Abstract
Background
Cushing’s Syndrome (CS) is associated with increased cardiovascular morbidity and mortality. In endogenous CS, cardiovascular mortality remains increased for up to 15 years post remission of hypercortisolism. Similarly, patients with exogenous CS have 4-fold increased incidence of cardiovascular events, regardless of pre-existing cardiovascular disease (CVD).
Objective
To present the pathophysiology, prognosis, clinical and imaging phenotype of cardiac disease in CS.
Methods
A Pubmed search for cardiac disease in CS over the last 20 years was conducted using combinations of relevant terms. Preclinical and clinical studies, as well as review papers reporting on subclinical heart failure (HF), cardiomyopathy, coronary heart disease (CHD), and cardiovascular imaging were selected.
Results
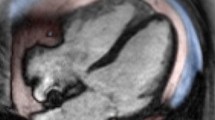
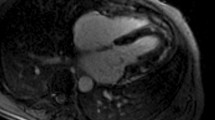
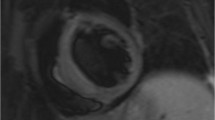
Cardiac disease in CS is associated with direct mineralocorticoid and glucocorticoid receptor activation, increased responsiveness to angiotensin II, ectopic epicardial adiposity, arterial stiffness and endothelial dysfunction, as well as with diabetes mellitus, hypertension, hyperlipidemia, obesity and prothrombotic diathesis. Subclinical HF and cardiomyopathy are principally related to direct glucocorticoid (GC) effects and markedly improve or regress post hypercortisolism remission. In contrast, CHD is related to both direct GC effects and CS comorbidities and persists post cure. In patients without clinical evidence of CVD, echocardiography and cardiac magnetic resonance (CMR) imaging reveal left ventricular hypertrophy, fibrosis, diastolic and systolic dysfunction, with the latter being underestimated by echocardiography. Finally, coronary microvascular disease is encountered in one third of cases.
Conclusion
Cardiovascular imaging is crucial in evaluation of cardiac involvement in CS. CMR superiority in terms of reproducibility, operator independency, unrestricted field of view and capability of tissue characterisation makes this modality ideal for future studies.



Similar content being viewed by others
References
L.K. Nieman, B.M.K. Biller, J.W. Findling, J. Newell-Price, M.O. Savage, P.M. Stewart, V.M. Montori, H. Edwards, The diagnosis of Cushing’s syndrome: An endocrine society clinical practice guideline. J. Clin. Endocrinol. Metab. 93, 1526–1540 (2008). https://doi.org/10.1210/jc.2008-0125
L. Fardet, B. Fève, Systemic glucocorticoid therapy: A review of its metabolic and cardiovascular adverse events. Drugs 74, 1731–1745 (2014). https://doi.org/10.1007/s40265-014-0282-9
O.A. Hakami, S. Ahmed, N. Karavitaki, Epidemiology and mortality of Cushing’s syndrome. Best Pract.Res. Clin. Endocrinol. Metabol. 35, (2021). https://doi.org/10.1016/j.beem.2021.101521
R. Pivonello, A.M. Isidori, M.C. De Martino, J. Newell-Price, B.M.K. Biller, A. Colao, Complications of Cushing’s syndrome: State of the art. Lancet Diabetes Endocrinol. 4, 611–629 (2016). https://doi.org/10.1016/S2213-8587(16)00086-3
A.M. Isidori, C. Graziadio, R.M. Paragliola, A. Cozzolino, A.G. Ambrogio, A. Colao, S.M. Corsello, R. Pivonello, The hypertension of Cushing’s syndrome: Controversies in the pathophysiology and focus on cardiovascular complications. J. Hypertens. 33, 44–60 (2015). https://doi.org/10.1097/HJH.0000000000000415
