Abstract
Purpose
Acromegaly is closely related to increased oxidative stress and endothelial dysfunction (ED). This study aimed to evaluate, for the first time in the literature, signal peptide-CUB-EGF domain-containing protein 1 (SCUBE-1) and endothelial nitric oxide synthase e(NOS) levels in the setting of acromegaly.
Method
A total of 56 acromegaly patients and a control group composed of 30 healthy volunteers were included in this study. In the postoperative follow-up, patients were grouped as active or in-remission according to their GH and IGF-1 levels in oral glucose stimulation test (OGST). After detailed physical examination of acromegaly patients and the control subjects, 8-hour fasting blood samples were collected to evaluate biochemical parameters including lipid profile, anterior pituitary hormones, and SCUBE-1 and e(NOS) levels.
Results
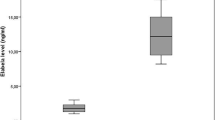
Inactive and active acromegaly was noted in 78.6% and 21.4% of patients, respectively. The median (min-max) SCUBE-1 levels were significantly higher in the inactive acromegaly and active acromegaly groups than in the control group (1.6(0.4–2.4) and 1.8(1.1–2.5) vs. 0.4(0.2–1.0) ng/mL, respectively, p < 0.001 for each). The median (min-max) e(NOS) levels were significantly higher in the inactive acromegaly and active acromegaly groups than in the control group (132.7 (26.8–602.9) and 137.3 (69.7–488.7) vs. 83.9 (16.4–218.7) pg/mL, p = 0.018 and p = 0.048, respectively). We have also detected positive correlations of e(NOS) with leukocyte (r = 0.307, p = 0.021) and neutrophil counts (r = 0.309, p = 0.021).
Conclusion
Our study revealed for the first time in literature that SCUBE-1 levels, being a novel marker for ED, were significantly higher in acromegaly patients than in control subjects. When supported with clinical studies, SCUBE-1can be used as an early indicator of endothelial damage in acromegaly patients.


Similar content being viewed by others
References
A. Fernandez, N. Karavitaki, J.A. Wass, Prevalence of pituitary adenomas: a community-based, cross-sectional study in Banbury (Oxfordshire, UK). Clin. Endocrinol. (Oxf) 72(3), 377–382 (2010). https://doi.org/10.1111/j.1365-2265.2009.03667.x
S. Bekkering, R.J.W. Arts, B. Novakovic, I. Kourtzelis, C.D.C.C. van der Heijden, Y. Li, C.D. Popa, R. Ter Horst, J. van Tuijl, R.T. Netea-Maier, F.L. van de Veerdonk, T. Chavakis, L.A.B. Joosten, J.W.M. van der Meer, H. Stunnenberg, N.P. Riksen, M.G. Netea, Metabolic Induction of Trained Immunity through the Mevalonate Pathway. Cell 172(1-2), 135–146.e9 (2018). https://doi.org/10.1016/j.cell.2017.11.025
T.L.C. Wolters, M.G. Netea, A.R.M.M. Hermus, J.W.A. Smit, R.T. Netea-Maier, IGF1 potentiates the pro-inflammatory response in human peripheral blood mononuclear cells via MAPK. J. Mol. Endocrinol. 59(2), 129–139 (2017). https://doi.org/10.1530/JME-17-0062
L. Liu, D. Leitner, Simultaneous pursuit of innovation and efficiency in complex engineering projects—A study of the antecedents and impacts of ambidexterity in project teams. Proj. Manag. J. 43(6), 97–110 (2012). https://doi.org/10.1002/pmj.21301
G. Bodart, K. Farhat, C. Charlet-Renard, R. Salvatori, V. Geenen, H. Martens, The Somatotrope Growth Hormone-Releasing Hormone/Growth Hormone/Insulin-Like Growth Factor-1 Axis in Immunoregulation and Immunosenescence. Front. Horm. Res. 48, 147–159 (2017). https://doi.org/10.1159/000452913
