile and adult mice offspring. Brain Behav Immun 79:207–215. https://doi.org/10.1016/j.bbi.2019.02.002
Daher I, Le Dieu-Lugon B, Lecointre M, Dupré N, Voisin C, Leroux P, Dourmap N, Gonzalez BJ, Marret S, Leroux-Nicollet I, Cleren C (2018) Time- and sex-dependent efficacy of magnesium sulfate to prevent behavioral impairments and cerebral damage in a mouse model of cerebral palsy. Neurobiol Dis 120:151–164. https://doi.org/10.1016/j.nbd.2018.08.020
Ginsberg Y, Khatib N, Weiss B, Arison S, Ross MG, Weiner Z, Beloosesky R (2017) Magnesium sulfate (MG) prevents maternal inflammation induced offspring cerebral injury evident on MRI but not via IL-1β. Neuroscience 353:98–105. https://doi.org/10.1016/j.neuroscience.2017.03.046
Guma E, Plitman E, Chakravarty MM (2019) The role of maternal immune activation in altering the neurodevelopmental trajectories of offspring: a translational review of neuroimaging studies with implications for autism spectrum disorder and schizophrenia. Neurosci Biobehav Rev 104:141–157. https://doi.org/10.1016/j.neubiorev.2019.06.020
Guo F, Maeda Y, Ko EM, Delgado M, Horiuchi M, Soulika A, Miers L, Burns T, Itoh T, Shen H, Lee E, Sohn J, Pleasure D (2012) Disruption of NMDA receptors in oligodendroglial lineage cells does not alter their susceptibility to experimental autoimmune encephalomyelitis or their normal development. J Neurosci 32:639–645. https://doi.org/10.1523/JNEUROSCI.4073-11.2012
Hemmerle AM, Ahlbrand R, Bronson SL, Lundgren KH, Richtand NM, Seroogy KB (2015) Modulation of schizophrenia-related genes in the forebrain of adolescent and adult rats exposed to maternal immune activation. Schizophr Res 168:411–420. https://doi.org/10.1016/j.schres.2015.07.006
Iriye BK, Gregory KD, Saade GR, Grobman WA, Brown HL (2017) Quality measures in high-risk pregnancies: executive Summary of a Cooperative Workshop of the Society for Maternal-Fetal Medicine, National Institute of Child Health and Human Development, and the American College of Obstetricians and Gynecologists, in: American Journal of Obstetrics and Gynecology. Elsevier Inc., pp B2–B25. https://doi.org/10.1016/j.ajog.2017.07.018
Iwakura Y, Nawa H (2013) ErbB1-4-dependent EGF/neuregulin signals and their cross talk in the central nervous system: pathological implications in schizophrenia and Parkinson’s disease. Front Cell Neurosci 7:1–13. https://doi.org/10.3389/fncel.2013.00004
Izvolskaia M, Sharova V (2018) Prenatal programming of neuroendocrine system development by lipopolysaccharide: long-term effects. https://doi.org/10.3390/ijms19113695
Kampa BM, Clements J, Jonas P, Stuart GJ (2004) Kinetics of Mg2+ unblock of NMDA receptors: implications for spike-timing dependent synaptic plasticity. J Physiol 556:337–345. https://doi.org/10.1113/jphysiol.2003.058842
Knuesel I, Chicha L, Britschgi M, Schobel SA, Bodmer M, Hellings JA, Toovey S, Prinssen EP (2014) Maternal immune activation and abnormal brain development across CNS disorders. Nat Rev Neurol 10:643–660. https://doi.org/10.1038/nrneurol.2014.187
Lamhot VB, Khatib N, Ginsberg Y, Anunu R, Richter-Levin G, Weiner Z, Ross MG, Divon MY, Hallak M, Beloosesky R (2015) Magnesium sulfate prevents maternal inflammation-induced impairment of learning ability and memory in rat offspring presented in poster format at the 34th annual meeting of the Society for Maternal-Fetal Medicine, New Orleans, LA, Feb. 3-8, 2014. Am J Obstet Gynecol 213:851.e1–851.e8. https://doi.org/10.1016/j.ajog.2015.07.042