M. Kadmiel, J.A. Cidlowski, Glucocorticoid receptor signaling in health and disease. Trends Pharm. Sci. 34, 518–530 (2013). https://doi.org/10.1016/j.tips.2013.07.003
E.B. Geer, W. Shen, D. Gallagher, M. Punyanitya, H.C. Looker, K.D. Post, P.U. Freda, MRI assessment of lean and adipose tissue distribution in female patients with Cushing’s disease. Clin. Endocrinol. (Oxf.) 73, 469–475 (2010). https://doi.org/10.1111/j.1365-2265.2010.03829.x
A.G. Rockall, S.A. Sohaib, D. Evans, G. Kaltsas, A.M. Isidori, J.P. Monson, G.M. Besser, A.B. Grossman, R.H. Reznek, Computed tomography assessment of fat distribution in male and female patients with Cushing’s syndrome. Eur. J. Endocrinol. 149, 561–567 (2003)
C. Scaroni, M. Zilio, M. Foti, M. Boscaro, Glucose metabolism abnormalities in cushing syndrome: from molecular basis to clinical management. Endocr. Rev. 38, 189–219 (2017). https://doi.org/10.1210/er.2016-1105
M. Fleseriu, R. Auchus, I. Bancos, A. Ben-Shlomo, J. Bertherat, N.R. Biermasz, C.L. Boguszewski, M.D. Bronstein, M. Buchfelder, J.D. Carmichael, F.F. Casanueva, F. Castinetti, P. Chanson, J. Findling, M. Gadelha, E.B. Geer, A. Giustina, A. Grossman, M. Gurnell, K. Ho, A.G. Ioachimescu, U.B. Kaiser, N. Karavitaki, L. Katznelson, D.F. Kelly, A. Lacroix, A. McCormack, S. Melmed, M. Molitch, P. Mortini, J. Newell-Price, L. Nieman, A.M. Pereira, S. Petersenn, R. Pivonello, H. Raff, M. Reincke, R. Salvatori, C. Scaroni, I. Shimon, C.A. Stratakis, B. Swearingen, A. Tabarin, Y. Takahashi, M. Theodoropoulou, S. Tsagarakis, E. Valassi, E.V. Varlamov, G. Vila, J. Wass, S.M. Webb, M.C. Zatelli, B.M.K. Biller, Consensus on diagnosis and management of Cushing’s disease: a guideline update. Lancet Diabetes Endocrinol. 9, 847–875 (2021). https://doi.org/10.1016/S2213-8587(21)00235-7
E. Gomez-Sanchez, C.E. Gomez-Sanchez, The multifaceted mineralocorticoid receptor. Compr. Physiol. 4, 965–994 (2014). https://doi.org/10.1002/cphy.c130044
A. Coulden, R. Hamblin, J. Wass, N. Karavitaki, Cardiovascular health and mortality in Cushing’s disease. Pituitary 25, 750–753 (2022). https://doi.org/10.1007/s11102-022-01258-4
E.V. Varlamov, F. Langlois, G. Vila, M. Fleseriu, Cardiovascular risk assessment, thromboembolism, and infection prevention in Cushing’s syndrome: A practical approach. Eur. J. Endocrinol. 184, R207–R224 (2021). https://doi.org/10.1530/EJE-20-1309
L.K. Nieman, Hypertension and cardiovascular mortality in patients with cushing syndrome. Endocrinol. Metab. Clin. North Am. 48, 717–725 (2019). https://doi.org/10.1016/j.ecl.2019.08.005
R.N. Clayton, P.W. Jones, R.C. Reulen, P.M. Stewart, Z.K. Hassan-Smith, G. Ntali, N. Karavitaki, O.M. Dekkers, A.M. Pereira, M. Bolland, I. Holdaway, J. Lindholm, Mortality in patients with Cushing’s disease more than 10 years after remission: A multicentre, multinational, retrospective cohort study. Lancet Diabetes Endocrinol. 4, 569–576 (2016). https://doi.org/10.1016/S2213-8587(16)30005-5
G. Ntali, A. Asimakopoulou, T. Siamatras, J. Komninos, D. Vassiliadi, M. Tzanela, S. Tsagarakis, A.B. Grossman, J.A.H. Wass, N. Karavitaki, Mortality in Cushing’s syndrome: Systematic analysis of a large series with prolonged follow-up. Eur. J. Endocrinol. 169, 715–723 (2013). https://doi.org/10.1530/EJE-13-0569