A. Colao, D. Ferone, P. Marzullo, G. Lombardi, Systemic complications of acromegaly: epidemiology, pathogenesis, and management. Endocr. Rev. 25(1), 102–152 (2004). https://doi.org/10.1210/er.2002-0022
S. Tellatin, P. Maffei, E. Osto, F. Dassie, G. Famoso, R. Montisci, C. Martini, F. Fallo, M.P. Marra, R. Mioni, S. Iliceto, R. Vettor, F. Tona, Coronary microvascular dysfunction may be related to IGF-1 in acromegalic patients and can be restored by therapy. Atherosclerosis 269, 100–105 (2018). https://doi.org/10.1016/j.atherosclerosis.2017.12.019
M.P. Warren, J. Brooks-Gunn, R.P. Fox, C.C. Holderness, E.P. Hyle, W.G. Hamilton, Osteopenia in exercise-associated amenorrhea using ballet dancers as a model: a longitudinal study. J. Clin. Endocrinol. Metab. 87(7), 3162–3168 (2002). https://doi.org/10.1210/jcem.87.7.8637
A. Colao, S. Spiezia, G. Cerbone, R. Pivonello, P. Marzullo, D. Ferone, C. Di Somma, A.P. Assanti, G. Lombardi, Increased arterial intima-media thickness by B-M mode echodoppler ultrasonography in acromegaly. Clin. Endocrinol. (Oxf) 54(4), 515–524 (2001). https://doi.org/10.1046/j.1365-2265.2001.01256.x
I. Kartal, H. Oflaz, B. Pamukçu, M. Meriç, F. Aral, N. Ozbey, F. Alagöl, Investigation of early atherosclerotic changes in acromegalic patients. Int. J. Clin. Pract 64(1), 39–44 (2010). https://doi.org/10.1111/j.1742-1241.2008.01750.x
B.A. Bengtsson, S. Edén, I. Ernest, A. Odén, B. Sjögren, Epidemiology and long-term survival in acromegaly. A study of 166 cases diagnosed between 1955 and 1984. Acta Med. Scand. 223(4), 327–335 (1988). https://doi.org/10.1111/j.0954-6820.1988.tb15881.x
W. Che, N. Lerner-Marmarosh, Q. Huang, M. Osawa, S. Ohta, M. Yoshizumi, M. Glassman, J.D. Lee, C. Yan, B.C. Berk, J. Abe, Insulin-like growth factor-1 enhances inflammatory responses in endothelial cells: role of Gab1 and MEKK3 in TNF-alpha-induced c-Jun and NF-kappaB activation and adhesion molecule expression. Circ. Res. 90(11), 1222–1230 (2002). https://doi.org/10.1161/01.res.0000021127.83364.7d
L. Maione, T. Brue, A. Beckers, B. Delemer, P. Petrossians, F. Borson-Chazot, O. Chabre, P. François, J. Bertherat, C. Cortet-Rudelli, P. Chanson; French Acromegaly Registry Group, Changes in the management and comorbidities of acromegaly over three decades: the French Acromegaly Registry. Eur. J. Endocrinol. 176(5), 645–655 (2017). https://doi.org/10.1530/EJE-16-1064
R. Pivonello, R.S. Auriemma, L.F. Grasso, C. Pivonello, C. Simeoli, R. Patalano, M. Galdiero, A. Colao, Complications of acromegaly: cardiovascular, respiratory and metabolic comorbidities. Pituitary 20(1), 46–62 (2017). https://doi.org/10.1007/s11102-017-0797-7
M. Yang, M. Guo, Y. Hu, Y. Jiang, Scube regulates synovial angiogenesis-related signaling. Med. Hypotheses 81(5), 948–953 (2013). https://doi.org/10.1016/j.mehy.2013.09.001
R.B. Yang, C.K. Ng, S.M. Wasserman, S.D. Colman, S. Shenoy, F. Mehraban, L.G. Komuves, J.E. Tomlinson, J.N. Topper, Identification of a novel family of cell-surface proteins expressed in human vascular endothelium. J. Biol. Chem. 277(48), 46364–46373 (2002). https://doi.org/10.1074/jbc.M207410200
L. Bronze, SCUBE 1: A novel biomarker related to platelet activation and atherothrombosis. SCUBE1 – Um novo biomarcador associado a ativação plaquetária e aterotrombose. Rev. Port. Cardiol. (Engl Ed). 37(5), 383–385 (2018). https://doi.org/10.1016/j.repc.2018.04.004