Lecuyer M, Rubio M, Chollat C, Lecointre M, Jégou S, Leroux P, Cleren C, Leroux-Nicollet I, Marpeau L, Vivien D, Marret S, Gonzalez BJ (2017) Experimental and clinical evidence of differential effects of magnesium sulfate on neuroprotection and angiogenesis in the fetal brain. Pharmacol Res Perspect 5:1–20. https://doi.org/10.1002/prp2.315
Lingam I, Robertson NJ (2018) Magnesium as a neuroprotective agent: a review of its use in the fetus, term infant with neonatal encephalopathy, and the adult stroke patient. Dev Neurosci 40:1–12. https://doi.org/10.1159/000484891
Mor G, Cardenas I, Abrahams V, Guller S (2012) Author manuscript the implantation site. Ann N Y Acad Sci 1221:80–87. https://doi.org/10.1111/j.1749-6632.2010.05938.x.Inflammation
Muraki K, Tanigaki K (2015) Neuronal migration abnormalities and its possible implications for schizophrenia. Front Neurosci 9:1–10. https://doi.org/10.3389/fnins.2015.00074
Nadeau-Vallée (2016) Sterile inflammation and pregnancy complications : a review. Reproduction. https://doi.org/10.1530/REP-16-0453
Nicola NA, Babon JJ (2015) Leukemia inhibitory factor (LIF). Cytokine Growth Factor Rev 26:533–544. https://doi.org/10.1016/j.cytogfr.2015.07.001
Patten BA, Peyrin JM, Weinmaster G, Corfas G (2003) Sequential signaling through Notch1 and erbB receptors mediates radial glia differentiation. J Neurosci 23:6132–6140. https://doi.org/10.1523/jneurosci.23-14-06132.2003
Shaw OEF, Yager JY (2019) Preventing childhood and lifelong disability: maternal dietary supplementation for perinatal brain injury. Pharmacol Res 139:228–242. https://doi.org/10.1016/j.phrs.2018.08.022
Shaw JC, Crombie GK, Zakar T, Palliser HK, Hirst JJ (2020) Perinatal compromise contributes to programming of GABAergic and glutamatergic systems leading to long-term effects on offspring behaviour. J Neuroendocrinol 32:1–12. https://doi.org/10.1111/jne.12814
Simamura E, Shimada H, Higashi N, Uchishiba M, Otani H, Hatta T (2010) Maternal leukemia inhibitory factor (LIF) promotes fetal neurogenesis via a LIF-ACTH-LIF signaling relay pathway. Endocrinology 151:1853–1862. https://doi.org/10.1210/en.2009-0985
Solek CM, Farooqi N, Verly M, Lim TK, Ruthazer ES (2018) Maternal immune activation in neurodevelopmental disorders. Dev Dyn 247:588–619. https://doi.org/10.1002/dvdy.24612
Tam Tam HB, Dowling O, Xue X, Lewis D, Rochelson B, Metz CN (2011) Magnesium sulfate ameliorates maternal and fetal inflammation in a rat model of maternal infection. Am J Obstet Gynecol 204:364.e1–364.e8. https://doi.org/10.1016/j.ajog.2010.11.006
Tsukada T, Simamura E, Shimada H, Arai T, Higashi N, Akai T, Iizuka H, Hatta T (2015) The suppression of maternal-fetal leukemia inhibitory factor signal relay pathway by maternal immune activation impairs brain development in mice. PLoS One 10:1–14. https://doi.org/10.1371/journal.pone.0129011
Author information
Authors and Affiliations
Corresponding authors
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 27 kb)
Rights and permissions
About this article
Cite this article
Dabbah-Assadi, F., Khatib, N., Ginsberg, Y. et al. Short-Term Effect of MgSO4 on the Expression of NRG-ErbB, Dopamine, GABA, and Glutamate Systems in the Fetal Rat Brain. J Mol Neurosci 71, 446–454 (2021). https://doi.org/10.1007/s12031-020-01665-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12031-020-01665-x