O. Ragnarsson, D.S. Olsson, E. Papakokkinou, D. Chantzichristos, P. Dahlqvist, E. Segerstedt, T. Olsson, M. Petersson, K. Berinder, S. Bensing, C. Hoybye, B. Eden-Engstrom, P. Burman, L. Bonelli, C. Follin, D. Petranek, E.M. Erfurth, J. Wahlberg, B. Ekman, A.K. Akerman, E. Schwarcz, I.L. Bryngelsson, G. Johannsson, Overall and disease-specific mortality in patients with cushing disease: A Swedish nationwide study. J. Clin. Endocrinol. Metab. 104, 2375–2384 (2019). https://doi.org/10.1210/jc.2018-02524
R.N. Clayton, D. Raskauskiene, R.C. Reulen, P.W. Jones, Mortality and morbidity in Cushing’s disease over 50 Years in Stoke-on-Trent, UK: Audit and meta-analysis of literature. J. Clin. Endocrinol. Metab. 96, 632–642 (2011). https://doi.org/10.1210/jc.2010-1942
D. Bengtsson, O. Ragnarsson, K. Berinder, P. Dahlqvist, B.E. Engström, B. Ekman, C. Höybye, J. Järås, S. Valdemarsson, P. Burman, J. Wahlberg, Increased Mortality Persists after Treatment of Cushing’s Disease: A Matched Nationwide Cohort Study. J. Endocr. Soc. 6, 1–10 (2022). https://doi.org/10.1210/jendso/bvac045
L. Fardet, I. Petersen, I. Nazareth, Risk of cardiovascular events in people prescribed glucocorticoids with iatrogenic Cushing’s syndrome: Cohort study. BMJ (Online). 345, (2012). https://doi.org/10.1136/bmj.e4928
R.N. Clayton, Cardiovascular complications of Cushing’s syndrome: Impact on morbidity and mortality. J Neuroendocrinol. 34, (2022). https://doi.org/10.1111/jne.13175
P.M. Toja, G. Branzi, F. Ciambellotti, P. Radaelli, M. De Martin, L.M. Lonati, M. Scacchi, G. Parati, F. Cavagnini, F.P. Giraldi, Clinical relevance of cardiac structure and function abnormalities in patients with Cushing’s syndrome before and after cure. Clin. Endocrinol. 76, 332–338 (2012). https://doi.org/10.1111/j.1365-2265.2011.04206.x
A.M. Pereira, V. Delgado, J.A. Romijn, J.W.A. Smit, J.J. Bax, R.A. Feelders, Cardiac dysfunction is reversed upon successful treatment of Cushing’s syndrome. Eur. J. Endocrinol. 162, 331–340 (2010). https://doi.org/10.1530/EJE-09-0621
M.L. Muiesan, M. Lupia, M. Salvetti, C. Grigoletto, N. Sonino, M. Boscaro, E. Agabiti Rosei, F. Mantero, F. Fallo, Left ventricular structural and functional characteristics in Cushing’s syndrome. J. Am. Coll. Cardiol. 41, 2275–2279 (2003). https://doi.org/10.1016/S0735-1097(03)00493-5
E. Avenatti, A. Rebellato, A. Iannaccone, M. Battocchio, F. Dassie, F. Veglio, A. Milan, F. Fallo, Left ventricular geometry and 24-h blood pressure profile in Cushing’s syndrome. Endocrine 55, 547–554 (2017). https://doi.org/10.1007/s12020-016-0986-6
K.H. Yiu, N.A. Marsan, V. Delgado, N.R. Biermasz, E.R. Holman, J.W.A. Smit, R.A. Feelders, J.J. Bax, A.M. Pereira, Increased myocardial fibrosis and left ventricular dysfunction in Cushing’s syndrome. Eur. J. Endocrinol. 166, 27–34 (2012). https://doi.org/10.1530/EJE-11-0601
C. Roux, N. Kachenoura, Z. Raissuni, E. Mousseaux, J. Young, M.J. Graves, C. Jublanc, P. Cluzel, P. Chanson, P. Kamenický, A. Redheuil, Effects of cortisol on the heart: characterization of myocardial involvement in Cushing’s disease by longitudinal cardiac MRI T1 mapping. J. Magn. Reson. Imaging 45, 147–156 (2017). https://doi.org/10.1002/jmri.25374