S. Turkmen, U. Eryigit, Y. Karaca, A. Mentese, U.A. Sumer, E. Yulug, N. Aksut, S. Gazioglu, A. Gunduz, Diagnostic value of plasma signal peptide-Cub-Egf domain-containing protein-1 (SCUBE-1) in an experimental model of acute ischemic stroke. Am. J. Emerg. Med. 33(2), 262–265 (2015). https://doi.org/10.1016/j.ajem.2014.11.051
G.A. Laughlin, E. Barrett-Connor, M.H. Criqui, D. Kritz-Silverstein, The prospective association of serum insulin-like growth factor I (IGF-I) and IGF-binding protein-1 levels with all cause and cardiovascular disease mortality in older adults: the Rancho Bernardo Study. J. Clin. Endocrinol. Metab. 89(1), 114–120 (2004). https://doi.org/10.1210/jc.2003-030967
M. Andreassen, I. Raymond, C. Kistorp, P. Hildebrandt, J. Faber, L.Ø. Kristensen, IGF1 as predictor of all cause mortality and cardiovascular disease in an elderly population. Eur. J. Endocrinol. 160(1), 25–31 (2009). https://doi.org/10.1530/EJE-08-0452
S. Kul, O.T. Caklili, Y. Tutuncu, F.B. Ozcan, F. Aksu, O.F. Baycan, A. Atici, U.Z. Bilgili, M. Takir, M. Caliskan, Endothelial dysfunction in patients with acromegaly and It’s association with Endocan. Growth Horm. IGF Res. 56, 101362 (2021). https://doi.org/10.1016/j.ghir.2020.101362
D.F. Dai, P. Thajeb, C.F. Tu, F.T. Chiang, C.H. Chen, R.B. Yang, J.J. Chen, Plasma concentration of SCUBE1, a novel platelet protein, is elevated in patients with acute coronary syndrome and ischemic stroke. J. Am. Coll. Cardiol. 51(22), 2173–2180 (2008). https://doi.org/10.1016/j.jacc.2008.01.060
H. Uyanikoglu, N.G. Hilali, M. Yardimciel, I. Koyuncu, A new biomarker for the early diagnosis of ovarian torsion: SCUBE-1. Clin. Exp. Reprod. Med. 45(2), 94–99 (2018). https://doi.org/10.5653/cerm.2018.45.2.94
A.A. Capkin, S. Demir, A. Mentese, Ç. Bulut, A. Ayar, Can signal peptide-CUB-EGF domain-containing protein (SCUBE) levels be a marker of angiogenesis in patients with psoriasis? Arch. Dermatol. Res. 309(3), 203–207 (2017). https://doi.org/10.1007/s00403-017-1722-7
T.L.C. Wolters, M.G. Netea, N.P. Riksen, A.R.M.M. Hermus, R.T. Netea-Maier, Acromegaly, inflammation and cardiovascular disease: a review. Rev. Endocr. Metab. Disord. 21(4), 547–568 (2020). https://doi.org/10.1007/s11154-020-09560-x
M.F. Walsh, M. Barazi, G. Pete, R. Muniyappa, J.C. Dunbar, J.R. Sowers, Insulin-like growth factor I diminishes in vivo and in vitro vascular contractility: role of vascular nitric oxide. Endocrinology 137(5), 1798–1803 (1996). https://doi.org/10.1210/endo.137.5.8612517
H. Nishizawa, A.E. Handayaningsih, G. Iguchi, Y. Cho, M. Takahashi, M. Yamamoto, K. Suda, K. Kasahara, F. Hakuno, K. Yamanouchi, M. Nishihara, S. Seino, S. Takahashi, Y. Takahashi, Enhanced oxidative stress in GH-transgenic rat and acromegaly in humans. Growth Horm. IGF Res. 22(2), 64–68 (2012). https://doi.org/10.1016/j.ghir.2012.02.001
H. Ozisik, B.S. Yurekli, A. Suner, O. Copur, E.Y. Sozmen, S.S. Ozbek, A.K. Karabulut, I.Y. Simsir, M. Erdogan, S. Cetinkalp, F. Saygili, High chitotriosidase and AGE levels in acromegaly: a case-control study. Hormones (Athens) 22(1), 61–69 (2023). https://doi.org/10.1007/s42000-022-00409-3