P. Kamenický, A. Redheuil, C. Roux, S. Salenave, N. Kachenoura, Z. Raissouni, L. Macron, L. Guignat, C. Jublanc, A. Azarine, S. Brailly, J. Young, E. Mousseaux, P. Chanson, Cardiac structure and function in cushing’s syndrome: A cardiac magnetic resonance imaging study. J. Clin. Endocrinol. Metab. 99, E2144–E2153 (2014). https://doi.org/10.1210/jc.2014-1783
K.E. Sheppard, D.J. Autelitano, 1-Hydroxysteroid Dehydrogenase 1 Transforms 11-Dehydrocorticosterone into Transcriptionally Active Glucocorticoid in Neonatal Rat Heart. Endocrinology 143, 198–204 (2002)
G.A. Gray, C.I. White, R.F.P. Castellan, S.J. McSweeney, K.E. Chapman, Getting to the heart of intracellular glucocorticoid regeneration: 11β-HSD1 in the myocardium. J. Mol. Endocrinol. 58, R1–R13 (2017). https://doi.org/10.1530/JME-16-0128
K. Nagata, K. Obata, J. Xu, S. Ichihara, A. Noda, H. Kimata, T. Kato, H. Izawa, T. Murohara, M. Yokota, Mineralocorticoid receptor antagonism attenuates cardiac hypertrophy and failure in low-aldosterone hypertensive rats. Hypertension 47, 656–664 (2006). https://doi.org/10.1161/01.HYP.0000203772.78696.67
A. Frustaci, C. Letizia, R. Verardo, C. Grande, L. Petramala, M.A. Russo, C. Chimenti, Cushing syndrome cardiomyopathy.clinicopathologic impact of cortisol normalization. Circ. Cardiovasc. Imaging. 9, (2016)
A. Frustaci, C. Letizia, R. Verardo, C. Grande, C. Calvieri, M.A. Russo, C. Chimenti, Atrogin-1 pathway activation in cushing syndrome cardiomyopathy. J. Am. Coll. Cardiol. 67, 116–117 (2016). https://doi.org/10.1016/j.jacc.2015.10.040
F. Maurice, B. Gaborit, C. Vincentelli, I. Abdesselam, M. Bernard, T. Graillon, F. Kober, T. Brue, F. Castinetti, A. Dutour, Cushing syndrome is associated with subclinical LV dysfunction and increased epicardial adipose tissue. J. Am. Coll. Cardiol. 72, 2276–2277 (2018). https://doi.org/10.1016/j.jacc.2018.07.096
P. Wolf, B. Marty, K. Bouazizi, N. Kachenoura, C. Piedvache, A. Blanchard, S. Salenave, M. Prigent, C. Jublanc, C. Ajzenberg, C. Droumaguet, J. Young, A.L. Lecoq, E. Kuhn, H. Agostini, S. Trabado, P.G. Carlier, B. Fève, A. Redheuil, P. Chanson, P. Kamenický, Epicardial and pericardial adiposity without myocardial steatosis in cushing syndrome. J. Clin. Endocrinol. Metab. 106, 3505–3514 (2021). https://doi.org/10.1210/clinem/dgab556
F. Tona, M. Boscaro, M. Barbot, L. Maritan, G. Famoso, L.C. Dal, R. Montisci, F. Fallo, S. Iliceto, C. Scaroni, New insights to the potential mechanisms driving coronary flow reserve impairment in Cushing’s syndrome: A pilot noninvasive study by transthoracic Doppler echocardiography. Microvasc. Res. 128, (2020). https://doi.org/10.1016/j.mvr.2019.103940
R. Sagara, T. Inoue, N. Sonoda, C. Yano, M. Motoya, H. Umakoshi, R. Sakamoto, Y. Ogawa, Association between cortisol and left ventricular diastolic dysfunction in patients with diabetes mellitus. J. Diabetes Investig. 13, 344–350 (2022). https://doi.org/10.1111/jdi.13653
O.M. Dekkers, E. Horváth-Puh́o, J.O.L. Jørgensen, S.C. Cannegieter, V. Ehrenstein, J.P. Vandenbroucke, A.M. Pereira, H.T. Srøensen, Multisystem morbidity and mortality in Cushing’s syndrome: A cohort study. J. Clin. Endocrinol. Metab. 98, 2277–2284 (2013). https://doi.org/10.1210/jc.2012-3582
S. Miao, L. Lu, L. Li, Y. Wang, Z. Lu, H. Zhu, L. Wang, L. Duan, X. Xing, Y. Yao, M. Feng, R. Wang, Clinical characteristics for the improvement of cushing’s syndrome complicated with cardiomyopathy after treatment with a literature review. Front. Cardiovasc. Med. 8, (2021). https://doi.org/10.3389/fcvm.2021.777964