L. Boero, L. Cuniberti, N. Magnani, M. Manavela, V. Yapur, M. Bustos, L. Gómez Rosso, T. Meroño, L. Marziali, L. Viale, P. Evelson, G. Negri, F. Brites, Increased oxidized low density lipoprotein associated with high ceruloplasmin activity in patients with active acromegaly. Clin. Endocrinol. (Oxf) 72(5), 654–660 (2010). https://doi.org/10.1111/j.1365-2265.2009.03685.x
S. Yarman, T.A. Ozden, C. Gökkuşu, The evaluation of lipid peroxidation and acute effect of octreotide on lipid peroxidation in patients with active acromegaly. Clin. Chim. Acta. 336(1-2), 45–48 (2003). https://doi.org/10.1016/s0009-8981(03)00328-0
C. Ozkan, A.E. Altinova, E.T. Cerit, C. Yayla, A. Sahinarslan, D. Sahin, A.S. Dincel, F.B. Toruner, M. Akturk, M. Arslan, Markers of early atherosclerosis, oxidative stress and inflammation in patients with acromegaly. Pituitary 18(5), 621–629 (2015). https://doi.org/10.1007/s11102-014-0621-6
M. Ilhan, S. Turgut, S. Turan, S. Demirci Cekic, H.A. Ergen, G. Korkmaz Dursun, B. Mezani, O. Karaman, I. Yaylim, M.R. Apak, E. Tasan, The assessment of total antioxidant capacity and superoxide dismutase levels, and the possible role of manganese superoxide dismutase polymorphism in acromegaly. Endocr. J. 65(1), 91–99 (2018). https://doi.org/10.1507/endocrj.EJ17-0300
T.L.C. Wolters, C.D.C.C. van der Heijden, N. van Leeuwen, B.T.P. Hijmans-Kersten, M.G. Netea, J.W. Smit, D.H.J. Thijssen, A. Hermus, N.P. Riksen, R. Netea-Maier, Persistent inflammation and endothelial dysfunction in patients with treated acromegaly. Endocr. Connect 8(12), 1553–1567 (2019). https://doi.org/10.1530/EC-19-0430
D.S. Celermajer, K.E. Sorensen, C. Bull, J. Robinson, J.E. Deanfield, Endothelium-dependent dilation in the systemic arteries of asymptomatic subjects relates to coronary risk factors and their interaction. J. Am. Coll. Cardiol. 24(6), 1468–1474 (1994). https://doi.org/10.1016/0735-1097(94)90141-4
P. Maffei, F. Dassie, A. Wennberg, M. Parolin, R. Vettor, The endothelium in acromegaly. Front. Endocrinol. (Lausanne) 10, 437 (2019). https://doi.org/10.3389/fendo.2019.00437
G. Ozkan, S. Ulusoy, A. Menteşe, S.C. Karahan, M. Cansız, New marker of platelet activation, SCUBE1, is elevated in hypertensive patients. Am. J. Hypertens. 26(6), 748–753 (2013). https://doi.org/10.1093/ajh/hpt007
Author information
Authors and Affiliations
Contributions
CRediT authorship contribution statement. Conceptualization: D.T., I.N., S.C.K. Methodology: D.T., I.N., H.C. Data curation: D.T., I.N., H.C., M.K., O.U., C.B., Y.E.G. Formal analysis: S.S.A., S.C.K., S.O.Y., M.C.B. Investigation: S.C.K., M.K., O.U. Project administration: S.S.A. Writing-original draft preparation: D.T., S.O.Y., M.C.B. Writing- review & editing: D.T., M.K., H.C., M.C.B. Supervision: S.S.A., O.U., M.C.B.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Consent to participate
Written informed consent was obtained from each subject following a detailed explanation of the objectives and protocol of the study.
Ethics approval
The study protocol was approved by the Karadeniz Teknik University Ethics Committee (Date of Approval : 30/06/2021, Protocol no:2021/595-24237859).
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Tufekci, D., Nuhoglu, I., Ayan, S.S. et al. Can signal peptide-CUB-EGF domain-containing protein 1 (SCUBE-1) be used as an indicator of endothelial dysfunction in acromegaly patients?. Endocrine 82, 152–160 (2023). https://doi.org/10.1007/s12020-023-03444-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12020-023-03444-1