M. Sakota, S. Tatebe, K. Sugimura, T. Aoki, S. Yamamoto, H. Sato, N. Kikuchi, R. Konno, Y. Terui, K. Satoh, Y. Tezuka, R. Morimoto, M. Saito, S. Kuniyoshi, H. Shimokawa, Successful management of acute congestive heart failure by emergent caesarean section followed by adrenalectomy in a pregnant woman with cushing’s syndrome-induced cardiomyopathy. Intern. Med. 58, 2819–2824 (2019). https://doi.org/10.2169/internalmedicine.2427-18
M. Peppa, I. Ikonomidis, D. Hadjidakis, V. Pikounis, I. Paraskevaidis, T. Economopoulos, S.A. Raptis, D.T. Kremastinos, Dilated cardiomyopathy as the predominant feature of Cushing’s syndrome. Am. J. Med. Sci. 338, 252–253 (2009). https://doi.org/10.1097/MAJ.0b013e3181a927e0
T. Sheikh, H. Shuja, S.R. Zaidi, A. Haque, Glucocorticoid-induced cardiomyopathy: Unexpected conclusion. BMJ Case Rep. 13, (2020). https://doi.org/10.1136/bcr-2020-237173
L. Marchand, B. Segrestin, M. Lapoirie, V. Favrel, J. Dementhon, E. Jouanneau, G. Raverot, Dilated Cardiomyopathy Revealing Cushing Disease. A Case Report and Literature Review. Med. (U. S.) 94, e2011 (2015). https://doi.org/10.1097/MD.0000000000002011
T.M. Hey, J.S. Dahl, T.H. Brix, E.V. Søndergaard, D. Thomas, M. Hey, Biventricular hypertrophy and heart failure as initial presentation of Cushing’s disease. BMJ Case Rep. (2013). https://doi.org/10.1136/bcr-2013
L. Wei, T.M. Macdonald, B.R. Walker, Taking glucocorticoids by prescription is associated with subsequent cardiovascular disease background: glucocorticoids have adverse systemic effects, in. Ann. Intern. Med. 141, 764–770 (2004)
M.J. Bolland, I.M. Holdaway, J.E. Berkeley, S. Lim, W.J. Dransfield, J.V. Conaglen, M.S. Croxson, G.D. Gamble, P.J. Hunt, R.J. Toomath, Mortality and morbidity in Cushing’s syndrome in New Zealand. Clin. Endocrinol. 75, 436–442 (2011). https://doi.org/10.1111/j.1365-2265.2011.04124.x
P.C. Souverein, A. Berard, T.P. Van Staa, C. Cooper, A.C.G. Egberts, H.G.M. Leufkens, B.R. Walker, Use of oral glucocorticoids and risk of cardiovascular and cerebrovascular disease in a population based case-control study. Heart 90, 859–865 (2004). https://doi.org/10.1136/hrt.2003.020180
N.M. Neary, O.J. Booker, B.S. Abel, J.R. Matta, N. Muldoon, N. Sinaii, R.I. Pettigrew, L.K. Nieman, A.M. Gharib, Hypercortisolism is associated with increased coronary arterial atherosclerosis: Analysis of noninvasive coronary angiography using multidetector computerized tomography. J. Clin. Endocrinol. Metab. 98, 2045–2052 (2013). https://doi.org/10.1210/jc.2012-3754
M.J. Barahona, E. Resmini, D. Viladés, G. Pons-Lladó, R. Leta, T. Puig, S.M. Webb, Coronary artery disease detected by multislice computed tomography in patients after long-term cure of cushing’s syndrome. J. Clin. Endocrinol. Metab. 98, 1093–1099 (2013). https://doi.org/10.1210/jc.2012-3547
M. De Leo, R. Pivonello, R.S. Auriemma, A. Cozzolino, P. Vitale, C. Simeoli, M.C. De Martino, G. Lombardi, A. Colao, Cardiovascular disease in Cushing’s syndrome: Heart versus vasculature. Neuroendocrinology 92, 50–54 (2010). https://doi.org/10.1159/000318566
C. Yano, M. Yokomoto-Umakoshi, M. Fujita, H. Umakoshi, S. Yano, N. Iwahashi, S. Katsuhara, H. Kaneko, M. Ogata, T. Fukumoto, E. Terada, Y. Matsuda, R. Sakamoto, Y. Ogawa, Coexistence of bone and vascular disturbances in patients with endogenous glucocorticoid excess. Bone Rep. 17, (2022). https://doi.org/10.1016/j.bonr.2022.101610
M. Battocchio, A. Rebellato, A. Grillo, F. Dassie, P. Maffei, S. Bernardi, B. Fabris, R. Carretta, F. Fallo, Ambulatory arterial stiffness indexes in cushing’s syndrome. Horm. Metab. Res. 49, 214–220 (2017). https://doi.org/10.1055/s-0043-100385
I. Akaza, T. Yoshimoto, K. Tsuchiya, Y. Hirata, Endothelial dysfunction associated with hypercortisolism is reversible in cushing’s syndrome. Endocr. J. 57, 245–252 (2010)
G. Kirilov, A. Tomova, L. Dakovska, P. Kumanov, A. Shinkov, A.S. Alexandrov, Elevated plasma endothelin as an additional cardiovascular risk factor in patients with Cushing’s syndrome. Eur. J. Endocrinol. 149, 549–553 (2003)
A. Faggiano, D. Melis, R. Alfieri, M.C. De Martino, M. Filippella, F. Milone, G. Lombardi, A. Colao, R. Pivonello, Sulfur amino acids in Cushing’s disease: Insight in homocysteine and taurine levels in patients with active and cured disease. J. Clin. Endocrinol. Metabol. 90, 6616–6622 (2005). https://doi.org/10.1210/jc.2005-0656
G. Markousis‐Mavrogenis, F. Bacopoulou, C. Mavragani, P. Voulgari, G. Kolovou, G.D. Kitas, G.P. Chrousos, S.I. Mavrogeni, Coronary microvascular disease: The “Meeting Point” of Cardiology, Rheumatology and Endocrinology. Eur J Clin Invest. 52, (2022). https://doi.org/10.1111/eci.13737
R.B. Devereux, B. Dahlöf, E. Gerdts, K. Boman, M.S. Nieminen, V. Papademetriou, J. Rokkedal, K.E. Harris, J.M. Edelman, K. Wachtell, Regression of hypertensive left ventricular hypertrophy by Losartan compared with atenolol: The Losartan Intervention for Endpoint Reduction in Hypertension (LIFE) trial. Circulation 110, 1456–1462 (2004). https://doi.org/10.1161/01.CIR.0000141573.44737.5A
F. Grothues, G.C. Smith, J.C. Moon, M. BCh, N.G. Bellenger, P. Collins, H.U. Klein, D.J. Pennell, Comparison of Interstudy Reproducibility of Cardiovascular Magnetic Resonance With Two-Dimensional Echocardiography in Normal Subjects and in Patients With Heart Failure or Left Ventricular Hypertrophy. Am. J. Cardiol. 90, 29–34 (2002)
J.H. Park, K. Negishi, D.H. Kwon, Z.B. Popovic, R.A. Grimm, T.H. Marwick, Validation of global longitudinal strain and strain rate as reliable markers of right ventricular dysfunction: Comparison with cardiac magnetic resonance and outcome. J. Cardiovasc Ultrasound 22, 113–120 (2014). https://doi.org/10.4250/jcu.2014.22.3.113
M. Aurich, F. André, M. Keller, S. Greiner, A. Hess, S.J. Buss, H.A. Katus, D. Mereles, Assessment of left ventricular volumes with echocardiography and cardiac magnetic resonance imaging: Real-life evaluation of standard versus new semiautomatic methods. J. Am. Soc. Echocardiogr. 27, 1017–1024 (2014). https://doi.org/10.1016/j.echo.2014.07.006
L. Perdrix, N. Mansencal, B. Cocheteux, G. Chatellier, A. Bissery, B. Diebold, E. Mousseaux, E. Abergel, How to calculate left ventricular mass in routine practice? An echocardiographic versus cardiac magnetic resonance study. Arch. Cardiovasc Dis. 104, 343–351 (2011). https://doi.org/10.1016/j.acvd.2011.04.003
H. Oe, T. Hozumi, K. Arai, Y. Matsumura, K. Negishi, K. Sugioka, K. Ujino, Y. Takemoto, Y. Inoue, J. Yoshikawa, Comparison of accurate measurement of left ventricular mass in patients with hypertrophied hearts by real-time three-dimensional echocardiography versus magnetic resonance imaging. Am. J. Cardiol. 95, 1263–1267 (2005). https://doi.org/10.1016/j.amjcard.2005.01.065
K. Kusunose, D.H. Kwon, H. Motoki, S.D. Flamm, T.H. Marwick, Comparison of three-dimensional echocardiographic findings to those of magnetic resonance imaging for determination of left ventricular mass in patients with ischemic and non-ischemic cardiomyopathy. Am. J. Cardiol. 112, 604–611 (2013). https://doi.org/10.1016/j.amjcard.2013.04.028
K. Alfakih, K. Walters, T. Jones, J. Ridgway, A.S. Hall, M. Sivananthan, New gender-specific partition values for ECG criteria of left ventricular hypertrophy: Recalibration against cardiac MRI. Hypertension 44, 175–179 (2004). https://doi.org/10.1161/01.HYP.0000135249.66192.30
K. Moschetti, R.Y. Kwong, S.E. Petersen, M. Lombardi, J. Garot, D. Atar, F.E. Rademakers, L.M. Sierra-Galan, S. Mavrogeni, K. Li, J.L. Fernandes, S. Schneider, C. Pinget, Y. Ge, P. Antiochos, C. Deluigi, O. Bruder, H. Mahrholdt, J. Schwitter, Cost-Minimization Analysis for Cardiac Revascularization in 12 Health Care Systems Based on the EuroCMR/SPINS Registries. JACC Cardiovasc Imaging 15, 607–625 (2022). https://doi.org/10.1016/j.jcmg.2021.11.008
S.I. Mavrogeni, A. Kallifatidis, T. Kourtidou, N. Lama, A. Christidi, E. Detorakis, G. Chatzantonis, T. Vrachliotis, T. Karamitsos, K. Kouskouras, N. Kelekis. Cardiovascular Magnetic Resonance for evaluation of patients with cardiovascular disease. An overview of current indications, limitations and procedures. Hell. J. Cardiol. (2023). https://doi.org/10.1016/j.hjc.2023.01.003
S. Mavrogeni, A. Pepe, R. Nijveldt, N. Ntusi, L.M. Sierra-Galan, K. Bratis, J. Wei, M. Mukherjee, G. Markousis-Mavrogenis, L. Gargani, L.E. Sade, N. Ajmone-Marsan, P. Seferovic, E. Donal, M. Nurmohamed, M.M. Cerinic, P. Sfikakis, G. Kitas, J. Schwitter, J.A.C. Lima, D. Dawson, M. Dweck, K.H. Haugaa, N. Keenan, J. Moon, I. Stankovic, E. Donal, B. Cosyns, Cardiovascular magnetic resonance in autoimmune rheumatic diseases: a clinical consensus document by the European Association of Cardiovascular Imaging. Eur. Heart J. Cardiovasc Imaging 23, e308–e322 (2022). https://doi.org/10.1093/ehjci/jeac134
K. Ehrlich, C. Morbach, T. Reiter, P.U. Heuschmann, A. Hannemann, M. Fassnacht, S. Störk, S. Hahner, T. Deutschbein, Rationale and design of the cardiovascular status in patients with endogenous cortisol excess study (CV-CORT-EX): a prospective non-interventional follow-up study. BMC Endocr Disord. 21, (2021). https://doi.org/10.1186/s12902-020-00665-7
Author contributions
S.M. and A.V. conceived the idea of the manuscript. Literature search was performed by M.M., G.M.-M. and S.M. The first draft of the manuscript was written by M.M. and G.M.-M. The manuscript was revised for intellectual content by S.M., S.A.P. and A.V. All authors revised and approved the final version of manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Moustaki, M., Markousis-Mavrogenis, G., Vryonidou, A. et al. Cardiac disease in Cushing’s syndrome. Emphasis on the role of cardiovascular magnetic resonance imaging. Endocrine 83, 548–558 (2024). https://doi.org/10.1007/s12020-023-03623-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12020-023-03623-0